Revolutionizing Phenazine Production: A Green Aqueous Copper-Catalyzed Route for Commercial Scale-up
Revolutionizing Phenazine Production: A Green Aqueous Copper-Catalyzed Route for Commercial Scale-up
The pharmaceutical and agrochemical industries are constantly seeking sustainable methodologies that align with green chemistry principles without compromising on yield or purity. Patent CN102206189A introduces a groundbreaking approach for the preparation of phenazine compounds by catalyzing o-halogenated anilines in a pure aqueous phase. This technology represents a significant paradigm shift from traditional organic solvent-based syntheses, utilizing water-soluble coordination compounds as efficient catalysts to drive the cyclization reaction. By leveraging the unique properties of water as a reaction medium, this invention offers an environmentally friendly, operationally simple, safe, and cost-effective pathway for generating high-value phenazine derivatives. The method is particularly notable for its ability to accommodate a wide range of functional groups while maintaining high selectivity and yield, addressing critical pain points in the manufacturing of complex heterocyclic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phenazine and its derivatives has relied on methodologies that are increasingly viewed as obsolete in the context of modern sustainable manufacturing. Early approaches, such as the condensation of catechol and o-phenylenediamine, often suffered from low yields and the formation of complex by-product mixtures that were difficult to separate. Other traditional routes involving the ring-closing of 2-amino or nitrodiphenylamines typically required harsh reaction conditions, including high temperatures and the use of expensive, toxic organic solvents. These conventional processes frequently necessitated the protection and deprotection of functional groups, adding unnecessary steps that increased both material costs and waste generation. Furthermore, the reliance on heavy metal catalysts in non-aqueous media often led to significant environmental pollution and posed serious safety risks due to the flammability and volatility of the solvents used. The difficulty in controlling these reactions often resulted in inconsistent product quality, making scale-up a challenging and risky endeavor for procurement and supply chain teams.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN102206189A utilizes a novel aqueous phase reaction system that fundamentally simplifies the production workflow. This approach employs water-soluble metal copper complexes to efficiently catalyze the coupling of o-halogenated anilines, eliminating the need for hazardous organic solvents entirely. 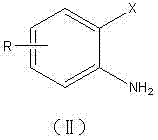 As illustrated by the substrate scope, the method is robust enough to handle various substituents such as hydrogen, chlorine, nitro, methoxy, methyl, and acetyl groups at different positions on the aromatic ring. The reaction proceeds under relatively mild conditions, typically between 60°C and 130°C, which significantly reduces energy consumption compared to high-temperature fusion methods. The simplicity of the operation allows for straightforward product isolation, often requiring only extraction and concentration, which streamlines the purification process. This novel route not only enhances safety by removing flammable solvents but also ensures a single major product profile, thereby facilitating easier downstream processing and quality control.
As illustrated by the substrate scope, the method is robust enough to handle various substituents such as hydrogen, chlorine, nitro, methoxy, methyl, and acetyl groups at different positions on the aromatic ring. The reaction proceeds under relatively mild conditions, typically between 60°C and 130°C, which significantly reduces energy consumption compared to high-temperature fusion methods. The simplicity of the operation allows for straightforward product isolation, often requiring only extraction and concentration, which streamlines the purification process. This novel route not only enhances safety by removing flammable solvents but also ensures a single major product profile, thereby facilitating easier downstream processing and quality control.
Mechanistic Insights into Copper-Catalyzed Cyclization in Water
The core of this technological advancement lies in the specific design and application of the water-soluble copper catalyst system. The patent highlights two primary catalytic species: an in-situ catalyst formed from copper sulfate and pyridine-2-carboxylate, and a pre-formed water-soluble metal copper complex (I). 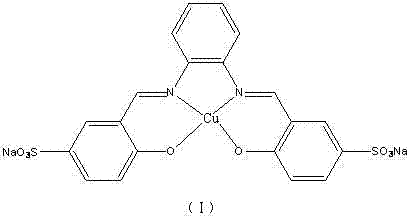 This complex features a specific coordination environment that stabilizes the copper center in the aqueous medium, preventing precipitation and maintaining high catalytic activity throughout the reaction cycle. The mechanism likely involves the oxidative addition of the o-halogenated aniline to the copper center, followed by intramolecular nucleophilic attack by the amine nitrogen to form the pyrazine ring. The presence of the base, such as potassium carbonate or sodium hydroxide, plays a crucial role in deprotonating the intermediate species, driving the equilibrium towards the formation of the phenazine product. The water molecules in the solvent shell may also participate in stabilizing transition states through hydrogen bonding, enhancing the reaction rate and selectivity. This sophisticated interplay between the ligand, the metal center, and the aqueous environment allows for the activation of carbon-halogen bonds under much milder conditions than traditionally possible.
This complex features a specific coordination environment that stabilizes the copper center in the aqueous medium, preventing precipitation and maintaining high catalytic activity throughout the reaction cycle. The mechanism likely involves the oxidative addition of the o-halogenated aniline to the copper center, followed by intramolecular nucleophilic attack by the amine nitrogen to form the pyrazine ring. The presence of the base, such as potassium carbonate or sodium hydroxide, plays a crucial role in deprotonating the intermediate species, driving the equilibrium towards the formation of the phenazine product. The water molecules in the solvent shell may also participate in stabilizing transition states through hydrogen bonding, enhancing the reaction rate and selectivity. This sophisticated interplay between the ligand, the metal center, and the aqueous environment allows for the activation of carbon-halogen bonds under much milder conditions than traditionally possible.
From an impurity control perspective, this aqueous catalytic system offers distinct advantages over organic phase reactions. The high polarity of water tends to suppress side reactions that are common in non-polar organic solvents, such as homocoupling of the aryl halide or over-alkylation. The selectivity of the copper complex ensures that the cyclization occurs specifically at the ortho-position relative to the amine, minimizing the formation of regio-isomers. Additionally, the use of water facilitates the removal of inorganic salts and polar by-products during the workup phase, as these remain in the aqueous layer while the organic phenazine product is extracted. This inherent partitioning behavior significantly reduces the burden on purification columns and crystallization steps, leading to a cleaner final product with a superior impurity profile. For R&D directors, this means a more predictable synthesis with fewer unknown peaks in the chromatogram, accelerating the timeline from lab scale to clinical supply.
How to Synthesize Phenazine Derivatives Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific parameters to maximize efficiency and yield. The process begins with the precise weighing of the o-halogenated aniline substrate, the chosen copper catalyst, and the inorganic base, which are then suspended or dissolved in deionized water. The reaction mixture is heated to the optimal temperature range, typically around 120°C, and maintained under stirring for a period sufficient to achieve full conversion, usually between 10 to 40 hours depending on the substrate reactivity. Detailed standard operating procedures regarding stoichiometry, temperature ramping, and extraction protocols are essential for reproducibility. For a comprehensive guide on the exact experimental conditions and workup procedures, please refer to the standardized synthesis steps outlined below.
- Prepare the reaction mixture by combining o-halogenated aniline substrate, a water-soluble copper catalyst (such as Complex I or in-situ CuSO4/pyridine-2-carboxylate), and an inorganic base like potassium carbonate in pure water.
- Heat the aqueous reaction system to a temperature range of 60°C to 130°C and maintain stirring for a duration of 10 to 40 hours to ensure complete conversion.
- Upon completion, cool the mixture to room temperature, extract the product using ethyl acetate, concentrate under reduced pressure, and purify via column chromatography to obtain the high-purity phenazine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this aqueous-phase technology offers substantial strategic benefits that extend beyond mere technical feasibility. The elimination of expensive and regulated organic solvents translates directly into a significant reduction in raw material costs and waste disposal fees. By replacing volatile organic compounds with water, the process inherently mitigates the risks associated with solvent storage, handling, and recovery, leading to a safer working environment and lower insurance premiums. The simplified workup procedure, which avoids complex distillation or extensive chromatographic purification, reduces the overall processing time and equipment occupancy, thereby increasing throughput capacity. These factors combine to create a more resilient and cost-efficient supply chain capable of responding rapidly to market demands for phenazine intermediates.
- Cost Reduction in Manufacturing: The economic impact of switching to an aqueous system is profound, primarily driven by the drastic reduction in solvent costs and waste treatment expenses. Traditional organic synthesis often requires large volumes of solvents that must be purchased, stored, and subsequently recovered or disposed of, all of which incur significant operational expenditures. In this new method, water serves as a cheap, non-toxic, and non-flammable medium that requires minimal processing before discharge or recycling. Furthermore, the high catalytic efficiency allows for lower catalyst loading in many cases, reducing the consumption of precious metal resources. The avoidance of protection and deprotection steps further trims the bill of materials, resulting in a leaner and more cost-effective manufacturing process that improves overall profit margins.
- Enhanced Supply Chain Reliability: Relying on water as a solvent removes the supply chain vulnerabilities associated with petrochemical-derived organic solvents, whose prices and availability can fluctuate wildly with crude oil markets. The raw materials for this process, specifically o-halogenated anilines and simple copper salts, are commodity chemicals with robust and established global supply networks. This stability ensures consistent production schedules and reduces the risk of delays caused by raw material shortages. Additionally, the safety profile of the process allows for manufacturing in a wider range of facilities without requiring specialized explosion-proof infrastructure, expanding the potential supplier base and enhancing supply continuity. This reliability is critical for long-term contracts and just-in-time delivery models required by major pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling up chemical processes often introduces new challenges, but the aqueous nature of this reaction simplifies the transition from gram to ton scale. Heat transfer and mixing are generally more manageable in water than in viscous organic media, reducing the engineering hurdles associated with reactor design. From an environmental compliance standpoint, this method aligns perfectly with increasingly stringent global regulations regarding VOC emissions and hazardous waste generation. The "green" credentials of the process facilitate easier regulatory approval and enhance the corporate sustainability profile of the manufacturer. This environmental compatibility future-proofs the supply chain against tightening ecological laws, ensuring long-term operational viability without the need for costly retrofits or process changes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aqueous copper-catalyzed synthesis. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance. Understanding these details is crucial for evaluating the feasibility of adopting this technology for your specific production needs. We encourage technical teams to review these points carefully to assess the alignment with their current manufacturing capabilities.
Q: What are the primary advantages of using water as a solvent for phenazine synthesis compared to organic solvents?
A: Using water eliminates the need for volatile organic compounds (VOCs), drastically reducing environmental pollution and safety hazards associated with flammability. Furthermore, water simplifies the workup process as products often precipitate or can be easily extracted, avoiding complex solvent recovery systems required in traditional organic synthesis.
Q: Can this copper-catalyzed method tolerate various functional groups on the aniline substrate?
A: Yes, the method demonstrates excellent functional group tolerance. The patent data indicates successful synthesis with substrates containing methyl, nitro, chloro, methoxy, and ethoxycarbonyl groups, making it highly versatile for producing diverse phenazine libraries for drug discovery.
Q: Is the catalyst system suitable for large-scale industrial production?
A: Absolutely. The use of inexpensive copper salts or stable water-soluble complexes, combined with the safety and low cost of water as a medium, makes this process inherently scalable. The absence of strict anhydrous conditions further lowers the barrier for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenazine Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the aqueous copper-catalyzed synthesis described in CN102206189A for the production of high-quality phenazine compounds. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical intermediates. Our facilities are equipped with state-of-the-art rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch meets the highest industry standards for pharmaceutical and agrochemical applications. We are committed to leveraging this green chemistry innovation to deliver superior value to our global partners.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in phenazine chemistry can support your next breakthrough project.
