Advanced Microwave-Assisted Synthesis of 2'-Substituted Benzothiazoles for Commercial Scale-up
The landscape of heterocyclic chemistry is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic pathways, particularly for high-value scaffolds like 2'-substituted benzothiazoles. As detailed in the recent patent disclosure CN116217513A, a novel preparation method has emerged that fundamentally shifts the paradigm from traditional organic solvent-based systems to an aqueous microwave-assisted protocol. This technological breakthrough addresses critical pain points in the synthesis of these versatile compounds, which serve as essential building blocks for luminescent materials, molecular probes, and a vast array of pharmaceutical agents exhibiting antibacterial, antitumor, and antimalarial activities. By leveraging pure water as the sole solvent and employing a minimal amount of commercially available phase transfer catalyst, this innovation achieves reaction completion within 15 minutes with yields consistently exceeding 86%, marking a substantial leap forward in process intensification for fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2'-substituted benzothiazoles has relied heavily on condensation reactions involving 2-aminobenzenethiol and various carbonyl sources such as orthoesters, carboxylic acids, esters, or aldehydes, often conducted in hazardous organic solvents like toluene or o-dichlorobenzene. As illustrated in prior art reaction schemes, these conventional routes frequently necessitate the use of strong Bronsted acids or complex, expensive catalysts that require rigorous preparation involving corrosive substances, thereby increasing both the operational risk and the environmental footprint of the process. 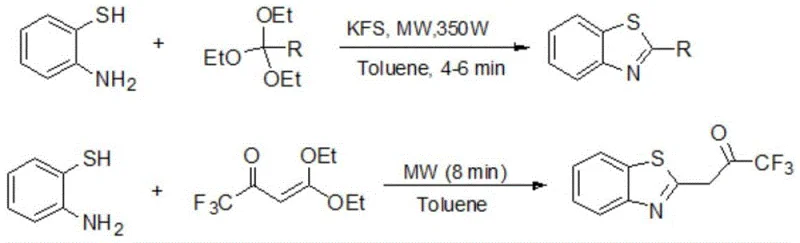
Furthermore, the downstream processing associated with these traditional methods is notoriously cumbersome, typically requiring organic solvent extraction, washing with saturated brine, drying over anhydrous sodium sulfate, and often purification via column chromatography or recrystallization. These additional steps not only consume significant energy and time but also generate substantial volumes of chemical waste, creating a bottleneck for cost reduction in API manufacturing and complicating the supply chain for high-purity pharmaceutical intermediates. The reliance on volatile organic compounds (VOCs) also poses serious regulatory compliance challenges regarding emissions and worker safety, making these legacy processes increasingly unsustainable for modern industrial applications.
The Novel Approach
In stark contrast, the methodology disclosed in CN116217513A introduces a streamlined, eco-friendly pathway that utilizes water as the reaction medium, effectively eliminating the need for toxic organic solvents and the complex catalyst preparation steps associated with older techniques. The core of this innovation lies in the synergistic combination of microwave irradiation and phase transfer catalysis, which allows for the rapid and efficient condensation of 2-aminobenzenethiol with aldehydes under mild conditions. 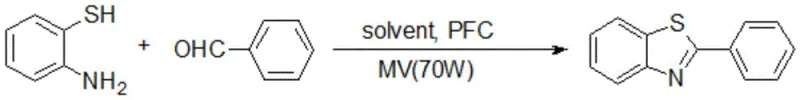
This novel approach ensures that the reactants are effectively dispersed in boiling water, absorbing microwave energy to reach the target temperature of 100°C rapidly, thereby accelerating the reaction kinetics to completion in merely 15 minutes. The simplicity of the workup procedure, which involves simply pouring the reaction mixture into ice water to precipitate the product followed by filtration and drying, represents a drastic simplification of the manufacturing workflow. This shift not only enhances the overall atom economy by producing only water as a byproduct alongside the target molecule but also significantly lowers the barrier for commercial scale-up of complex heterocyclic intermediates by removing the dependency on specialized purification infrastructure.
Mechanistic Insights into Microwave-Assisted Phase Transfer Catalysis
The efficacy of this synthetic route is underpinned by the precise interplay between dielectric heating and phase transfer mechanisms, which together overcome the inherent solubility limitations of organic reactants in an aqueous environment. The phase transfer catalyst, typically a quaternary ammonium salt such as tetrabutylammonium bromide (TBAB) used at a molar ratio of approximately 2% relative to the thiol, acts as a molecular shuttle that transports the hydrophobic 2-aminobenzenethiol and aldehyde species into the aqueous phase where the reaction occurs. This facilitates a homogeneous-like reaction environment despite the heterogeneous nature of the initial mixture, ensuring that the nucleophilic attack of the amino group on the carbonyl carbon proceeds with high efficiency and selectivity.
From an impurity control perspective, the use of water as a solvent and the specific stoichiometric balance of reactants (1:1 molar ratio) play a crucial role in minimizing side reactions. Unlike acidic conditions that might promote polymerization or degradation of sensitive functional groups, the neutral to slightly basic environment facilitated by the phase transfer catalyst preserves the integrity of the substrates. The microwave irradiation provides uniform internal heating, preventing local hot spots that could lead to thermal decomposition, thus ensuring a clean impurity profile that is critical for reliable pharmaceutical intermediate supplier standards. The structural versatility of this method is further evidenced by its compatibility with a wide range of aldehydes, including aromatic aldehydes with various substituents and aliphatic aldehydes, as defined by the general structural formulas where R1 and R2 can be hydrogen, hydroxyl, alkyl, or alkoxy groups. 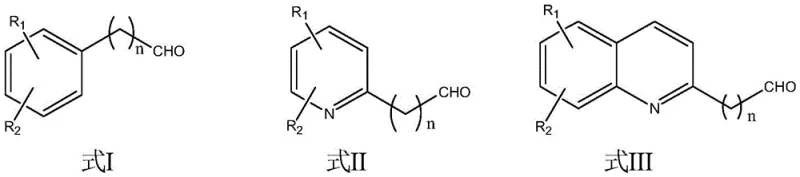
How to Synthesize 2'-Substituted Benzothiazole Efficiently
The practical implementation of this technology involves a straightforward protocol that can be easily adapted for both laboratory optimization and pilot-scale production. The process begins with the preparation of the reaction mixture in a standard round-bottom flask, where distilled water serves as the green solvent matrix. To this, the key reactants, 2-aminobenzenethiol and the specific aldehyde derivative, are added along with the catalytic amount of the phase transfer agent. The detailed standardized synthesis steps are outlined in the guide below to ensure reproducibility and optimal yield.
- Prepare the reaction mixture by adding 2-aminobenzenethiol, a commercially available phase transfer catalyst (such as TBAB), and the selected aldehyde into distilled water within a round-bottom flask.
- Place the flask into a microwave reactor, set the temperature to 100°C, and irradiate at 70W power for approximately 15 minutes while stirring.
- Upon completion, pour the reaction mixture into ice water to precipitate the solid product, then filter, wash with distilled water, and dry to obtain the high-purity target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this water-based microwave synthesis offers compelling strategic benefits that extend beyond mere technical feasibility. The elimination of expensive and hazardous organic solvents directly translates to a reduction in raw material procurement costs and lowers the logistical burden associated with the storage and handling of flammable liquids. Moreover, the simplified workup procedure, which avoids energy-intensive distillation and complex chromatographic separation, drastically reduces the utility consumption and labor hours required per batch, leading to substantial cost savings in manufacturing operations without compromising on product quality.
- Cost Reduction in Manufacturing: The economic advantage of this process is primarily derived from the substitution of high-cost organic solvents and complex catalysts with inexpensive, commodity-grade water and readily available quaternary ammonium salts. By removing the need for column chromatography and extensive solvent recovery systems, the operational expenditure is significantly lowered, allowing for more competitive pricing strategies in the global market for fine chemical intermediates. Additionally, the high conversion rates and selectivity minimize the loss of valuable starting materials, further enhancing the overall cost-efficiency of the production cycle.
- Enhanced Supply Chain Reliability: The reliance on universally available raw materials such as water and common aldehydes mitigates the risk of supply disruptions that often plague specialty chemical markets dependent on niche reagents. The robustness of the reaction conditions, which tolerate a broad scope of substrates including those with sensitive functional groups, ensures consistent production output even when slight variations in feedstock quality occur. This stability is crucial for maintaining continuous supply lines to downstream pharmaceutical manufacturers who demand rigorous adherence to delivery schedules and quality specifications.
- Scalability and Environmental Compliance: From a sustainability standpoint, the use of water as a solvent aligns perfectly with increasingly stringent environmental regulations regarding VOC emissions and waste disposal. The process generates minimal hazardous waste, primarily consisting of aqueous filtrates that are easier to treat than organic solvent mixtures, thereby reducing the environmental compliance costs for the manufacturing facility. The scalability of microwave technology, combined with the simplicity of the filtration-based isolation method, facilitates a smooth transition from gram-scale development to multi-ton commercial production, ensuring long-term viability and regulatory approval.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology, providing clarity on its operational parameters and strategic value for industry stakeholders. These insights are derived directly from the experimental data and process descriptions found in the patent literature, ensuring accuracy and relevance for decision-makers evaluating this methodology for their own production portfolios.
Q: What are the primary advantages of using water as a solvent in this benzothiazole synthesis?
A: Using water eliminates the need for toxic organic solvents like toluene or o-dichlorobenzene, significantly reducing environmental impact and disposal costs. Furthermore, water acts as an excellent medium for microwave absorption, facilitating rapid heating and improved reaction kinetics without requiring complex catalyst preparation.
Q: How does the phase transfer catalyst improve the reaction efficiency?
A: The phase transfer catalyst, such as tetrabutylammonium bromide (TBAB), facilitates the interaction between the organic reactants and the aqueous phase. This ensures effective dispersion of the hydrophobic 2-aminobenzenethiol and aldehydes in boiling water, leading to higher conversion rates and selectivity within a short 15-minute timeframe.
Q: Is this method suitable for large-scale commercial production of pharmaceutical intermediates?
A: Yes, the method is highly scalable due to its simplicity and safety profile. The absence of corrosive strong acids and the use of readily available, low-cost catalysts make it economically viable for industrial scale-up, while the simple filtration workup reduces processing time compared to traditional column chromatography methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2'-Substituted Benzothiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this water-based microwave synthesis technology in reshaping the supply chain for critical heterocyclic intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry approach are fully realized in a GMP-compliant manufacturing environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of 2'-substituted benzothiazole delivered meets the exacting standards required for drug research and development, providing our clients with a secure and high-quality source of supply.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic impact of switching to this sustainable method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and competitiveness in your pharmaceutical manufacturing operations.
