Advanced One-Step Synthesis of 5-Chloro-2-Methyl-4-Trifluoromethylaniline for Commercial Scale-Up
The pharmaceutical industry continuously seeks efficient pathways for constructing complex fluorinated aniline derivatives, which serve as critical building blocks for next-generation therapeutics. Patent CN112174832A introduces a groundbreaking methodology for the synthesis of 5-chloro-2-methyl-4-(trifluoromethyl)aniline, addressing long-standing challenges in yield optimization and process simplicity. This innovation represents a paradigm shift from traditional multi-step protocols to a streamlined, one-step catalytic approach that leverages advanced organometallic chemistry. By utilizing a specific palladium-catalyzed cross-coupling strategy, the technology enables the direct installation of a methyl group onto a highly functionalized aromatic core with exceptional precision. For R&D directors and process chemists, this patent offers a robust solution that minimizes waste generation while maximizing atom economy, setting a new benchmark for the manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted anilines bearing both chloro and trifluoromethyl groups has been fraught with synthetic inefficiencies and operational hazards. Conventional routes often rely on tedious sequences involving nitration, reduction, and halogenation steps, each introducing potential points of failure and impurity accumulation. These multi-step processes typically suffer from low overall yields due to the cumulative loss of material at each stage, necessitating extensive purification efforts that drive up production costs. Furthermore, the handling of reactive intermediates in traditional methods often requires stringent safety controls and specialized equipment, complicating the supply chain logistics for large-scale manufacturing. The reliance on harsh reagents and extreme conditions in older methodologies also poses significant environmental compliance challenges, making them increasingly untenable for modern green chemistry standards.
The Novel Approach
In stark contrast, the method disclosed in CN112174832A employs a sophisticated yet operationally simple one-step transformation that directly converts a readily available iodo-aniline precursor into the target methylated product. This novel approach utilizes a palladium-catalyzed cross-coupling reaction between Compound A (5-chloro-2-amino-4-trifluoromethyl iodobenzene) and Compound B (a cyclic boronate ester serving as the methyl source). The reaction proceeds smoothly in the presence of a carbonate base and a dioxane solvent system, effectively bypassing the need for intermediate isolation. 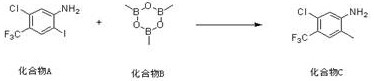 This direct methylation strategy not only drastically reduces the number of unit operations but also ensures superior control over the reaction profile, leading to consistently high purity levels. The elimination of multiple synthetic steps translates directly into reduced processing time and lower energy consumption, providing a compelling economic argument for adopting this technology in commercial settings.
This direct methylation strategy not only drastically reduces the number of unit operations but also ensures superior control over the reaction profile, leading to consistently high purity levels. The elimination of multiple synthetic steps translates directly into reduced processing time and lower energy consumption, providing a compelling economic argument for adopting this technology in commercial settings.
Mechanistic Insights into Pd-Catalyzed Methylation
The core of this technological breakthrough lies in the precise orchestration of the palladium catalytic cycle, which facilitates the difficult carbon-carbon bond formation between the aryl iodide and the methyl group. The catalyst, Pd(Dppf)Cl2, plays a pivotal role in activating the carbon-iodine bond of the substrate through oxidative addition, forming a reactive organopalladium intermediate. Subsequently, the transmetallation step involves the transfer of the methyl group from the boron species to the palladium center, a process that is critically dependent on the presence of the carbonate base to activate the boronate ester. The final reductive elimination step releases the desired 5-chloro-2-methyl-4-(trifluoromethyl)aniline product and regenerates the active palladium catalyst, allowing the cycle to continue efficiently. Understanding this mechanism is crucial for process optimization, as it highlights the importance of maintaining an inert nitrogen atmosphere to prevent catalyst deactivation and ensure consistent turnover numbers throughout the reaction duration.
From an impurity control perspective, the choice of reagents and conditions in this patent is meticulously designed to suppress side reactions that commonly plague cross-coupling chemistries. The use of a specific cyclic boronate ester minimizes the formation of homocoupling byproducts, which are often difficult to separate from the target molecule. Additionally, the moderate reaction temperature range of 100-105°C strikes an optimal balance between reaction kinetics and thermal stability, preventing the decomposition of sensitive functional groups like the trifluoromethyl moiety. The inclusion of carbonate bases such as sodium or potassium carbonate serves a dual purpose: neutralizing the acid generated during the coupling and facilitating the transmetallation step without promoting hydrolysis of the product. This careful tuning of reaction parameters results in a crude product profile that is remarkably clean, simplifying downstream purification and ensuring that the final API intermediate meets rigorous quality specifications.
How to Synthesize 5-Chloro-2-Methyl-4-(Trifluoromethyl)Aniline Efficiently
The practical implementation of this synthesis route is designed for seamless integration into existing manufacturing infrastructure, requiring standard reactor setups and common laboratory techniques. The process begins with the charging of Compound A, the palladium catalyst, and dioxane into a reactor under a protective nitrogen blanket to exclude oxygen and moisture. Following an initial stirring period to ensure homogeneous mixing, Compound B and the carbonate base are introduced, and the reaction mixture is heated to the specified temperature range. This straightforward protocol eliminates the need for exotic reagents or cryogenic conditions, making it accessible for facilities aiming to ramp up production of this valuable intermediate without significant capital expenditure on new equipment.
- Charge Compound A, Pd(Dppf)Cl2 catalyst, and dioxane into a reactor under nitrogen protection.
- Add Compound B and carbonate base, then heat the mixture to 100-105°C.
- Stir for 15-20 hours, filter through diatomite, concentrate, and purify to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this one-step synthesis method offers transformative benefits that extend far beyond simple chemical yield improvements. The consolidation of multiple synthetic steps into a single operation fundamentally alters the cost structure of manufacturing, reducing the demand for solvents, reagents, and labor hours associated with intermediate handling. By streamlining the production workflow, companies can achieve substantial cost savings in pharmaceutical intermediate manufacturing, allowing for more competitive pricing strategies in the global market. The simplified process flow also mitigates supply chain risks by reducing the number of critical raw materials required, thereby decreasing the vulnerability to disruptions in the sourcing of specialized reagents.
- Cost Reduction in Manufacturing: The elimination of transition metal removal steps often required in other coupling reactions, combined with the high efficiency of the Pd(Dppf)Cl2 catalyst, leads to a drastic reduction in overall production costs. The high yield reported in the patent examples implies that less raw material is wasted per kilogram of finished product, directly improving the gross margin for manufacturers. Furthermore, the ability to use inexpensive carbonate bases instead of more costly organic bases contributes to a leaner bill of materials, enhancing the economic viability of the process on a multi-ton scale.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials such as the iodo-aniline precursor and the boronate ester ensures a stable and predictable supply chain. Unlike proprietary reagents that may be subject to exclusive supply agreements or long lead times, the inputs for this reaction are sourced from a broad network of chemical suppliers. This diversity in sourcing options empowers procurement teams to negotiate better terms and secure inventory buffers, ensuring continuous production schedules even during periods of market volatility. The robustness of the reaction conditions further guarantees consistent output quality, reducing the frequency of batch failures that can disrupt delivery timelines.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively with straightforward heating and stirring protocols that translate easily from pilot plant to full commercial production. The use of dioxane as a solvent, while requiring appropriate recovery systems, is well-understood in industrial settings, and the minimal generation of hazardous byproducts simplifies waste treatment procedures. This alignment with green chemistry principles not only reduces environmental liability but also positions the manufacturer favorably regarding increasingly strict regulatory frameworks governing chemical production emissions and effluent discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the detailed specifications of the patent documentation. These insights are intended to provide clarity for technical teams evaluating the feasibility of integrating this route into their current manufacturing portfolios.
Q: What is the primary advantage of the one-step synthesis method described in CN112174832A?
A: The primary advantage is the direct conversion of the iodo-aniline precursor to the methylated product in a single step, eliminating complex multi-step sequences and significantly improving overall yield and purity.
Q: Which catalyst system is utilized for this transformation?
A: The process utilizes Pd(Dppf)Cl2 as the catalyst, paired with a carbonate base such as sodium or potassium carbonate in a dioxane solvent system.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method features mild reaction conditions (100-105°C), readily available raw materials, and simple post-reaction workup, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Chloro-2-Methyl-4-(Trifluoromethyl)Aniline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable supply chains for high-performance pharmaceutical intermediates like 5-chloro-2-methyl-4-(trifluoromethyl)aniline. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch against the highest industry standards. Our capability to adapt and optimize patented processes allows us to offer a stable supply of this key intermediate, bridging the gap between innovative academic research and industrial reality.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this efficient one-step method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your development timeline remains on track with a trusted partner dedicated to excellence in fine chemical manufacturing.
