Advanced Asymmetric Synthesis of Prostaglandin Intermediates for Commercial Scale Manufacturing
Advanced Asymmetric Synthesis of Prostaglandin Intermediates for Commercial Scale Manufacturing
The pharmaceutical industry continuously seeks robust and scalable methodologies for the production of complex bioactive molecules, particularly prostaglandins and their analogs which serve as critical therapeutics for conditions ranging from glaucoma to cardiovascular diseases. Patent CN101003504B introduces a groundbreaking asymmetric synthesis strategy for preparing formula I lactones and formula II cyclopentanones, which act as pivotal advanced intermediates in the manufacture of prostaglandin F2α compounds. This technology addresses long-standing challenges in stereochemical control and impurity management, offering a viable pathway for reliable prostaglandin intermediate supplier operations to deliver high-purity materials essential for downstream API synthesis. The innovation lies not merely in the reaction sequence but in the profound understanding of chirality transfer and physical property manipulation, specifically crystallinity, to facilitate purification.
Traditionally, the synthesis of these key lactone structures has been plagued by inefficiencies associated with racemic starting materials and cumbersome purification protocols. The present invention provides a novel process that leverages enantiomerically enriched starting materials to achieve optical purities exceeding 99.8% e.e., thereby drastically reducing the burden on downstream processing. For procurement managers and supply chain heads, this translates to a more predictable and stable supply of critical raw materials, minimizing the risk of batch failures due to isomeric contamination. The ability to produce these intermediates with such high fidelity positions this technology as a cornerstone for cost reduction in API manufacturing, particularly for high-volume ophthalmic and cardiovascular drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of formula I lactones has relied heavily on the so-called Corey aldehyde route, a linear synthesis originating from cyclopentadiene. As illustrated in the conventional flow process, this method typically comprises more than 12 reaction steps, inherently accumulating yield losses and operational complexity at each stage. 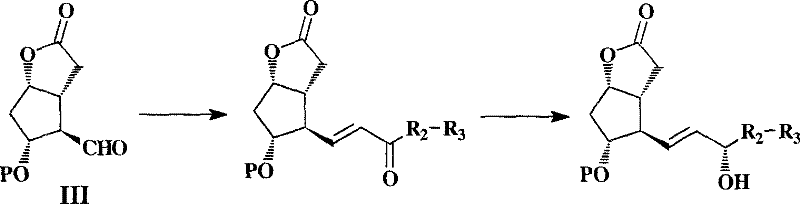 A primary drawback of this legacy approach is the low selectivity observed during the reduction of the 15-ketone functional group on the omega-side chain. This lack of selectivity invariably generates significant quantities of the 15-beta isomer as a major impurity, which possesses physical properties remarkably similar to the desired 15-alpha product. Consequently, removing this isomer necessitates rigorous chromatographic separation, a technique that is notoriously difficult to scale and economically prohibitive for large-scale commercial production. Furthermore, alternative racemic routes, such as those reported by Kalnins involving conjugate addition to racemic cyclopentenones, suffer from poor overall yields of approximately 27% and generate substantial amounts of diastereomeric byproducts that cannot be easily removed by crystallization.
A primary drawback of this legacy approach is the low selectivity observed during the reduction of the 15-ketone functional group on the omega-side chain. This lack of selectivity invariably generates significant quantities of the 15-beta isomer as a major impurity, which possesses physical properties remarkably similar to the desired 15-alpha product. Consequently, removing this isomer necessitates rigorous chromatographic separation, a technique that is notoriously difficult to scale and economically prohibitive for large-scale commercial production. Furthermore, alternative racemic routes, such as those reported by Kalnins involving conjugate addition to racemic cyclopentenones, suffer from poor overall yields of approximately 27% and generate substantial amounts of diastereomeric byproducts that cannot be easily removed by crystallization.
The Novel Approach
In stark contrast to these inefficient legacy methods, the novel approach detailed in the patent utilizes a convergent asymmetric conjugate addition strategy that dramatically streamlines the synthesis. 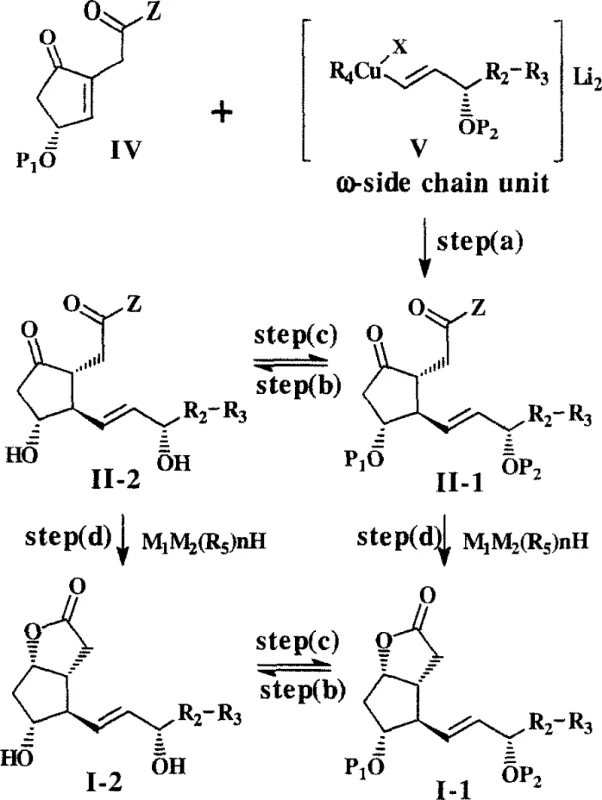 This method couples an optically active cyclopentenone of formula IV with an enantiomerically enriched omega-side chain unit of formula V, derived from cuprates, vinyl stannanes, or alkynes. A key breakthrough in this methodology is the observation that the enantiomeric purity of the resulting cyclopentanone of formula II is often enhanced relative to the starting materials, a phenomenon known as chiral amplification. Moreover, the inventors discovered that modifying the ester protecting group (R1) from a small methyl group to bulkier substituents such as ethyl, benzyl, or menthyl induces unexpected crystallinity in the intermediates. This physical state change is transformative for manufacturing, as it allows for the removal of unwanted diastereomers, particularly the persistent 15-beta isomer, through simple and scalable crystallization techniques rather than chromatography. This shift from chromatographic to crystalline purification represents a paradigm shift in process chemistry, enabling the production of high-purity intermediates suitable for direct progression to final drug substances.
This method couples an optically active cyclopentenone of formula IV with an enantiomerically enriched omega-side chain unit of formula V, derived from cuprates, vinyl stannanes, or alkynes. A key breakthrough in this methodology is the observation that the enantiomeric purity of the resulting cyclopentanone of formula II is often enhanced relative to the starting materials, a phenomenon known as chiral amplification. Moreover, the inventors discovered that modifying the ester protecting group (R1) from a small methyl group to bulkier substituents such as ethyl, benzyl, or menthyl induces unexpected crystallinity in the intermediates. This physical state change is transformative for manufacturing, as it allows for the removal of unwanted diastereomers, particularly the persistent 15-beta isomer, through simple and scalable crystallization techniques rather than chromatography. This shift from chromatographic to crystalline purification represents a paradigm shift in process chemistry, enabling the production of high-purity intermediates suitable for direct progression to final drug substances.
Mechanistic Insights into Asymmetric Conjugate Addition and Chirality Transfer
The core of this technological advancement rests on the precise mechanistic execution of the asymmetric conjugate addition reaction. The process involves the generation of a higher-order cuprate species from the omega-side chain unit, which then attacks the beta-position of the optically active cyclopentenone. The reaction is preferably conducted within a temperature range of -100°C to 40°C, with careful control required to maintain the integrity of the stereocenters. The mechanism ensures that the chirality present in the cyclopentenone starting material is effectively transferred to the newly formed stereocenters in the cyclopentanone product. Crucially, the patent highlights that this transfer is highly efficient, often resulting in a product with higher optical purity than the starting reagents, suggesting a kinetic resolution or selective reaction pathway that favors the formation of the desired enantiomer. This self-correction capability is invaluable for maintaining quality standards without requiring ultra-high purity starting materials, thus relaxing upstream specifications and reducing costs.
Following the coupling reaction, the subsequent reduction and lactonization steps are equally critical for defining the final product profile. The reduction of the ketone functionality is performed using specialized hydride reagents such as lithium tri-sec-butylborohydride, which offers superior stereoselectivity compared to standard reducing agents like sodium borohydride. 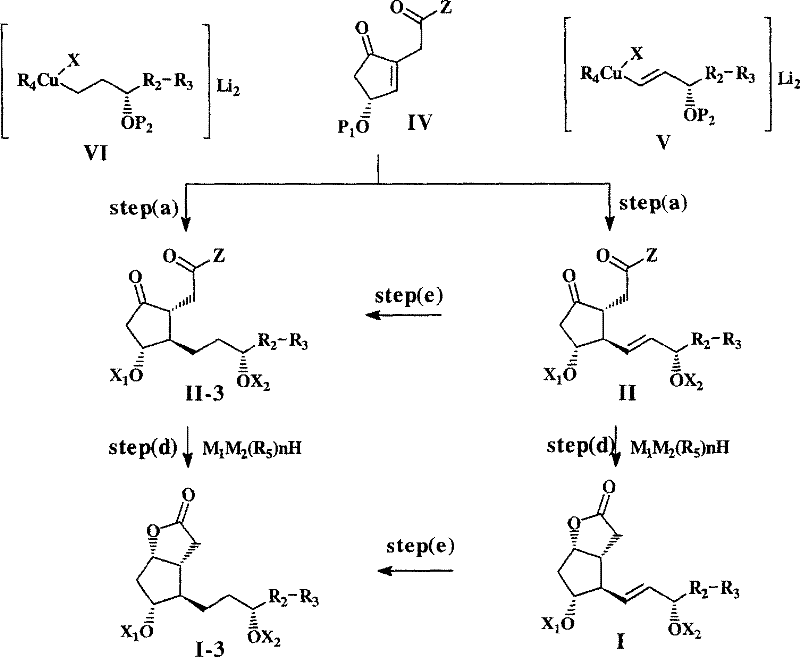 The choice of reducing agent and reaction conditions (-120°C to room temperature) is optimized to minimize the formation of the 9-beta isomer, a common byproduct in lactonization reactions that arises from steric hindrance. By employing bulky ester groups (Z) that serve as leaving groups during lactonization, the process facilitates the intramolecular cyclization while suppressing competing side reactions. The result is a clean conversion to the formula I lactone with minimal epimerization, ensuring that the hard-won optical purity from the conjugate addition step is preserved throughout the synthesis. This mechanistic robustness is what allows the process to consistently deliver intermediates with less than 0.1% of unwanted enantiomers and diastereomers.
The choice of reducing agent and reaction conditions (-120°C to room temperature) is optimized to minimize the formation of the 9-beta isomer, a common byproduct in lactonization reactions that arises from steric hindrance. By employing bulky ester groups (Z) that serve as leaving groups during lactonization, the process facilitates the intramolecular cyclization while suppressing competing side reactions. The result is a clean conversion to the formula I lactone with minimal epimerization, ensuring that the hard-won optical purity from the conjugate addition step is preserved throughout the synthesis. This mechanistic robustness is what allows the process to consistently deliver intermediates with less than 0.1% of unwanted enantiomers and diastereomers.
How to Synthesize Enantiomerically Enriched Prostaglandin Lactones Efficiently
The implementation of this synthesis route requires strict adherence to the optimized reaction parameters defined in the patent to ensure reproducibility and high yield. The process begins with the preparation of the omega-side chain unit, which may involve the conversion of alkynes to vinyl stannanes or iodides, followed by transmetallation to form the reactive cuprate species. This reactive intermediate is then added to the cyclopentenone solution under inert atmosphere conditions to prevent oxidation. Following the coupling, the workup involves careful quenching and extraction, often followed by a deprotection step if silyl protecting groups were utilized on the side chain. The resulting crude cyclopentanone is then subjected to the reduction/lactonization sequence. Detailed standard operating procedures for each unit operation, including specific solvent ratios, addition rates, and temperature ramps, are essential for successful technology transfer.
- Perform asymmetric conjugate addition of an enantiomerically enriched omega-side chain cuprate (Formula V) to an optically active cyclopentenone (Formula IV) at temperatures between -100°C to 40°C to generate the cyclopentanone intermediate (Formula II).
- Purify the resulting cyclopentanone intermediate via crystallization, leveraging bulky ester groups (R1) to enhance crystallinity and remove diastereomeric impurities such as the 15-beta isomer.
- Execute a one-pot reduction and lactonization reaction using a selective reducing agent like lithium tri-sec-butylborohydride to convert the cyclopentanone into the final high-purity lactone (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers compelling strategic advantages that extend beyond mere technical feasibility. The primary value proposition lies in the drastic simplification of the purification train. By eliminating the reliance on preparative chromatography for isomer removal, the process significantly reduces the consumption of expensive silica gel and organic solvents, leading to substantial cost savings in raw materials and waste disposal. Furthermore, the ability to purify intermediates via crystallization enhances the throughput capacity of existing manufacturing facilities, as crystallization tanks are generally more available and easier to operate at scale than chromatography columns. This operational efficiency directly contributes to cost reduction in API manufacturing, allowing for more competitive pricing of the final prostaglandin drugs.
- Cost Reduction in Manufacturing: The elimination of chromatographic purification steps removes a major bottleneck and cost center in the production of prostaglandin intermediates. Chromatography is not only expensive in terms of consumables but also limits batch size and increases cycle times. By leveraging the crystallinity of the bulky ester intermediates, manufacturers can achieve high purity through filtration and washing, processes that are inherently cheaper and faster. Additionally, the shorter synthetic route compared to the Corey aldehyde method reduces the total number of unit operations, further lowering labor and utility costs associated with prolonged processing times.
- Enhanced Supply Chain Reliability: The robustness of the asymmetric conjugate addition process ensures consistent quality across batches, which is critical for maintaining regulatory compliance and avoiding supply disruptions. The tolerance for slightly lower optical purity in starting materials, due to the chiral amplification effect, broadens the supplier base for raw materials and reduces the risk of shortages. Moreover, the crystalline nature of the intermediates improves their stability and shelf-life, facilitating easier storage and transportation logistics. This reliability is paramount for securing the supply chain of life-saving medications like those used for glaucoma treatment.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reagents and conditions that are amenable to large-scale reactor operations. The reduction in solvent usage and waste generation associated with the removal of chromatography steps aligns with modern green chemistry principles and environmental regulations. This makes the technology attractive for manufacturers looking to minimize their environmental footprint while expanding production capacity. The ability to scale from kilogram to multi-ton quantities without fundamental process changes ensures a seamless transition from clinical trial material to commercial supply.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. Understanding these nuances is essential for R&D teams evaluating the feasibility of adopting this route for their specific product portfolios. The answers provided are derived directly from the experimental data and embodiments disclosed in the patent documentation, ensuring accuracy and relevance.
Q: How does this novel process improve upon the traditional Corey aldehyde route?
A: The traditional Corey route involves over 12 steps and suffers from low selectivity during the reduction of the 15-ketone group, leading to difficult-to-remove 15-beta isomers. This novel asymmetric conjugate addition process significantly shortens the synthetic route and utilizes crystallization to efficiently remove diastereomeric impurities, ensuring higher optical purity without extensive chromatography.
Q: What is the significance of the ester group (R1) modification in this synthesis?
A: The patent reveals a critical discovery that changing the ester group from a small methyl group to bulkier groups like ethyl, benzyl, or menthyl induces crystallinity in the cyclopentanone intermediates. This physical property change allows for the removal of unwanted isomers via simple crystallization rather than complex chromatographic separation, which is vital for industrial scalability.
Q: Can this method be applied to synthesize specific drugs like Latanoprost?
A: Yes, the process is highly versatile. By selecting specific omega-side chain units (such as those containing phenyl groups or specific protecting groups), the method can produce intermediates for various prostaglandin analogs including Latanoprost, Bimatoprost, and Travoprost, as demonstrated by the synthesis of 13,14-dihydro lactones described in the patent.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prostaglandin Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical supply chain. Our team of expert chemists has extensively analyzed the technological advancements presented in patent CN101003504B and possesses the capability to implement this sophisticated asymmetric synthesis on a commercial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the detection of trace isomers at levels below 0.1%, guaranteeing that every batch meets the highest industry standards.
We invite you to collaborate with us to leverage this cutting-edge technology for your prostaglandin development programs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate your time to market with our reliable supply of advanced prostaglandin intermediates.
