Scalable Metal-Free Synthesis of N-phenyl-3-(phenyldisulfanyl)propionamide for Pharmaceutical Applications
Scalable Metal-Free Synthesis of N-phenyl-3-(phenyldisulfanyl)propionamide for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex sulfur-containing scaffolds, particularly asymmetric disulfides which serve as pivotal motifs in bioactive natural products and drug candidates. Patent CN111777536A, published in October 2020, discloses a groundbreaking synthetic strategy for preparing N-phenyl-3-(phenyldisulfanyl)propionamide, a valuable asymmetric disulfide intermediate. This innovation represents a significant departure from traditional protocols by employing a metal-free oxidative coupling system that utilizes readily available sulfides and symmetrical disulfides. The technical breakthrough lies in the identification of N-fluorobisbenzenesulfonamide (NFSI) as a highly specific additive that drives the reaction efficiency without the need for toxic transition metals or hazardous thiol reagents. For R&D directors and process chemists, this patent offers a compelling alternative route that simplifies purification workflows and enhances the overall safety profile of the manufacturing process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of asymmetric disulfide bonds has been plagued by significant operational and safety challenges that hinder large-scale adoption in commercial manufacturing. The most prevalent classical approach involves the oxidative coupling of two different thiols; however, this method suffers from severe drawbacks including the generation of unpleasant odors and the formation of symmetric homocoupling byproducts which are difficult to separate. Furthermore, alternative strategies relying on transition metal catalysis, while effective in some contexts, introduce heavy metal contaminants that require rigorous and costly removal processes to meet stringent pharmaceutical purity standards. These legacy methods often necessitate specialized equipment for odor containment and complex downstream processing to scavenge residual metals, thereby inflating production costs and extending lead times for high-purity pharmaceutical intermediates.
The Novel Approach
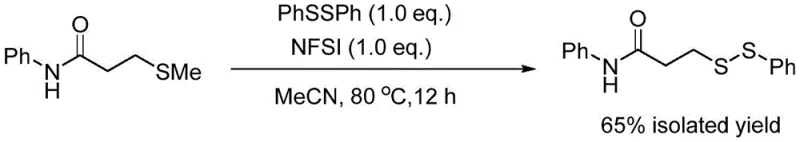
In stark contrast to these cumbersome traditional techniques, the methodology outlined in the patent utilizes a direct, one-pot oxidative cross-coupling between 3-methylthio-N-phenylpropionamide and diphenyl disulfide. This novel pathway leverages the stability and low odor profile of sulfide precursors, effectively bypassing the safety hazards associated with volatile thiols. The reaction proceeds smoothly in acetonitrile at moderate temperatures, driven by the unique reactivity of NFSI which activates the sulfur center for nucleophilic attack by the disulfide species. This streamlined approach not only simplifies the reaction setup but also ensures a cleaner impurity profile by avoiding metal catalysts entirely. The visual representation of this efficient transformation highlights the direct conversion of stable starting materials into the target asymmetric disulfide with impressive selectivity.
Mechanistic Insights into NFSI-Mediated Electrophilic Sulfur Activation
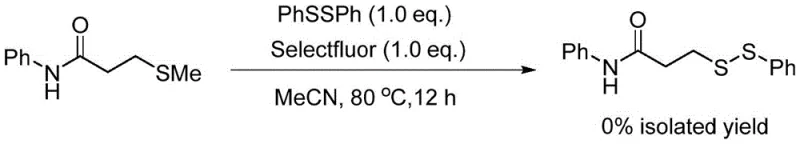
The success of this synthetic route hinges on the precise mechanistic role of N-fluorobisbenzenesulfonamide (NFSI) as an electrophilic activator within the reaction medium. Unlike generic oxidants, NFSI appears to facilitate the generation of a reactive sulfenium species from the methylthio precursor, which is subsequently intercepted by the nucleophilic sulfur atom of the diphenyl disulfide. This specific activation mode prevents the random radical scrambling often seen in other oxidative conditions, thereby favoring the formation of the desired unsymmetrical bond over symmetric byproducts. The mechanistic elegance of this system is further underscored by control experiments which demonstrate that substituting NFSI with other common electrophilic fluorinating agents, such as Selectfluor, results in a complete failure of the reaction. This specificity suggests that the steric and electronic properties of the bisbenzenesulfonamide moiety are critical for stabilizing the transition state and enabling the sulfur-sulfur bond formation under mild thermal conditions.
From an impurity control perspective, the absence of transition metals fundamentally alters the purification landscape for this intermediate. In metal-catalyzed processes, trace amounts of palladium, copper, or iron can coordinate with sulfur atoms, creating stable complexes that are notoriously difficult to remove and can poison downstream biological assays. By utilizing an organocatalytic-like oxidative system, the resulting crude mixture contains primarily organic byproducts that are more amenable to standard separation techniques like chromatography or crystallization. This inherent cleanliness of the reaction matrix reduces the burden on quality control laboratories and minimizes the risk of metal-induced degradation of the final active pharmaceutical ingredient. Consequently, this method aligns perfectly with the industry's push towards greener chemistry and stricter regulatory limits on elemental impurities in drug substances.
How to Synthesize N-phenyl-3-(phenyldisulfanyl)propionamide Efficiently
The practical implementation of this synthesis is designed for straightforward execution in standard laboratory or pilot plant settings, requiring no specialized high-pressure or cryogenic equipment. The protocol dictates the sequential addition of the sulfide substrate, the symmetrical disulfide coupling partner, and the NFSI additive into a reaction vessel containing acetonitrile solvent. Maintaining a precise 1:1:1 molar ratio is essential to maximize atom economy and minimize the formation of unreacted starting materials. The mixture is then heated to 80°C and agitated vigorously for a duration of 12 hours to ensure full conversion. Following the reaction period, the workup involves a simple concentration step followed by purification, typically via column chromatography, to isolate the target pale yellow solid. For detailed standardized operating procedures and safety data sheets regarding this specific transformation, please refer to the technical guidelines below.
- Charge a sealed tube with acetonitrile solvent, 3-methylthio-N-phenylpropionamide, diphenyl disulfide, and N-fluorobisbenzenesulfonamide (NFSI) in a 1: 1:1 molar ratio.
- Heat the reaction mixture to 80°C and stir vigorously for 12 hours to ensure complete conversion to the asymmetric disulfide product.
- Upon completion, concentrate the reaction solution under reduced pressure and purify the crude residue via column chromatography to isolate the pale yellow solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthetic route offers tangible strategic benefits that extend beyond mere chemical yield. The elimination of transition metal catalysts removes a significant cost center associated with the purchase of expensive noble metals and the subsequent disposal or recycling of metal-laden waste streams. Furthermore, the substitution of malodorous thiols with stable, solid sulfide reagents drastically simplifies facility requirements, as there is no longer a need for extensive scrubbing systems or specialized containment infrastructure to manage noxious vapors. This shift translates directly into lower capital expenditure for new production lines and reduced operational overhead for existing facilities, making the manufacturing of complex sulfur-containing intermediates more economically viable.
- Cost Reduction in Manufacturing: The economic impact of removing transition metals from the process cannot be overstated, as it eliminates the need for costly scavenging resins and extensive analytical testing for heavy metal residues. Additionally, the use of commodity chemicals like diphenyl disulfide and NFSI, which are available in bulk quantities from multiple global suppliers, ensures competitive pricing and reduces dependency on single-source specialty reagents. The simplified workup procedure also reduces solvent consumption and labor hours required for purification, contributing to substantial overall cost savings in the production of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: By relying on robust, shelf-stable starting materials rather than sensitive thiols or air-sensitive metal catalysts, the supply chain becomes significantly more resilient to disruptions. The reagents employed in this method are not subject to the same strict transportation regulations as hazardous malodorous compounds, facilitating smoother logistics and faster delivery times. This stability allows for larger batch sizes and longer storage periods without degradation, enabling manufacturers to maintain strategic stockpiles and ensuring continuous supply for critical drug development programs without the risk of raw material spoilage.
- Scalability and Environmental Compliance: The benign nature of the reagents and the absence of toxic metals make this process inherently scalable and environmentally compliant. Waste streams generated from this reaction are easier to treat and dispose of compared to those containing heavy metals, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The mild reaction conditions (80°C) also reduce energy consumption compared to high-temperature or high-pressure alternatives, further enhancing the green chemistry profile of the manufacturing process and reducing the carbon footprint of the final product.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the scalability, safety, and regulatory compliance of this novel asymmetric disulfide synthesis method. These insights are derived directly from the experimental data and comparative studies presented in the patent documentation, providing clarity for technical teams evaluating this route for commercial adoption. Understanding these nuances is critical for assessing the feasibility of integrating this technology into existing manufacturing portfolios.
Q: Why is NFSI critical for this asymmetric disulfide synthesis compared to other oxidants?
A: Experimental data indicates that alternative oxidants like Selectfluor fail to promote the reaction, yielding 0% product. NFSI specifically facilitates the electrophilic activation of the sulfide sulfur without causing decomposition or side reactions common with harsher fluorinating agents.
Q: Does this process require transition metal catalysts?
A: No, the method described in patent CN111777536A is entirely metal-free. It utilizes an organic oxidative system (NFSI) rather than traditional transition metal catalysts, eliminating the need for expensive metal scavenging steps and reducing heavy metal impurities in the final API intermediate.
Q: How does this method address the safety issues associated with thiol-based synthesis?
A: Traditional methods often rely on volatile and malodorous thiols which pose significant occupational health hazards. This novel route uses stable, odorless sulfide and disulfide starting materials, significantly improving the working environment and simplifying waste gas treatment protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-phenyl-3-(phenyldisulfanyl)propionamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic routes that balance efficiency with regulatory compliance. Our team of expert process chemists has extensively evaluated the methodology described in CN111777536A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of N-phenyl-3-(phenyldisulfanyl)propionamide meets the exacting standards required for pharmaceutical applications. Our commitment to quality assurance means that we can deliver this complex intermediate with a consistent impurity profile suitable for direct use in downstream API synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this metal-free technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your R&D timelines and production budgets.
