Advanced Nickel-Catalyzed Synthesis of Allylamine Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex nitrogen-containing scaffolds, particularly allylamine derivatives which serve as critical building blocks for bioactive molecules. Patent CN112552215A introduces a groundbreaking methodology that leverages earth-abundant nickel catalysis to achieve the direct dehydrogenative coupling of amines and alkynes. This innovation represents a significant leap forward in synthetic efficiency, as it utilizes the hydrogen inherent in the amine reactant to reduce the alkyne, thereby eliminating the need for external reducing agents. For a reliable pharmaceutical intermediate supplier, mastering such atom-economical transformations is essential for delivering high-purity compounds while adhering to green chemistry principles. The technology described allows for the construction of diverse allylamine structures from readily available benzylamine and alkyne precursors, offering a versatile platform for drug discovery and process development.
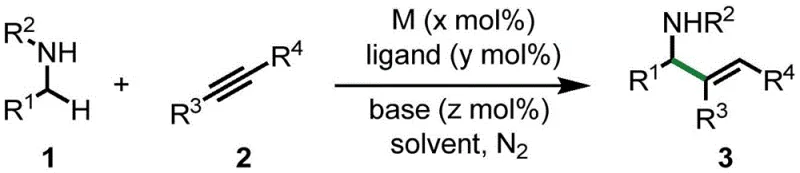
This novel approach fundamentally shifts the paradigm of allylamine synthesis by addressing the limitations of conventional transition metal catalysis. Traditionally, the preparation of these valuable motifs often relied on precious metal catalysts such as rhodium or required the pre-formation of unstable imine intermediates. Furthermore, existing reductive coupling methods frequently necessitated the addition of stoichiometric amounts of reducing agents like triethylborane or zinc alkyls, which generate substantial waste and complicate downstream purification. In stark contrast, the method disclosed in CN112552215A operates through a hydrogen-transfer mechanism where the amine itself acts as the hydrogen donor. This not only simplifies the reaction setup by removing the need for hazardous reductants but also drastically improves the overall atom economy. The ability to bypass the isolation of sensitive imine intermediates further enhances the operational simplicity, making this route highly attractive for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Nickel-Catalyzed Dehydrogenative Coupling
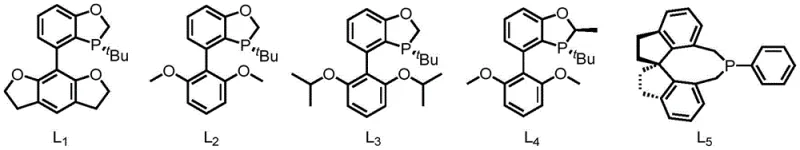
The success of this transformation hinges on the precise interplay between the nickel center and the phosphine ligand environment. The catalytic cycle likely initiates with the oxidative addition of the benzylic C-H bond of the amine to the Ni(0) species, forming a nickel-hydride intermediate. This step is facilitated by the electron-rich nature of ligands such as tricyclohexylphosphine (PCy3) or dicyclohexylphenylphosphine, which stabilize the active metal center. Subsequent insertion of the alkyne into the nickel-carbon bond followed by reductive elimination yields the allylamine product while regenerating the catalyst. The choice of ligand is critical; for instance, the use of bulky monophosphine ligands prevents catalyst deactivation and promotes the desired coupling pathway over competing side reactions. When chiral variants of these ligands, such as the specialized structure L1 shown below, are employed, the steric environment around the nickel center becomes asymmetric, enabling the differentiation of prochiral faces during the bond-forming events.
Impurity control in this system is inherently managed by the specificity of the nickel catalyst and the stability of the sulfonamide protecting group, typically the mesitylenesulfonyl (Mts) group. The Mts group not only activates the alpha-C-H bond of the amine for metallation but also provides robust protection against over-oxidation or polymerization of the reactive allylic system. The reaction conditions, typically involving potassium phosphate as a mild base in solvents like 1,4-dioxane, are optimized to minimize the formation of homocoupling byproducts or isomerized alkene impurities. By maintaining a strict nitrogen atmosphere and controlling the temperature between 80°C and 140°C, the process ensures that the hydrogen transfer occurs selectively. This level of control is vital for producing high-purity allylamine derivatives that meet the stringent quality standards required for clinical applications, ensuring that the final API intermediates possess the necessary chemical integrity.
How to Synthesize Allylamine Derivatives Efficiently
The practical implementation of this nickel-catalyzed protocol offers a streamlined workflow for laboratory and pilot-scale synthesis. The procedure involves the straightforward mixing of the amine substrate, alkyne, base, catalyst, and ligand in a single vessel, followed by heating to drive the dehydrogenative coupling to completion. This one-pot strategy significantly reduces labor time and solvent consumption compared to multi-step sequences involving imine formation and separate reduction steps. The detailed standardized synthesis steps for this specific transformation are outlined in the guide below, providing a clear roadmap for technical teams looking to adopt this methodology.
- Mix amine substrate, alkyne, inorganic base (K3PO4), Ni(cod)2 catalyst, and phosphine ligand under nitrogen atmosphere.
- Add solvent such as 1,4-Dioxane or THF and heat the mixture to 80-140°C for 24 hours to facilitate hydrogen transfer and coupling.
- Cool the reaction, concentrate the mixture, and purify the crude product via column chromatography to isolate the target allylamine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this nickel-catalyzed technology offers substantial strategic benefits that extend beyond mere chemical yield. The shift from precious metals like rhodium to abundant nickel complexes results in a drastic reduction in raw material costs, insulating the supply chain from the volatility associated with noble metal markets. Furthermore, the elimination of stoichiometric reducing agents simplifies the waste stream, reducing the environmental burden and the costs associated with hazardous waste disposal. This aligns perfectly with modern sustainability goals and regulatory pressures facing chemical manufacturers today. The robustness of the reaction conditions also implies a lower risk of batch failure, enhancing supply continuity for critical drug intermediates.
- Cost Reduction in Manufacturing: The replacement of expensive rhodium catalysts with economical nickel systems, combined with the removal of external reducing agents, leads to significant savings in bill of materials. The simplified workup procedure, which avoids complex quenching of reactive hydrides, further lowers processing costs and increases throughput capacity without requiring capital-intensive equipment upgrades.
- Enhanced Supply Chain Reliability: Since the starting materials, including various substituted benzylamines and alkynes, are commercially available and stable, sourcing risks are minimized. The method's tolerance to diverse functional groups means that a single robust platform can be used to synthesize a wide library of analogs, reducing the need for multiple specialized supply lines and streamlining inventory management for complex pharmaceutical portfolios.
- Scalability and Environmental Compliance: The patent data confirms that the reaction maintains high efficiency even when scaled up to gram levels, demonstrating its readiness for commercial scale-up of complex pharmaceutical intermediates. The atom-economical nature of the hydrogen transfer mechanism minimizes waste generation, facilitating easier compliance with increasingly strict environmental regulations regarding solvent use and chemical effluent treatment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed synthesis route. These insights are derived directly from the experimental data and scope limitations defined in the patent documentation, providing clarity for R&D and operations teams evaluating this technology for their specific production needs.
Q: What is the primary advantage of this nickel-catalyzed method over traditional rhodium catalysis?
A: The primary advantage is the elimination of expensive noble metals like rhodium and the avoidance of stoichiometric reducing agents, significantly lowering raw material costs and improving atom economy.
Q: Can this process produce chiral allylamines with high optical purity?
A: Yes, by employing specific chiral monophosphine ligands such as L1, the method achieves high enantioselectivity, making it suitable for synthesizing chiral drug intermediates.
Q: Is this synthesis method scalable for industrial production?
A: The patent demonstrates successful scale-up to gram levels with maintained yields, indicating robust potential for commercial manufacturing and supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Allylamine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of nickel-catalyzed dehydrogenative coupling in modern medicinal chemistry. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative academic discoveries like CN112552215A are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of allylamine derivative meets the exacting standards required for global pharmaceutical supply chains.
We invite you to collaborate with our technical team to evaluate the feasibility of this cost-effective route for your specific project requirements. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis tailored to your target molecule. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and economical supply of high-quality chemical intermediates.
