Advanced Synthesis of S-3-Chloro-D-Alanine Methyl Ester Hydrochloride for Commercial API Production
The pharmaceutical industry is constantly seeking robust pathways for critical antibiotic intermediates, particularly those serving dual purposes in treating tuberculosis and emerging neurological indications. Patent CN116332780A introduces a significant technological breakthrough in the synthesis of S-3-chloro-D-alanine methyl ester hydrochloride, a pivotal precursor for the antibiotic Cycloserine. This novel methodology addresses long-standing inefficiencies in traditional manufacturing by replacing hazardous reagents and energy-intensive separation techniques with a streamlined, thionyl chloride-mediated protocol. For R&D directors and procurement strategists, this patent represents a shift towards safer, higher-yielding processes that directly impact the bottom line through reduced operational complexity and enhanced product consistency. The ability to produce high-purity intermediates without relying on dangerous hydrogen chloride gas or violent phosphorus reagents marks a substantial evolution in amino acid derivative synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of S-3-chloro-D-alanine methyl ester hydrochloride has been plagued by significant safety hazards and operational bottlenecks that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Traditional routes often relied heavily on phosphorus pentachloride for the chlorination step, a reagent known for its violent reactivity and the generation of substantial phosphorous waste byproducts that are notoriously difficult to separate from the final API intermediate. Furthermore, earlier methodologies frequently necessitated the use of gaseous hydrogen chloride for esterification, requiring specialized equipment for gas handling and nitrogen replacement operations to maintain anhydrous conditions, thereby increasing capital expenditure and safety risks. Another critical drawback in prior art involves the reliance on rotary evaporation for isolating intermediates, a process that is not only energy-intensive but also poses thermal degradation risks to sensitive chiral molecules, ultimately compromising the optical purity and overall yield of the final product.
The Novel Approach
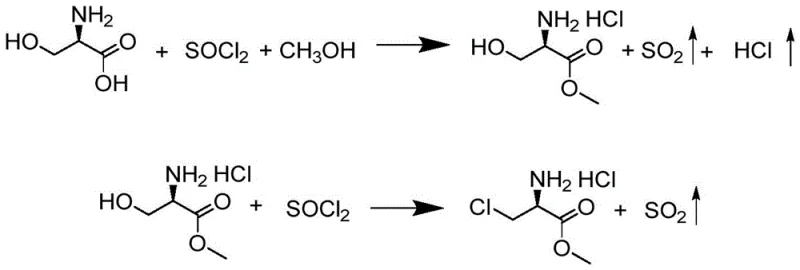
The innovative strategy outlined in patent CN116332780A fundamentally reengineers the synthetic route by utilizing thionyl chloride as a dual-function reagent for both esterification and chlorination under mild, controllable conditions. This approach eliminates the need for hazardous hydrogen chloride gas and avoids the chaotic side reactions associated with phosphorus pentachloride, resulting in a cleaner reaction profile with significantly reduced impurity loads. A key differentiator of this novel approach is the implementation of direct crystallization techniques using ethyl acetate and methanol, which bypasses the need for rotary evaporation entirely, thus preserving the structural integrity of the chiral center while drastically simplifying the workup procedure. By optimizing solvent ratios and temperature profiles, this method ensures that unreacted raw materials are consumed in subsequent steps rather than being lost during isolation, leading to a more atom-economical process that is inherently safer and more conducive to continuous industrial production.
Mechanistic Insights into Thionyl Chloride-Mediated Esterification and Chlorination
The chemical transformation described in this patent proceeds through a sophisticated two-stage mechanism where thionyl chloride acts first as an esterifying agent and subsequently as a chlorinating reagent, leveraging its ability to convert hydroxyl groups into good leaving groups. In the initial stage, D-serine reacts with thionyl chloride in methanol at controlled low temperatures to form D-serine methyl ester hydrochloride, where the carboxylic acid is converted to a methyl ester while the amine is protected as a hydrochloride salt. The subsequent chlorination step involves the activation of the beta-hydroxyl group by thionyl chloride in a dichloromethane medium, facilitating a nucleophilic substitution that installs the chlorine atom with retention or inversion depending on the specific mechanistic pathway, ultimately yielding the target S-3-chloro-D-alanine derivative. The precise control of molar ratios between thionyl chloride and the substrate is critical here, as it prevents over-chlorination at the alpha-position, a common side reaction that can severely degrade the quality of the pharmaceutical intermediate.
Impurity control in this synthesis is achieved through a combination of stoichiometric precision and strategic crystallization protocols that exploit solubility differences between the product and potential byproducts. The patent specifies that limiting the volume-to-mass ratio of solvents like methanol and dichloromethane ensures that the reaction mixture remains homogeneous enough for efficient conversion while allowing for selective precipitation of the product upon cooling. Furthermore, the addition of methanol in the second step serves a dual purpose: it quenches excess thionyl chloride to reduce acidic waste discharge and simultaneously acts as a recrystallization solvent to dissolve residual impurities while precipitating the pure hydrochloride salt. This mechanistic understanding allows process chemists to fine-tune parameters such as cooling rates and stirring times to maximize the rejection of sulfide contaminants and unreacted starting materials, ensuring that the final liquid phase purity consistently exceeds 94%.
How to Synthesize S-3-Chloro-D-Alanine Methyl Ester Hydrochloride Efficiently
Implementing this synthesis requires strict adherence to the temperature gradients and solvent addition sequences defined in the patent to ensure reproducibility and safety at scale. The process begins with the careful addition of thionyl chloride to a methanol suspension of D-serine at low temperatures to manage exothermicity, followed by a prolonged reaction period to ensure complete esterification before inducing crystallization with ethyl acetate. The detailed standardized synthesis steps see the guide below for exact operational parameters regarding reagent addition rates, specific temperature hold times, and filtration protocols necessary to achieve the reported high yields.
- Esterify D-serine in methanol with thionyl chloride at controlled temperatures, followed by ethyl acetate crystallization.
- React the resulting ester hydrochloride with thionyl chloride in dichloromethane, followed by methanol reflux and cooling crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers profound advantages in terms of cost reduction in API manufacturing and supply chain reliability. By eliminating the need for rotary evaporation and complex gas handling systems, the process significantly reduces energy consumption and equipment maintenance costs, translating into a more competitive pricing structure for the final intermediate. The simplification of the workflow, characterized by fewer unit operations and the avoidance of hazardous reagent handling, also minimizes the risk of production delays caused by safety incidents or equipment failures, thereby enhancing the overall resilience of the supply chain. Additionally, the high yield and purity reported in the patent examples suggest a substantial reduction in raw material waste, allowing manufacturers to optimize their inventory management and reduce the environmental footprint associated with waste disposal.
- Cost Reduction in Manufacturing: The elimination of energy-intensive rotary evaporation steps and the replacement of expensive or hazardous reagents like phosphorus pentachloride with cost-effective thionyl chloride leads to significant operational savings. The process design minimizes solvent usage through optimized volume-to-mass ratios, reducing the costs associated with solvent purchase, recovery, and disposal, while the high conversion efficiency ensures that raw material costs are maximized in the final product value.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route, which operates under mild conditions and utilizes common industrial solvents like methanol and dichloromethane, ensures consistent production output even in fluctuating market conditions. By avoiding reagents that require special storage or handling permits, such as gaseous hydrogen chloride, facilities can maintain uninterrupted production schedules, reducing lead time for high-purity pharmaceutical intermediates and ensuring timely delivery to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is designed for easy scale-up in standard glass-lined reactors without the need for specialized high-pressure or cryogenic equipment, facilitating rapid capacity expansion to meet market demand. Furthermore, the generation of gaseous byproducts like sulfur dioxide and hydrogen chloride, which are easily scrubbed, combined with the reduction of solid phosphorous waste, aligns the manufacturing process with stringent environmental regulations, mitigating compliance risks and potential fines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, derived directly from the comparative data and beneficial effects described in the patent documentation. These insights are intended to clarify the operational benefits and technical feasibility for stakeholders evaluating this technology for potential licensing or procurement partnerships.
Q: Why is thionyl chloride preferred over phosphorus pentachloride for this synthesis?
A: Thionyl chloride offers milder reaction conditions and generates gaseous byproducts (SO2, HCl) that are easily removed, whereas phosphorus pentachloride reacts violently and produces difficult-to-remove phosphorous byproducts.
Q: How does this patent improve product purity compared to conventional methods?
A: The method utilizes specific solvent crystallization techniques with ethyl acetate and methanol reflux, avoiding rotary evaporation which can degrade heat-sensitive intermediates, resulting in purity greater than 94%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process eliminates complex operations like nitrogen replacement and rotary evaporation, using standard glass-lined reactors and simple filtration, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-3-Chloro-D-Alanine Methyl Ester Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of life-saving antibiotics and neurological therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory patent data to full-scale manufacturing is seamless and compliant with international standards. We are committed to delivering products with stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of S-3-chloro-D-alanine methyl ester hydrochloride meets the exacting requirements of modern pharmaceutical synthesis.
We invite you to collaborate with us to leverage this advanced synthetic technology for your supply chain needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can drive efficiency and reliability in your production of cycloserine and related therapeutic agents.
