Scalable Synthesis of Polychlorinated Diphenyl Sulfides via Advanced Palladium Catalysis for Global Supply Chains
Scalable Synthesis of Polychlorinated Diphenyl Sulfides via Advanced Palladium Catalysis for Global Supply Chains
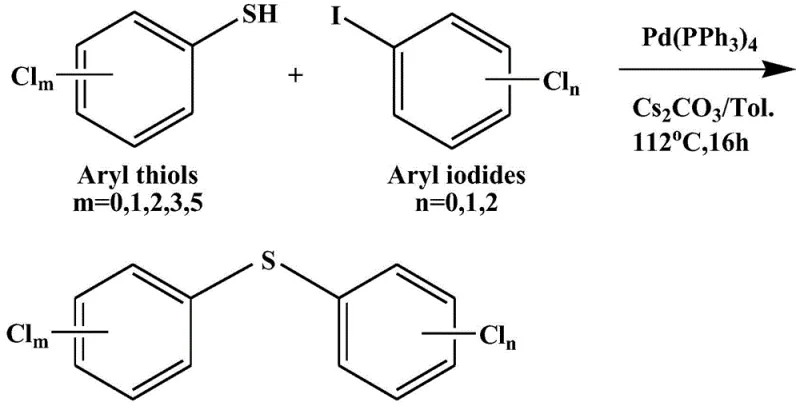
The development of efficient synthetic routes for sulfur-containing aromatic compounds remains a critical challenge in modern fine chemical manufacturing, particularly for high-value intermediates used in pharmaceuticals and agrochemicals. Patent CN113880739A introduces a significant technological advancement in this domain by detailing a robust method for synthesizing polychlorinated diphenyl sulfide compounds. This innovation addresses long-standing issues regarding catalyst poisoning and byproduct formation that have historically plagued the production of these essential molecular scaffolds. By utilizing a specific palladium catalyst system in conjunction with cesium carbonate, the disclosed method achieves high selectivity and yield under relatively mild thermal conditions. For R&D directors and procurement specialists, this represents a viable pathway to secure reliable supplies of complex sulfide intermediates without the baggage of cumbersome purification protocols associated with legacy technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of diaryl sulfides has relied heavily on nucleophilic aromatic substitution or copper-catalyzed coupling reactions, both of which present substantial drawbacks for large-scale commercial production. Traditional methods often utilize metallic copper or monovalent copper salts in highly polar solvents such as DMSO or DMF at elevated temperatures ranging from 80°C to 120°C. A major technical failure point in these copper-mediated processes is the propensity for oxidative dimerization of the thiol starting material, leading to the formation of diphenyl disulfide impurities. These disulfide byproducts possess physical properties similar to the target sulfide, making their removal via standard crystallization or distillation extremely difficult and costly. Furthermore, alternative nickel-based catalytic systems, while active, often involve ligands that are difficult to source commercially and may introduce heavy metal contamination risks that are unacceptable for pharmaceutical grade intermediates.
The Novel Approach
The methodology outlined in CN113880739A circumvents these historical bottlenecks by employing a palladium-catalyzed cross-coupling strategy that is specifically optimized to prevent catalyst deactivation by sulfur. The core innovation lies in the use of tetrakis(triphenylphosphine)palladium(0) [Pd(PPh3)4] paired with cesium carbonate (Cs2CO3) as the base in a toluene solvent system. This specific combination allows for the efficient formation of the carbon-sulfur (C-S) bond between chlorothiophenols and chloroiodobenzenes without generating significant amounts of disulfide side products. The reaction proceeds smoothly under reflux conditions, typically around 110°C, which is energetically favorable compared to more extreme conditions required by other methods. This novel approach not only simplifies the reaction profile but also drastically reduces the burden on downstream processing teams, as the absence of stubborn disulfide impurities means purification can be achieved through standard chromatographic and recrystallization techniques.
Mechanistic Insights into Pd-Catalyzed C-S Bond Formation
From a mechanistic perspective, the success of this synthesis relies on the delicate balance between oxidative addition and reductive elimination within the palladium catalytic cycle, managed effectively by the triphenylphosphine ligands. The cycle initiates with the oxidative addition of the aryl iodide substrate to the Pd(0) species, forming an aryl-palladium(II)-iodide complex. This step is facilitated by the electron-rich nature of the phosphine ligands, which stabilize the metal center against aggregation. Subsequently, the chlorothiophenol, activated by the cesium carbonate base to form a thiolate anion, undergoes transmetallation with the palladium complex. This step is critical; the use of cesium carbonate is particularly advantageous due to its high solubility in organic media and its ability to effectively deprotonate the thiol without promoting unwanted side reactions. The resulting aryl-palladium-thiolate intermediate then undergoes reductive elimination to forge the C-S bond, releasing the polychlorinated diphenyl sulfide product and regenerating the active Pd(0) catalyst for the next turnover.
Impurity control is inherently built into this catalytic system through the suppression of homocoupling pathways. In many sulfur couplings, the thiolate species can oxidize to form disulfides, or the aryl halide can undergo homocoupling to form biaryls. However, the specific stoichiometry and the choice of the soft palladium center favor the hetero-coupling between the distinct aryl iodide and aryl thiol partners. The patent data indicates that by maintaining an inert atmosphere (nitrogen or argon) and strictly controlling the reaction temperature between 105°C and 115°C, the formation of these off-target species is minimized. This high level of chemoselectivity is paramount for R&D teams aiming to produce reference standards or active ingredients where the impurity profile must be tightly controlled to meet regulatory specifications for agrochemical or pharmaceutical applications.
How to Synthesize Polychlorinated Diphenyl Sulfide Efficiently
The operational procedure for this synthesis is designed to be straightforward and adaptable to various scales of production, from gram-scale laboratory optimization to multi-kilogram pilot runs. The process begins by charging the chlorothiophenol substrate into toluene, followed by the sequential addition of the base, catalyst, and the chloroiodobenzene coupling partner. The reaction mixture is then heated to reflux for a period of 12 to 16 hours, ensuring complete consumption of the starting materials. Upon completion, the workup involves a simple aqueous wash to remove inorganic salts, followed by extraction and drying. The detailed standardized synthesis steps, including specific molar ratios and purification parameters, are provided in the guide below to assist technical teams in replicating these results.
- Charge a reactor with chlorothiophenol substrate and toluene solvent under inert atmosphere, followed by the addition of cesium carbonate base and tetrakis(triphenylphosphine)palladium(0) catalyst.
- Introduce the chloroiodobenzene coupling partner and heat the reaction mixture to reflux temperatures between 105-115°C for 12 to 16 hours to ensure complete conversion.
- Perform aqueous workup by washing with water, extract the organic phase with petroleum ether, dry over magnesium sulfate, and purify the crude product via silica gel chromatography and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this palladium-catalyzed route offers distinct strategic advantages over legacy copper or nickel-based methods, primarily driven by simplified processing and improved material throughput. The elimination of disulfide byproducts means that the yield of the desired product is not eroded by side reactions, leading to a more efficient utilization of raw materials. Furthermore, the use of toluene as a solvent is a significant logistical benefit; unlike high-boiling polar solvents like DMSO or DMF, toluene is easily recovered and recycled via distillation, reducing both solvent purchase costs and waste disposal fees. This solvent switch also facilitates easier product isolation, as the product can often be crystallized directly or with minimal anti-solvent addition, streamlining the manufacturing timeline.
- Cost Reduction in Manufacturing: The economic viability of this process is enhanced by the high catalytic efficiency and the avoidance of expensive or difficult-to-remove impurities. By preventing the formation of disulfide dimers, the process eliminates the need for complex preparative HPLC or multiple recrystallization steps that are often required to purge these specific contaminants in copper-catalyzed routes. Additionally, the catalyst loading is optimized to be effective at low concentrations, minimizing the cost contribution of the precious metal palladium. The overall simplification of the workup procedure translates directly into reduced labor hours and lower utility consumption per kilogram of product produced.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically various chlorothiophenols and chloroiodobenzenes, are commodity chemicals with well-established global supply chains. Unlike specialized nickel ligands or unstable copper salts that may face availability fluctuations, these substrates are readily sourced from multiple vendors, mitigating the risk of supply disruption. The robustness of the reaction conditions—tolerating standard reflux temperatures and common inert gases—means that the process can be transferred easily between different manufacturing sites or CDMO partners without requiring specialized equipment modifications, ensuring consistent supply continuity.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this method aligns well with modern green chemistry initiatives. The replacement of toxic polar aprotic solvents with toluene reduces the environmental footprint of the manufacturing process. Moreover, the high selectivity of the reaction minimizes the generation of hazardous waste streams associated with byproduct removal. The scalability is proven by the straightforward nature of the unit operations involved—heating, stirring, washing, and filtering—which are easily replicated in large-scale glass-lined reactors, allowing for seamless transition from pilot scale to commercial tonnage production without unforeseen engineering hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on process capabilities and product quality. Understanding these details is crucial for technical evaluators assessing the feasibility of integrating this route into existing production portfolios.
Q: Why is the Pd(PPh3)4/Cs2CO3 system preferred over copper catalysts for this synthesis?
A: Traditional copper-catalyzed methods often suffer from the formation of diphenyl disulfide byproducts (-S-S- bonds), which complicate purification. The palladium system described in CN113880739A selectively forms the C-S bond with higher specificity and avoids these oxidative dimerization side reactions.
Q: What are the typical reaction conditions for scaling this process?
A: The process operates under mild reflux conditions (105-115°C) in toluene, a standard industrial solvent. It requires an inert gas environment (nitrogen or argon) and runs for approximately 14 hours, making it highly compatible with standard glass-lined or stainless steel reactors used in fine chemical manufacturing.
Q: How is the final purity of the polychlorinated diphenyl sulfide ensured?
A: The patent outlines a rigorous purification protocol involving aqueous washing to remove inorganic salts, followed by silica gel column chromatography using petroleum ether. A final recrystallization step using an n-hexane and dichloromethane mixture ensures the removal of trace impurities and catalyst residues, yielding high-purity crystals suitable for sensitive applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polychlorinated Diphenyl Sulfide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation pharmaceuticals and agrochemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of polychlorinated diphenyl sulfide meets the exacting standards required for sensitive applications such as eosinophilic disease treatments or advanced insecticide formulations.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this palladium-catalyzed method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a secure and efficient supply of these vital chemical building blocks.
