Advanced Oxidation Strategy for High-Purity 2-(Methylsulfinyl)benzoic Acid Production
Introduction to Next-Generation Sulfoxide Synthesis
The synthesis of functionalized benzoic acid derivatives remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting antifungal and preservative activities. A pivotal advancement in this domain is documented in patent CN111848464A, which discloses a highly efficient, one-step method for preparing 2-(methylsulfinyl)benzoic acid. This specific compound serves as a critical building block for various bioactive molecules, yet its historical synthesis has been plagued by harsh conditions and toxic reagents. The disclosed methodology leverages the unique oxidative properties of Selectfluor (1-chloromethyl-4-fluoro-1,4-diazabicyclo[2.2.2]octane bis(tetrafluoroborate) salt) in a methanol medium. By operating at a mild temperature of 20 degrees Celsius, this process not only achieves exceptional conversion rates but also aligns with the growing industry demand for greener, safer chemical manufacturing protocols. For R&D directors and procurement specialists alike, understanding the nuances of this transformation is essential for securing a reliable supply chain of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the chemical community relied on three primary routes to access 2-(methylsulfinyl)benzoic acid, each fraught with significant operational and safety liabilities. The first conventional method utilized a complex catalytic system involving iron bromide and beta-cyclodextrin in acetonitrile, requiring unstable transition metal reagents like ferric nitrate that are prone to deliquescence. The second approach depended on trifluoroacetic acid as an oxidant, necessitating elevated temperatures and introducing severe corrosion risks to reactor vessels. Perhaps most concerning was the third method, which employed m-chloroperoxybenzoic acid (m-CPBA) in chloroform; this route combined the hazards of a potentially explosive peracid with the toxicity of chlorinated solvents. These legacy processes not only complicated waste treatment and regulatory compliance but also introduced variability in yield and purity, creating bottlenecks for the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
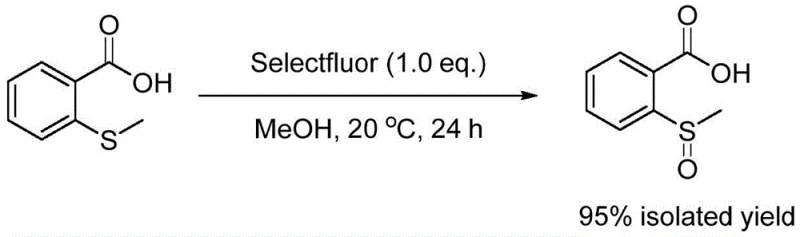
In stark contrast, the novel methodology presented in the patent data utilizes a direct oxidation strategy that bypasses these historical hurdles entirely. By employing Selectfluor as a stoichiometric oxidant in methanol, the reaction proceeds smoothly at ambient temperature (20 degrees Celsius) without the need for transition metals or corrosive acids. The simplicity of the workup—merely filtering the reaction mixture followed by reduced pressure distillation—drastically reduces processing time and solvent consumption. As illustrated in the reaction scheme below, the transformation of 2-(methylthio)benzoic acid to the target sulfoxide is achieved with remarkable efficiency, delivering an isolated yield of 95 percent under optimized conditions. This represents a paradigm shift from multi-step, hazard-heavy protocols to a streamlined, single-operation unit process suitable for modern GMP manufacturing environments.
Mechanistic Insights into Selectfluor-Mediated Oxidation
The efficacy of this transformation hinges on the specific interaction between the electrophilic fluorinating agent, Selectfluor, and the nucleophilic sulfur atom of the thioether substrate. Unlike traditional oxidants that may generate radical species leading to over-oxidation or side reactions, Selectfluor acts as a source of electrophilic positive charge, facilitating a controlled two-electron oxidation of the sulfide to the sulfoxide. The mechanism likely involves the initial attack of the sulfur lone pair on the nitrogen-fluorine bond of the reagent, forming a transient sulfonium intermediate which is subsequently hydrolyzed or solvolyzed by the methanol solvent to release the sulfoxide product. This pathway is inherently cleaner because it avoids the formation of metal salts or acidic byproducts that typically complicate downstream purification. Furthermore, the mildness of the reagent ensures that sensitive functional groups elsewhere on the aromatic ring remain intact, preserving the structural integrity required for subsequent coupling reactions in drug synthesis.
Crucially, the choice of solvent plays a decisive role in the success of this mechanistic pathway, as evidenced by comparative experimental data. While acetonitrile is a common polar aprotic solvent for many organic transformations, its use in this specific oxidation results in a precipitous drop in yield to merely 15 percent, as shown in the comparative reaction scheme. This suggests that methanol is not merely a passive medium but actively participates in stabilizing the charged intermediates or facilitating the proton transfer steps necessary for product release. Additionally, stoichiometric precision is vital; reducing the Selectfluor loading to 0.5 equivalents results in a complete failure of the reaction (0 percent yield), confirming that the oxidation requires a full equivalent of the oxidant to proceed to completion. These insights underscore the importance of adhering strictly to the patented parameters to ensure consistent quality in high-purity OLED material or pharmaceutical intermediate production.

How to Synthesize 2-(Methylsulfinyl)benzoic acid Efficiently
Implementing this synthesis on a laboratory or pilot scale requires strict adherence to the optimized parameters defined in the patent to maximize yield and minimize impurities. The process is designed to be operationally simple, avoiding the need for specialized high-pressure equipment or cryogenic cooling systems. Operators should focus on maintaining the precise molar ratio of reagents and ensuring adequate agitation to facilitate mass transfer between the solid Selectfluor and the solution phase. The following guide outlines the standardized procedure derived from the patent examples, serving as a foundational protocol for process chemists aiming to replicate these results. For a detailed breakdown of the specific quantities and handling precautions, please refer to the structured synthesis guide below.
- Dissolve 2-(methylthio)benzoic acid in methanol solvent within a reaction flask to achieve a concentration of approximately 0.2 mol/L.
- Add 1.0 equivalent of Selectfluor (1-chloromethyl-4-fluoro-1,4-diazabicyclo[2.2.2]octane bis(tetrafluoroborate) salt) to the reaction mixture.
- Stir the mixture vigorously at 20 degrees Celsius for 24 hours, then filter and distill under reduced pressure to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond mere chemical yield. By eliminating the reliance on volatile and regulated solvents like chloroform, as well as hazardous oxidants like m-CPBA, the process significantly reduces the regulatory burden associated with environmental health and safety compliance. This simplification translates directly into lower operational expenditures, as the costs associated with hazardous waste disposal and specialized containment infrastructure are drastically curtailed. Furthermore, the use of commercially available, stable reagents like Selectfluor ensures a robust supply chain that is less susceptible to the disruptions often seen with custom-synthesized catalysts or unstable peracids.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts such as iron bromide removes the necessity for expensive and time-consuming metal scavenging steps, which are often required to meet stringent residual metal limits in pharmaceutical ingredients. Additionally, the ability to run the reaction at ambient temperature (20 degrees Celsius) eliminates energy costs associated with heating or cooling large-scale reactors, leading to substantial cost savings in utility consumption. The high isolated yield of over 90 percent further enhances cost efficiency by maximizing the throughput of raw materials, ensuring that every kilogram of starting material contributes effectively to the final product inventory without significant loss to side reactions.
- Enhanced Supply Chain Reliability: The starting material, 2-(methylthio)benzoic acid, is a commodity chemical with a well-established global supply network, reducing the risk of raw material shortages that can plague proprietary precursor markets. Moreover, the stability of the reagents involved means that inventory can be held for longer periods without degradation, allowing manufacturers to maintain strategic stockpiles against market volatility. This reliability is critical for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream drug development timelines are not compromised by upstream synthesis delays or quality failures.
- Scalability and Environmental Compliance: The benign nature of the solvent system (methanol) and the absence of toxic byproducts make this process inherently scalable from gram to tonnage quantities without requiring major engineering redesigns. The simplified workup procedure, involving only filtration and distillation, facilitates easier technology transfer between sites and reduces the footprint of the manufacturing facility. From an environmental perspective, avoiding chlorinated solvents and heavy metals aligns with green chemistry principles, enhancing the corporate sustainability profile and ensuring compliance with increasingly strict international environmental regulations regarding industrial effluent.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this process for their specific applications, we have compiled answers to common inquiries regarding the reaction parameters and scalability. These responses are grounded in the experimental data provided in the patent documentation, addressing concerns about solvent selection, reagent stoichiometry, and purification methods. Understanding these technical details is crucial for assessing the compatibility of this route with existing manufacturing capabilities and quality control standards.
Q: Why is methanol preferred over acetonitrile for this oxidation?
A: Experimental data indicates that methanol provides a significantly higher isolated yield (95 percent) compared to acetonitrile (15 percent), likely due to better solubility of the transition state or stabilization of the ionic intermediates generated by Selectfluor.
Q: What is the minimum stoichiometry required for Selectfluor?
A: Using only 0.5 equivalents of Selectfluor results in zero percent yield, indicating that a full equivalent (1.0 eq.) is strictly necessary to drive the oxidation of the sulfide to the sulfoxide efficiently.
Q: Does this method require transition metal catalysts?
A: No, this novel approach eliminates the need for transition metal catalysts like iron bromide or ferric nitrate, thereby simplifying the purification process and removing the risk of heavy metal contamination in the final API intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(Methylsulfinyl)benzoic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality assurance. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields observed in the lab are faithfully reproduced on an industrial scale. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-(methylsulfinyl)benzoic acid meets the exacting standards required for pharmaceutical and fine chemical applications. We are committed to delivering not just a product, but a validated, reliable supply solution that supports your long-term R&D and commercial goals.
We invite you to engage with our technical procurement team to discuss how this advanced oxidation technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this metal-free protocol. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your project's unique volume and purity requirements, ensuring a seamless path from development to market.
