Advanced One-Pot Synthesis of Vilanterol Intermediate Fumarate for Scalable API Manufacturing
Introduction to Next-Generation Vilanterol Intermediate Manufacturing
The pharmaceutical landscape for Chronic Obstructive Pulmonary Disease (COPD) treatments continues to evolve, driven by the demand for high-purity Long-Acting Beta-Agonists (LABAs) like Vilanterol. A pivotal advancement in this domain is detailed in Chinese Patent CN115745945A, which discloses a revolutionary one-pot method for preparing the fumarate salt of a key Vilanterol intermediate. This technology addresses critical bottlenecks in traditional synthesis, offering a pathway to significantly enhanced product quality and process efficiency. Unlike conventional multi-step procedures that rely on complex purification techniques, this novel approach streamlines the production of the compound of Formula II fumarate, achieving exceptional purity levels exceeding 99.7% with minimal isomeric impurities. For global pharmaceutical manufacturers, this represents a strategic opportunity to optimize their supply chains for high-value respiratory APIs.
The core innovation lies in the direct conversion of the oxazolidinone precursor (Formula I) into the stable fumarate salt without isolating the free base intermediate. By leveraging specific alkaline conditions followed by immediate salification with fumaric acid, the process eliminates the need for hazardous reagents and extensive solvent usage associated with legacy methods. This technical breakthrough not only aligns with green chemistry principles by reducing the discharge of three wastes but also ensures a robust and repeatable manufacturing protocol. As the industry moves towards more sustainable and cost-effective production models, the adoption of such advanced synthetic routes becomes a competitive necessity for maintaining supply continuity and regulatory compliance.
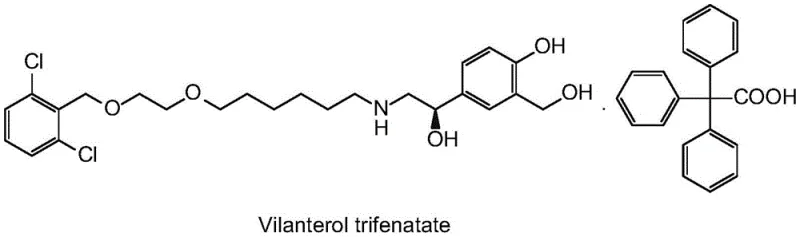
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Vilanterol intermediates has been plagued by inefficiencies and environmental burdens. Prior art, such as the methods described in international patent WO2003024439A1, typically involves a ring-opening reaction using potassium trimethylsilanolate followed by a separate deprotection step with hydrochloric acid. This multi-stage process necessitates the isolation of the free base (Formula III) before subsequent salification with triphenylacetic acid. Such workflows are inherently cumbersome, requiring multiple extraction cycles, solvent swaps, and often column chromatography to achieve acceptable purity. The reliance on these intensive purification steps not only inflates production costs but also introduces significant variability in yield and impurity profiles.
Furthermore, alternative salt forms like succinates or citrates, while attempting to improve stability, often fail to resolve the underlying process complexities. For instance, methods disclosed in CN111807973A for succinate salts still require extraction with large volumes of organic solvents like ethyl acetate and concentration steps that generate substantial waste liquid. The use of reducing agents like sodium borohydride in some prior routes also introduces safety hazards due to explosion risks. These conventional approaches result in lower overall yields, higher isomer content, and a larger environmental footprint, making them increasingly untenable for modern, large-scale industrial applications where efficiency and sustainability are paramount.
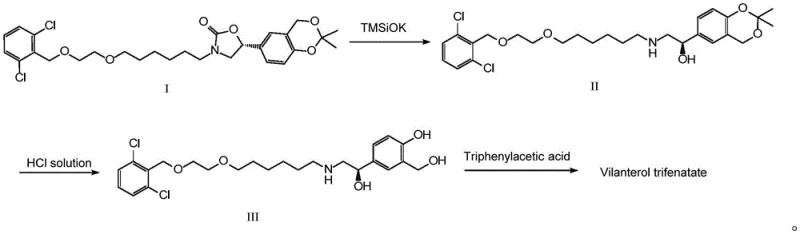
The Novel Approach
In stark contrast, the one-pot method disclosed in CN115745945A offers a paradigm shift in process design. This innovative route combines the ring-opening reaction and salt formation into a single, seamless operation. By reacting the Formula I precursor with a base such as potassium hydroxide or sodium ethoxide in an alcoholic solvent, the oxazolidinone ring is efficiently opened to generate the intermediate in situ. Instead of isolating this intermediate, fumaric acid is directly added to the reaction mixture to adjust the pH to a precise range of 6-7. This triggers the immediate precipitation of the fumarate salt of Formula II, bypassing the need for extraction, concentration, or chromatographic purification entirely.
The advantages of this streamlined approach are manifold. Firstly, it drastically reduces the consumption of organic solvents, as the reaction medium itself serves as the crystallization environment. Secondly, the direct precipitation yields a product with superior purity (99.7-100.0%) and exceptionally low isomer content (0.01-0.03%), outperforming traditional triphenylacetate or succinate salts. The simplicity of the operation—essentially a heat, cool, and filter sequence—enhances process robustness and repeatability. This method not only lowers the barrier for commercial scale-up but also significantly mitigates the environmental impact by minimizing waste discharge, positioning it as the preferred choice for forward-thinking chemical manufacturers.
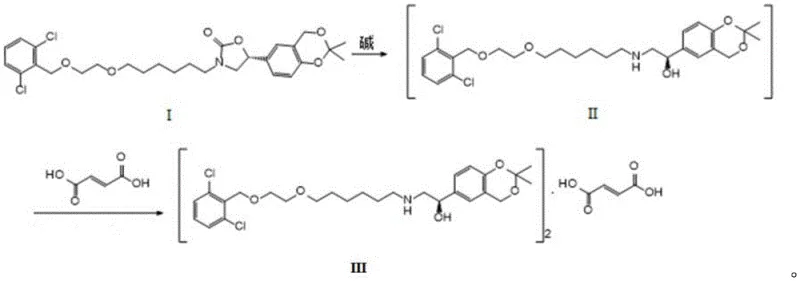
Mechanistic Insights into Base-Catalyzed Ring Opening and Salification
The chemical elegance of this process stems from the precise control of reaction kinetics and thermodynamics during the base-catalyzed ring opening. The oxazolidinone ring in Formula I is susceptible to nucleophilic attack by alkoxide ions generated from bases like potassium hydroxide or sodium ethoxide in alcoholic media. This attack cleaves the carbamate linkage, releasing the secondary amine and the hydroxyl group essential for the biological activity of the final API. The choice of base is critical; strong alkalis like KOH or NaOEt provide the necessary nucleophilicity to drive the reaction to completion at moderate temperatures (50-100°C), while avoiding the harsh conditions that could lead to degradation or racemization of the chiral centers.
Following the ring opening, the introduction of fumaric acid serves a dual purpose: quenching the excess base and forming the stable salt. Fumaric acid, being a dicarboxylic acid with low solubility in many organic solvents, acts as an effective precipitating agent. As the pH is adjusted to 6-7, the protonated amine of the intermediate forms an ionic bond with the fumarate anion. This interaction promotes the formation of a highly ordered crystal lattice, identified as Form A, which is characterized by specific X-ray powder diffraction peaks at 2θ angles of 4.6°, 9.2°, 14.0°, and 23.4°. This crystalline stability is crucial for preventing the formation of amorphous impurities and ensures that the isomeric purity is locked in, preventing epimerization during storage or subsequent processing steps.
Impurity control is further enhanced by the avoidance of aqueous workups and extractions. In traditional methods, partitioning between aqueous and organic phases can sometimes lead to the retention of polar impurities or the loss of product. By keeping the system homogeneous until the point of crystallization, the one-pot method ensures that impurities remain in the mother liquor while the pure product precipitates. The thermal profile, maintained between 0-40°C during acid addition, prevents thermal stress on the molecule, preserving the integrity of the sensitive phenolic and hydroxymethyl functionalities. This meticulous control over the reaction environment is what enables the consistent production of high-purity material suitable for stringent pharmaceutical applications.
How to Synthesize Vilanterol Intermediate Fumarate Efficiently
Implementing this advanced synthetic route requires careful attention to reagent stoichiometry and temperature control to maximize yield and purity. The process begins with the dissolution of the Formula I precursor in a suitable alcohol solvent, such as isopropanol or ethanol, followed by the addition of the base catalyst. The reaction mixture is then heated to facilitate the ring-opening transformation. Once the conversion is complete, typically monitored by HPLC, the mixture is cooled, and fumaric acid is introduced to induce crystallization. The resulting solid is filtered and dried to afford the final product. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Perform ring-opening reaction on the oxazolidinone precursor (Formula I) using an alkali catalyst such as potassium hydroxide or sodium ethoxide in an alcohol solvent like isopropanol at 50-100°C.
- Upon completion, directly add fumaric acid to the reaction mixture to adjust the pH to 6-7, inducing precipitation of the fumarate salt without intermediate extraction steps.
- Filter and dry the precipitated crystals to obtain the final fumarate salt with purity exceeding 99.7% and minimal isomer impurities.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this one-pot synthesis method offers tangible strategic benefits that extend beyond mere technical superiority. The elimination of complex purification steps such as column chromatography and multiple liquid-liquid extractions translates directly into reduced operational expenditures. By removing the need for large volumes of extraction solvents like ethyl acetate and the associated recovery infrastructure, manufacturers can achieve significant cost reductions in raw material procurement and waste management. Furthermore, the simplified workflow reduces the manpower and equipment time required per batch, enhancing overall plant throughput and asset utilization without compromising on quality standards.
Supply chain reliability is another critical area where this technology excels. The robustness of the one-pot process minimizes the risk of batch failures due to operational complexity, ensuring a more consistent and predictable output of high-purity intermediates. The use of common, non-hazardous reagents like potassium hydroxide and fumaric acid reduces dependency on specialized or regulated chemicals, thereby mitigating supply risks associated with raw material availability. Additionally, the high stability of the fumarate salt form facilitates easier storage and transportation, reducing the likelihood of degradation during logistics and extending the shelf life of inventory. This reliability is essential for maintaining uninterrupted production schedules for downstream API manufacturing.
From an environmental and regulatory perspective, the drastic reduction in solvent usage and waste generation aligns perfectly with global sustainability goals and increasingly stringent environmental regulations. The process generates significantly less hazardous waste compared to traditional routes, lowering the costs and complexities associated with waste disposal and environmental compliance. This green chemistry approach not only enhances the corporate social responsibility profile of the manufacturer but also future-proofs the supply chain against potential regulatory tightening. The combination of cost efficiency, supply reliability, and environmental stewardship makes this method a compelling choice for long-term partnerships in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Vilanterol intermediate fumarate. These insights are derived from the detailed experimental data and comparative analysis presented in the patent literature, providing a clear understanding of the method's capabilities and advantages. Understanding these nuances is vital for stakeholders evaluating the feasibility of integrating this intermediate into their existing manufacturing portfolios.
Q: What is the primary advantage of the one-pot fumarate synthesis method over traditional routes?
A: The primary advantage is the elimination of post-reaction extraction and column chromatography purification steps. By directly adjusting pH with fumaric acid in the reaction pot, the process significantly reduces solvent consumption and waste generation while achieving higher purity (>99.7%) and yield compared to prior art methods involving triphenylacetic acid or succinic acid salts.
Q: How does this method control isomeric impurities in the Vilanterol intermediate?
A: The method utilizes specific base catalysts like potassium hydroxide or sodium ethoxide in alcoholic solvents (e.g., isopropanol) at controlled temperatures (55-75°C). This optimized condition facilitates selective ring opening while minimizing racemization, resulting in isomer content as low as 0.01-0.03%, which is superior to conventional succinate or triphenylacetate salt forms.
Q: Is the fumarate salt form suitable for long-term storage and industrial scaling?
A: Yes, the fumarate salt exhibits excellent stability and physical properties, characterized by distinct X-ray powder diffraction peaks (e.g., at 2θ = 4.6°, 9.2°) and a sharp endothermic peak in DSC analysis around 140-146°C. These characteristics confirm a stable crystal lattice (Form A) that is easy to filter, dry, and store, making it highly suitable for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vilanterol Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving respiratory medications. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering Vilanterol intermediates with stringent purity specifications, utilizing rigorous QC labs to verify every batch against the highest international standards. Our state-of-the-art facilities are equipped to handle the specific requirements of base-catalyzed reactions and crystallization processes, guaranteeing a supply of material that meets the exacting demands of global pharmaceutical partners.
We invite you to collaborate with us to leverage these advanced synthetic technologies for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. By partnering with us, you gain access to not just a product, but a comprehensive solution that includes specific COA data and route feasibility assessments. Contact us today to discuss how we can support your supply chain with reliable, high-purity Vilanterol intermediates produced via cutting-edge, sustainable methods.
