Scalable Palladium-Catalyzed Synthesis of Phenyl-2-(2'-cyanophenyl)acetylene Intermediates for Pharma
Scalable Palladium-Catalyzed Synthesis of Phenyl-2-(2'-cyanophenyl)acetylene Intermediates for Pharma
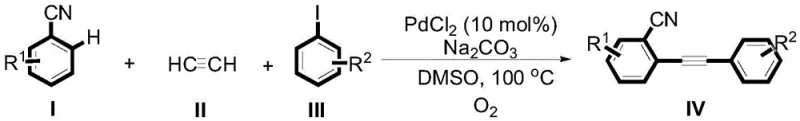
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways for constructing complex alkynyl scaffolds, which serve as critical building blocks for active pharmaceutical ingredients (APIs) and advanced materials. A significant breakthrough in this domain is detailed in Chinese Patent CN111704558A, which discloses a highly efficient, palladium-catalyzed method for preparing phenyl-2-(2'-cyanophenyl)acetylene compounds. This technology represents a paradigm shift from traditional multi-step syntheses to a streamlined, one-pot protocol that utilizes readily available starting materials: benzonitrile derivatives, acetylene gas, and iodobenzene derivatives. By leveraging a catalytic system composed of palladium chloride and sodium carbonate in dimethyl sulfoxide (DMSO) under an oxygen atmosphere, this process achieves remarkable yields ranging from 75% to 93%. For R&D directors and procurement strategists, this patent offers a compelling value proposition: a greener, safer, and economically superior route to high-value nitrile-containing intermediates that are essential for drug discovery and development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phenyl-2-(2'-cyanophenyl)acetylene derivatives has been plagued by significant operational and safety challenges that hinder commercial viability. Conventional routes typically rely on multi-step sequences involving the substitution of cyano groups, which not only prolongs the production timeline but also drastically increases the accumulation of impurities. A critical safety concern associated with these legacy methods is the potential generation of hydrocyanic acid (HCN), a highly toxic volatile gas, particularly under acidic or specific reaction conditions. Managing HCN requires specialized containment infrastructure, rigorous safety protocols, and expensive scrubbing systems, all of which inflate capital expenditure (CAPEX) and operational expenditure (OPEX). Furthermore, traditional metal-catalyzed approaches often suffer from low atom economy and require harsh reaction conditions that limit substrate scope, making them unsuitable for sensitive pharmaceutical intermediates. The cumulative effect of these inefficiencies is a supply chain vulnerable to delays, regulatory scrutiny, and unpredictable cost fluctuations.
The Novel Approach
In stark contrast, the methodology described in patent CN111704558A introduces a transformative one-pot strategy that elegantly bypasses these historical bottlenecks. By directly coupling benzonitrile, acetylene, and iodobenzene derivatives in a single vessel, the process eliminates the need for isolating unstable intermediates, thereby reducing unit operations and solvent consumption. The use of molecular oxygen as part of the reaction atmosphere serves as a clean, abundant oxidant that facilitates the catalytic cycle without generating heavy metal waste streams typical of stoichiometric oxidants. This approach not only enhances the overall yield—demonstrated by examples achieving up to 93% conversion—but also significantly improves the purity profile of the crude product. The versatility of this system is further evidenced by its compatibility with a wide array of functional groups, including trifluoromethyl, methoxy, and heterocyclic moieties, enabling the rapid synthesis of diverse chemical libraries for drug screening.
Mechanistic Insights into PdCl2-Catalyzed Oxidative Coupling
The core of this technological advancement lies in the sophisticated interplay between the palladium catalyst and the reaction components under oxidative conditions. The mechanism likely initiates with the oxidative addition of the aryl iodide (Component III) to the active palladium species, forming an aryl-palladium(II) intermediate. Simultaneously, the terminal alkyne (acetylene, Component II) undergoes activation, potentially facilitated by the basic environment provided by sodium carbonate, to form a copper-free or palladium-bound acetylide species. The subsequent migratory insertion of the alkyne into the palladium-aryl bond constructs the crucial carbon-carbon triple bond linkage. Uniquely, the presence of oxygen suggests a re-oxidation pathway that regenerates the active palladium catalyst, ensuring high turnover numbers (TON) and minimizing the required catalyst loading to just 10 mol%. This catalytic efficiency is paramount for commercial scale-up, as it reduces the residual metal burden in the final API, simplifying downstream purification processes.
From an impurity control perspective, the choice of sodium carbonate as a mild base is strategic. Unlike stronger bases that might promote side reactions such as homocoupling of the alkyne (Glaser coupling) or hydrolysis of the nitrile group, sodium carbonate maintains a pH window that favors the desired cross-coupling while suppressing degradation pathways. The solvent, DMSO, plays a dual role: it solubilizes the inorganic base and organic substrates effectively, and its high boiling point allows the reaction to proceed at 100°C, providing the necessary thermal energy to overcome activation barriers without requiring pressurized reactors. This mechanistic robustness ensures that even with electron-deficient or sterically hindered substrates, the reaction proceeds with high fidelity, delivering products with consistent quality suitable for stringent pharmaceutical specifications.
How to Synthesize Phenyl-2-(2'-cyanophenyl)acetylene Efficiently
Implementing this synthesis in a GMP environment requires precise adherence to the optimized parameters established in the patent data to ensure reproducibility and safety. The process is designed to be operationally simple, requiring standard glass-lined or stainless steel reactors capable of handling mild pressure from acetylene gas and oxygen. The key to success lies in the controlled addition of reagents and the maintenance of the oxygen atmosphere throughout the 10-hour heating period. Detailed standard operating procedures (SOPs) regarding the quenching of excess acetylene and the removal of palladium residues are essential for compliance. For a comprehensive understanding of the practical execution, the following guide outlines the standardized synthesis steps derived from the patent examples.
- Charge a reactor with benzonitrile derivative, acetylene gas, and iodobenzene derivative in a molar ratio of approximately 1: 1.2:1.
- Add palladium chloride catalyst (10 mol%), sodium carbonate base, and dimethyl sulfoxide (DMSO) solvent to the mixture.
- Heat the reaction to 100°C under an oxygen atmosphere for 10 hours, followed by extraction and silica gel chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this palladium-catalyzed protocol translates into tangible strategic benefits that extend beyond mere chemical yield. The consolidation of multiple synthetic steps into a single one-pot operation fundamentally alters the cost structure of manufacturing these intermediates. By eliminating intermediate isolation, drying, and purification stages, manufacturers can drastically reduce solvent usage, energy consumption, and labor hours. This streamlining effect leads to substantial cost savings in raw material procurement and waste disposal, making the final product more price-competitive in the global market. Furthermore, the reliance on commodity chemicals like acetylene, iodobenzene derivatives, and sodium carbonate ensures a stable supply base, insulating the production schedule from the volatility associated with exotic or custom-synthesized reagents.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by its high atom economy and reduced unit operations. Traditional multi-step routes incur costs at every stage of workup and purification; by collapsing these into a single reaction vessel, the process minimizes equipment occupancy time and utility usage. Additionally, the avoidance of toxic byproducts like hydrocyanic acid removes the need for expensive hazardous waste treatment facilities and specialized safety monitoring systems. The use of a relatively low loading of palladium chloride (10 mol%) combined with the potential for catalyst recovery further optimizes the cost per kilogram, allowing for significant margin improvement in high-volume production scenarios without compromising quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and flexibility of this catalytic system. The broad substrate tolerance means that a single manufacturing platform can produce a wide variety of analogues (e.g., compounds 4a through 4h) simply by swapping the starting iodobenzene or benzonitrile. This flexibility allows manufacturers to respond rapidly to changing demand from R&D clients without retooling entire production lines. Moreover, the reagents involved are commercially available in bulk quantities from multiple global suppliers, reducing the risk of single-source bottlenecks. The simplified workflow also shortens the overall lead time from order to delivery, enabling faster turnaround for clinical trial materials and commercial batches.
- Scalability and Environmental Compliance: Scaling this reaction from gram to tonnage is facilitated by the use of standard solvents and mild conditions (100°C, atmospheric or slight pressure). The absence of cryogenic conditions or highly pyrophoric reagents simplifies the engineering requirements for large-scale reactors. From an environmental standpoint, the process aligns with green chemistry principles by utilizing oxygen as a benign oxidant and generating minimal hazardous waste. This compliance profile eases the regulatory burden for environmental permits and supports corporate sustainability goals. The ability to run the reaction in DMSO, a solvent with well-established recovery and recycling protocols, further enhances the environmental footprint, making it an attractive option for companies aiming to reduce their carbon intensity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and claims within CN111704558A, providing clarity on the feasibility and advantages of the method for potential partners and licensees. Understanding these nuances is critical for evaluating the technology's fit within existing manufacturing portfolios.
Q: What are the primary safety advantages of this palladium-catalyzed method over traditional routes?
A: Traditional methods often involve multi-step substitutions that can generate highly toxic hydrocyanic acid (HCN) as a byproduct. This novel one-pot protocol operates under mild alkaline conditions using sodium carbonate, significantly mitigating the risk of toxic gas generation and enhancing operational safety for large-scale manufacturing.
Q: Does this catalytic system support heterocyclic substrates?
A: Yes, the patent data demonstrates excellent functional group tolerance. The system successfully couples heterocyclic iodides such as 2-iodopyridine, 2-iodothiophene, and quinoline derivatives, yielding products like 2-(pyridin-2-ylethynyl)benzonitrile with high efficiency (up to 93% yield).
Q: How does the use of oxygen affect the reaction economics?
A: Utilizing oxygen as the terminal oxidant or atmosphere eliminates the need for expensive stoichiometric oxidants or complex ligand systems often required in palladium cycles. This simplifies the reagent profile, reduces raw material costs, and minimizes waste generation, aligning with green chemistry principles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenyl-2-(2'-cyanophenyl)acetylene Supplier
The technological potential of the palladium-catalyzed synthesis of phenyl-2-(2'-cyanophenyl)acetylene compounds is immense, offering a pathway to high-purity intermediates essential for next-generation therapeutics. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate this laboratory-scale innovation into commercial reality. As a seasoned CDMO partner, we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facility is equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch meets the exacting standards required by global regulatory bodies. We understand the critical nature of these intermediates in your value chain and are committed to delivering reliability.
We invite you to collaborate with us to leverage this advanced synthetic route for your specific projects. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this process can optimize your budget. We encourage you to contact our technical procurement team today to request specific COA data for related compounds and to discuss route feasibility assessments for your target molecules. Let us be your partner in driving efficiency and innovation in your chemical supply chain.
