Advanced Metal-Free Grinding Synthesis of N,N-Dimethyl Dithioacetate Intermediates for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing carbon-sulfur bonds, a critical structural motif found in numerous bioactive molecules and functional materials. Patent CN112028802B introduces a groundbreaking approach for the preparation of N,N-dimethyl dithioacetic acid phenyl ester compounds, utilizing a novel grinding method that fundamentally shifts the paradigm from traditional metal-catalyzed processes to a more sustainable, metal-free protocol. This innovation addresses long-standing challenges in organic sulfide synthesis, such as catalyst poisoning by sulfur lone pairs and the generation of malodorous byproducts, by employing a three-component reaction system involving thiophenol derivatives, thionyl chloride, and N,N-dimethylbenzoyl allylamine. The significance of this patent lies not only in its chemical elegance but also in its potential to streamline the supply chain for reliable pharmaceutical intermediate suppliers who demand high purity and operational simplicity. By operating under ambient pressure and temperature, this technology offers a distinct advantage for cost reduction in API manufacturing, eliminating the energy-intensive requirements often associated with high-temperature coupling reactions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbon-sulfur bonds has relied heavily on transition metal catalysis, which presents significant hurdles for large-scale commercialization due to the inherent toxicity and cost of precious metals like palladium or copper. A major technical bottleneck in these conventional pathways is the strong coordination of sulfur atoms to metal centers, which frequently leads to catalyst deactivation or poisoning, thereby necessitating excessive catalyst loading and complicating the reaction kinetics. Furthermore, traditional methods often struggle with the oxidation sensitivity of thiol substrates, leading to the formation of disulfide impurities that are difficult to separate and can compromise the quality of high-purity OLED material or pharmaceutical precursors. The environmental footprint of these legacy processes is also substantial, as they typically require rigorous purification steps to remove trace metal residues to meet stringent regulatory limits for drug substances. Additionally, the handling of volatile and malodorous sulfur reagents in open systems poses serious occupational health and safety risks, increasing the complexity of facility management and waste treatment protocols.
The Novel Approach
In stark contrast to these legacy issues, the grinding method disclosed in the patent utilizes a mechanochemical approach that activates the reactants through physical shear forces rather than thermal energy or metal mediation. This technique allows for the efficient coupling of thiophenols with thionyl chloride and enamines in the presence of triethylamine, creating a reactive environment that bypasses the need for transition metals entirely. The process is conducted in dichloromethane at room temperature, which significantly lowers the energy consumption profile and reduces the thermal stress on sensitive functional groups that might be present on the aromatic ring. As illustrated in the reaction scheme below, the direct interaction of the three components leads to the formation of the dithioester backbone with remarkable efficiency and selectivity.
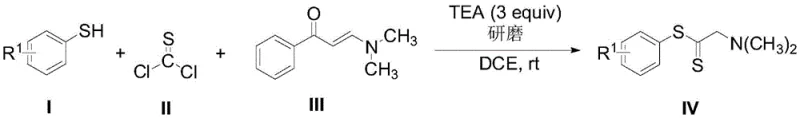
This metal-free strategy not only simplifies the downstream processing by removing the need for metal scavenging resins but also ensures that the final product is free from heavy metal contamination, a critical factor for reducing lead time for high-purity pharmaceutical intermediates. The use of readily available and relatively odorless reagents further enhances the operational safety, making this a superior choice for facilities aiming to improve their environmental compliance and worker safety standards without sacrificing yield or throughput.
Mechanistic Insights into Metal-Free Grinding Coupling
The mechanistic pathway of this transformation relies on the in situ generation of a highly electrophilic sulfur species from the reaction between the thiophenol and thionyl chloride, which is then trapped by the nucleophilic enamine component. Triethylamine plays a dual role in this system, acting both as a base to neutralize the hydrochloric acid byproduct generated during the activation of the thiol and as a promoter for the grinding efficiency by maintaining a homogeneous reaction medium. The absence of metal catalysts means that the reaction trajectory is governed purely by electronic effects and steric accessibility, allowing for a broad substrate scope that includes electron-rich and electron-deficient thiophenols alike. The grinding action itself likely enhances the mass transfer between the solid and liquid phases, ensuring that the local concentration of reactive intermediates remains high enough to drive the reaction to completion within a reasonable timeframe of 10 hours. This mechanistic simplicity is a key driver for the commercial scale-up of complex polymer additives and fine chemicals, as it reduces the number of variables that need to be controlled during production.
Impurity control in this system is inherently robust because the primary side reactions, such as over-oxidation to sulfoxides or sulfones, are minimized by the mild, non-oxidative conditions of the grinding process. The patent data indicates that various substituents on the phenyl ring, including methyl, methoxy, allyl, and even aldehyde groups, are well-tolerated, yielding the corresponding dithioesters in excellent purity as confirmed by NMR and mass spectrometry analysis. The structural diversity achievable with this method is demonstrated by the range of products synthesized, showcasing the versatility of the protocol for generating libraries of sulfur-containing building blocks.
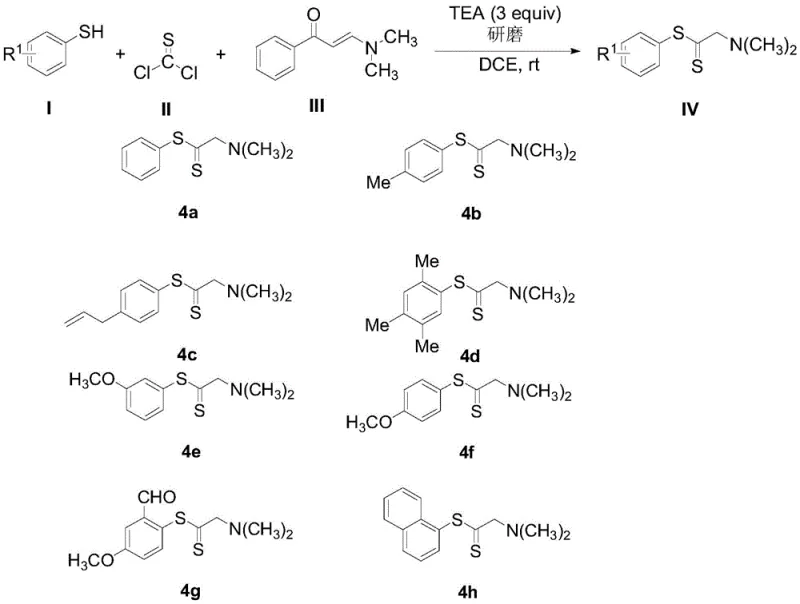
Such broad compatibility ensures that R&D teams can access a wide array of analogues for structure-activity relationship studies without needing to re-optimize reaction conditions for each new substrate. The consistency of the results across different electronic environments suggests that the rate-determining step is likely the initial activation of the thiol, which is efficiently managed by the stoichiometric excess of thionyl chloride and the basic conditions provided by the amine.
How to Synthesize N,N-Dimethyl Dithioacetate Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric ratios and mixing protocols to maximize the benefits of the grinding technique. The patent specifies a molar ratio of 1:1.2:1 for the thiophenol, thionyl chloride, and enamine respectively, with triethylamine added in a 3-equivalent excess to ensure complete acid scavenging throughout the 10-hour reaction window. Detailed standard operating procedures regarding the specific grinding equipment, solvent volumes, and workup parameters are essential for reproducibility and are outlined in the technical guide below.
- Combine thiophenol derivatives, thionyl chloride, and N,N-dimethylbenzoyl allylamine in dichloromethane with triethylamine.
- Perform the reaction under grinding conditions at room temperature (25°C) for approximately 10 hours to ensure complete conversion.
- Execute post-treatment via aqueous extraction, drying over anhydrous sodium sulfate, and purification using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this grinding methodology represents a strategic opportunity to optimize the cost structure and reliability of the sulfur-containing intermediate supply chain. By eliminating the dependency on volatile and expensive transition metal catalysts, manufacturers can achieve significant cost savings in raw material procurement and reduce the financial risk associated with fluctuating metal prices in the global market. The simplified workup procedure, which involves standard extraction and column chromatography rather than complex metal filtration or chelation steps, translates directly into reduced labor costs and shorter batch cycle times, enhancing overall plant throughput. Furthermore, the mild reaction conditions decrease the wear and tear on reactor vessels and auxiliary equipment, extending the lifespan of capital assets and lowering maintenance expenditures over the long term.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts from the process flow eliminates the need for costly metal scavengers and the associated waste disposal fees, leading to a leaner and more economical production model. Since the reaction proceeds at room temperature, there is no requirement for energy-intensive heating or cooling systems, which substantially lowers the utility costs per kilogram of product manufactured. The high yields reported in the patent, ranging from 83% to 95%, indicate excellent atom economy, meaning that less raw material is wasted as byproduct, further driving down the effective cost of goods sold. These cumulative efficiencies allow suppliers to offer more competitive pricing structures while maintaining healthy margins, a crucial advantage in the highly price-sensitive pharmaceutical intermediate market.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as thiophenols, thionyl chloride, and triethylamine ensures a stable and resilient supply chain that is less susceptible to the geopolitical disruptions often seen with specialty catalysts. The robustness of the reaction conditions means that production can be maintained consistently across different seasons and geographic locations without the need for highly specialized climate control or infrastructure. This stability is vital for securing long-term contracts with multinational corporations that require guaranteed continuity of supply for their critical drug development pipelines. Additionally, the ease of scaling this technology from gram to ton quantities minimizes the risk of supply bottlenecks during the transition from clinical trials to commercial launch phases.
- Scalability and Environmental Compliance: The metal-free nature of this synthesis aligns perfectly with modern green chemistry principles, facilitating easier regulatory approval and reducing the environmental burden of heavy metal discharge into wastewater streams. The absence of toxic metal residues simplifies the environmental impact assessment process and lowers the cost of effluent treatment, making the facility more sustainable and socially responsible. Scalability is further enhanced by the fact that the grinding mechanism can be adapted to various reactor configurations, allowing for flexible production capacities that can respond dynamically to market demand. This adaptability ensures that the manufacturing process remains viable and compliant even as environmental regulations become increasingly stringent in the future.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this grinding synthesis technology, derived directly from the experimental data and background analysis of the patent. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific advantages and operational parameters defined in the intellectual property, ensuring accuracy and relevance for decision-makers.
Q: Does this synthesis require transition metal catalysts?
A: No, the method described in patent CN112028802B operates without transition metal catalysts, utilizing a grinding mechanism promoted by triethylamine, which eliminates the need for expensive metal removal steps.
Q: What are the typical reaction conditions for this dithioester formation?
A: The reaction proceeds efficiently at room temperature (25°C) and atmospheric pressure in dichloromethane, requiring approximately 10 hours of grinding time to achieve high yields between 83% and 95%.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the mild conditions, absence of toxic metal catalysts, and simple workup procedures make this grinding method highly scalable and compliant with strict environmental and safety regulations for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N-Dimethyl Dithioacetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free grinding synthesis described in patent CN112028802B and have integrated similar advanced methodologies into our CDMO service offerings. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of dithioester intermediate meets the highest global standards for pharmaceutical applications. Our commitment to quality and technical excellence makes us the ideal partner for companies seeking to leverage this innovative chemistry for their next-generation drug candidates.
We invite you to engage with our technical procurement team to discuss how this specific synthetic route can be tailored to your unique project requirements and timeline constraints. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this metal-free protocol for your specific target molecule. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven industrial capability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
