Scalable Visible-Light Mediated Arylation for High-Purity Pharmaceutical Intermediates and Fine Chemicals
Scalable Visible-Light Mediated Arylation for High-Purity Pharmaceutical Intermediates and Fine Chemicals
The landscape of organic synthesis is undergoing a paradigm shift towards sustainable, metal-free methodologies, a trend vividly exemplified by the recent technological breakthroughs detailed in patent CN113956208A. This intellectual property introduces a robust and environmentally benign protocol for the construction of aryl heterocyclic derivatives, a class of compounds serving as critical scaffolds in modern drug discovery and material science. By leveraging visible-light photocatalysis, specifically utilizing Eosin Y as an organic photocatalyst, this method circumvents the traditional reliance on expensive and toxic transition metals. For R&D directors and procurement specialists alike, this represents a significant opportunity to streamline supply chains and enhance the purity profiles of complex intermediates. The process utilizes readily available iodine ylide compounds as aryl radical precursors, reacting them with diverse heterocyclic cores under mild LED illumination. This approach not only aligns with green chemistry principles but also offers a practical pathway for the cost reduction in pharmaceutical intermediate manufacturing by eliminating costly metal scavenging steps.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the introduction of aryl groups onto heterocyclic scaffolds has relied heavily on transition-metal-catalyzed cross-coupling reactions, such as Suzuki-Miyaura or Buchwald-Hartwig aminations. While effective, these conventional pathways impose significant logistical and financial burdens on large-scale production. They typically require pre-functionalized substrates, such as aryl halides or boronic acids, which increases the step count and overall raw material costs. Furthermore, the use of palladium, copper, or nickel catalysts introduces a persistent risk of heavy metal residue in the final active pharmaceutical ingredient (API), necessitating rigorous and expensive purification protocols to meet regulatory limits. These processes often demand harsh reaction conditions, including elevated temperatures and inert atmospheres, which escalate energy consumption and complicate reactor safety management. Consequently, the cumulative effect of these factors results in longer lead times for high-purity pharmaceutical intermediates and reduced overall process efficiency.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a direct C-H arylation strategy driven by visible-light photoredox catalysis. This innovative route employs iodine ylides as efficient radical generators, which, upon excitation by an LED light source in the presence of Eosin Y, release aryl radicals that directly functionalize the heterocyclic C-H bond. 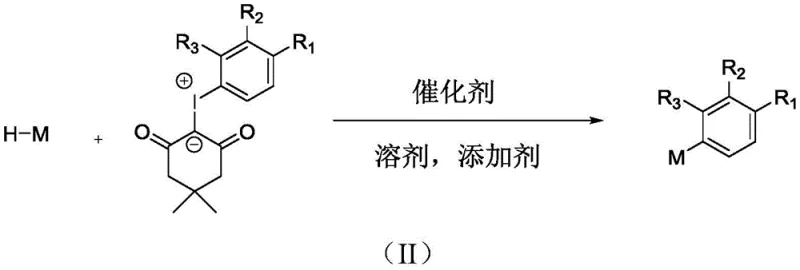 This eliminates the need for pre-halogenated substrates, thereby shortening the synthetic sequence and reducing waste generation. The reaction proceeds smoothly in polar aprotic solvents like DMSO at near-ambient temperatures (20-35°C), drastically lowering energy requirements. By replacing precious metal catalysts with an inexpensive organic dye, the process inherently reduces the potential for metal contamination, simplifying downstream processing. This novel approach offers a versatile platform for generating structural diversity, accommodating a wide range of substituents on both the aryl and heterocyclic components, thus providing a powerful tool for rapid library synthesis in medicinal chemistry.
This eliminates the need for pre-halogenated substrates, thereby shortening the synthetic sequence and reducing waste generation. The reaction proceeds smoothly in polar aprotic solvents like DMSO at near-ambient temperatures (20-35°C), drastically lowering energy requirements. By replacing precious metal catalysts with an inexpensive organic dye, the process inherently reduces the potential for metal contamination, simplifying downstream processing. This novel approach offers a versatile platform for generating structural diversity, accommodating a wide range of substituents on both the aryl and heterocyclic components, thus providing a powerful tool for rapid library synthesis in medicinal chemistry.
Mechanistic Insights into Eosin Y-Catalyzed Photoredox Arylation
The mechanistic elegance of this transformation lies in the photophysical properties of Eosin Y, which acts as a potent single-electron transfer (SET) agent under visible light irradiation. Upon absorption of photons from the LED source, the ground state Eosin Y is excited to its singlet state and rapidly undergoes intersystem crossing to a long-lived triplet state. This excited photocatalyst possesses sufficient redox potential to interact with the iodine ylide species, initiating a single-electron oxidation that generates a radical cation intermediate. Subsequent fragmentation of this unstable intermediate releases an aryl radical and an iodine byproduct. The highly reactive aryl radical then adds to the electron-deficient heterocyclic ring, forming a new carbon-carbon bond and a transient radical species on the heterocycle. 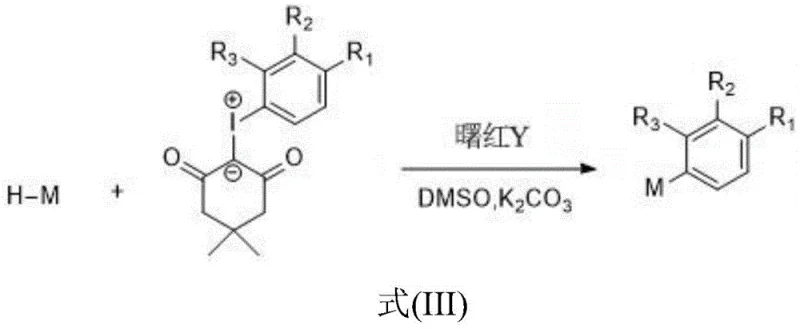 Finally, the catalytic cycle is closed through a subsequent electron transfer and deprotonation step, regenerating the aromaticity of the heterocycle and restoring the ground state photocatalyst. This radical-mediated pathway avoids the high-energy oxidative addition and reductive elimination steps typical of transition metal cycles, allowing the reaction to proceed under exceptionally mild thermal conditions.
Finally, the catalytic cycle is closed through a subsequent electron transfer and deprotonation step, regenerating the aromaticity of the heterocycle and restoring the ground state photocatalyst. This radical-mediated pathway avoids the high-energy oxidative addition and reductive elimination steps typical of transition metal cycles, allowing the reaction to proceed under exceptionally mild thermal conditions.
From an impurity control perspective, this mechanism offers distinct advantages for maintaining high chemical purity. Since the reaction does not involve transition metals, the formation of metal-complexed side products is entirely avoided, which is a common issue in Pd-catalyzed couplings. The primary byproducts are typically simple iodine salts and protonated base, which are easily removed during aqueous workup. The selectivity of the aryl radical addition is governed by the electronic nature of the heterocycle, favoring positions with high spin density, which leads to predictable regioselectivity. Furthermore, the use of mild bases like potassium carbonate minimizes the risk of base-sensitive functional group degradation, preserving the integrity of complex molecular architectures. This clean reaction profile ensures that the resulting aryl heterocyclic derivatives exhibit narrow impurity spectra, facilitating easier crystallization and purification, which is critical for meeting the stringent quality standards required for reliable pharmaceutical intermediate supplier status.
How to Synthesize Aryl Heterocyclic Derivatives Efficiently
The operational simplicity of this photocatalytic protocol makes it highly attractive for both laboratory optimization and pilot plant operations. The procedure involves charging a reaction vessel with the chosen heterocyclic substrate and the iodine ylide coupling partner in a defined molar ratio, typically ranging from 1:1 to 1:3 to ensure complete conversion of the limiting reagent. The system is then supplemented with the Eosin Y catalyst and a stoichiometric amount of an inorganic base, such as potassium carbonate or cesium carbonate, which serves to neutralize the acidic byproducts generated during the radical propagation. Dimethyl sulfoxide (DMSO) is identified as the preferred solvent due to its ability to dissolve both organic substrates and inorganic salts while stabilizing the charged intermediates involved in the photoredox cycle. The detailed standardized synthesis steps are outlined below.
- Prepare the reaction mixture by combining the heterocyclic compound (e.g., quinoxalinone) and iodine ylide compound in a molar ratio of 1: 1 to 1:3.
- Add the organic photocatalyst Eosin Y (0.02-0.05 equivalents) and an inorganic base additive such as potassium carbonate (1-1.5 equivalents) to the mixture.
- Dissolve the reagents in dimethyl sulfoxide (DMSO) and irradiate with an LED light source at 20-35°C for 6-12 hours to obtain the target aryl heterocyclic derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free photocatalytic technology translates into tangible strategic benefits regarding cost stability and supply continuity. The elimination of precious metal catalysts removes exposure to the volatile pricing markets associated with palladium and iridium, leading to more predictable raw material costs. Additionally, the simplified purification train reduces the consumption of specialized scavenger resins and silica gel, further driving down the cost of goods sold (COGS). The mild reaction conditions also imply lower energy overheads and reduced wear on reactor vessels, contributing to long-term operational savings. Moreover, the use of commodity chemicals like Eosin Y and potassium carbonate ensures a robust supply chain that is less susceptible to geopolitical disruptions compared to specialized organometallic reagents.
- Cost Reduction in Manufacturing: The substitution of expensive transition metal catalysts with organic dyes like Eosin Y fundamentally alters the cost structure of the synthesis. By removing the need for ppm-level metal removal technologies, manufacturers can significantly reduce downstream processing costs. The high atom economy of the direct C-H functionalization also means less waste disposal cost. Qualitative analysis suggests that the overall process mass intensity (PMI) is improved, leading to substantial cost savings in solvent usage and waste treatment. This economic efficiency allows for more competitive pricing strategies in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: The reliance on widely available, bulk chemical feedstocks enhances the resilience of the supply chain. Iodine ylides and heterocyclic precursors are commercially accessible from multiple vendors, mitigating the risk of single-source bottlenecks. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by equipment failures associated with high-pressure or high-temperature operations. This reliability ensures consistent delivery timelines, which is crucial for maintaining just-in-time inventory models in pharmaceutical manufacturing. The stability of the reagents also allows for longer shelf-life and easier logistics management.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging, but advancements in LED technology and flow chemistry have made this increasingly viable. The low thermal load of this reaction facilitates safer scale-up, as the risk of thermal runaway is minimal compared to exothermic metal-catalyzed reactions. From an environmental standpoint, the absence of heavy metals simplifies effluent treatment and helps facilities meet increasingly strict environmental regulations. The green nature of the process aligns with corporate sustainability goals, potentially qualifying the manufacturing site for green chemistry incentives and improving the company's environmental, social, and governance (ESG) rating.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic arylation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into existing production workflows.
Q: What are the primary advantages of using Eosin Y over transition metal catalysts for arylation?
A: Using Eosin Y eliminates the risk of heavy metal contamination in the final API, significantly simplifying purification processes and ensuring compliance with stringent pharmaceutical impurity guidelines.
Q: Can this photocatalytic method be scaled for industrial production?
A: Yes, the reaction operates at mild temperatures (20-35°C) and atmospheric pressure using standard LED sources, making it highly suitable for safe commercial scale-up without specialized high-pressure equipment.
Q: What types of heterocyclic substrates are compatible with this synthesis route?
A: The method demonstrates broad substrate scope, successfully functionalizing nitrogen-containing heterocycles like quinoxalinones and imidazopyridines, as well as oxygen-containing systems like coumarins and flavones.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Heterocyclic Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible-light mediated synthesis in the production of high-value pharmaceutical intermediates. Our technical team has extensively evaluated the scalability of such photocatalytic pathways and possesses the expertise to translate laboratory successes into commercial reality. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from gram-scale optimization to multi-ton manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch meets stringent purity specifications required by global regulatory bodies. We are committed to delivering high-purity aryl heterocyclic derivatives that empower your drug development programs.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific project needs. Our engineering team is ready to conduct a Customized Cost-Saving Analysis tailored to your target molecule, identifying opportunities to optimize yield and reduce waste. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain that prioritizes innovation, quality, and sustainability, ensuring a competitive edge in the fast-paced pharmaceutical industry.
