Advanced Green Esterification Process for High-Purity Trimethylolpropane Triacrylate Manufacturing
The global demand for high-performance photo-cured coatings continues to surge, driving the need for superior monomers like Trimethylolpropane Triacrylate (TMPTA), often commercially known as Viscoat 295. A pivotal advancement in this sector is detailed in patent CN103664599A, which discloses a robust preparation method for high-purity TMPTA that fundamentally reimagines the traditional esterification landscape. This technology leverages trimethylolpropane (TMP) as the core polyol substrate, reacting it with acrylic acid in the presence of specialized water-carrying agents, advanced catalysts, and polymerization inhibitors. The result is a streamlined process that consistently delivers product purity exceeding 98% and yields greater than 95%, addressing critical bottlenecks in the supply chain for reliable coating material suppliers. By shifting away from hazardous aromatic solvents and corrosive acid chlorides, this method offers a sustainable pathway for cost reduction in coating manufacturing while ensuring the structural integrity required for high-end applications.
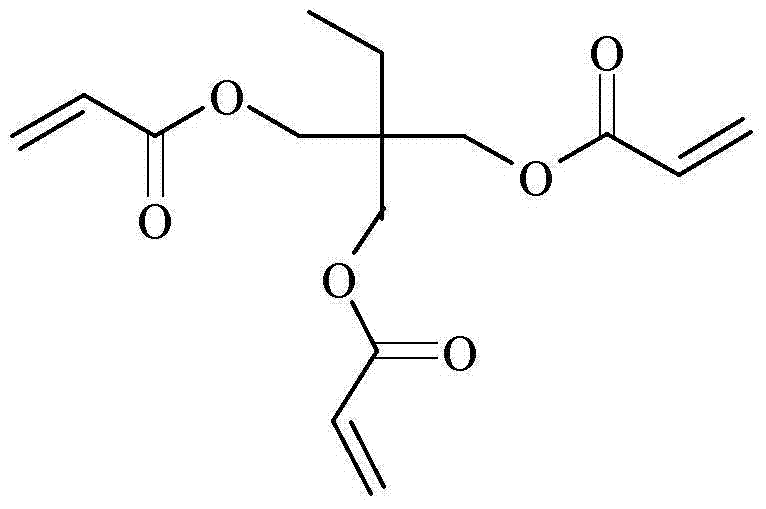
The structural complexity of TMPTA, characterized by its trifunctional acrylate groups attached to a neopentyl backbone, necessitates precise synthetic control to prevent premature polymerization and ensure high conversion rates. As illustrated in the molecular diagram, the three vinyl groups are highly reactive, making the choice of reaction conditions paramount to avoiding gelation during synthesis. The patent highlights that achieving this specific architecture with high fidelity requires not just stoichiometric precision but also a carefully curated environment that suppresses radical formation while promoting ester bond formation. This balance is the cornerstone of producing high-purity OLED material precursors and advanced polymer additives where even trace impurities can compromise the performance of the final cured film.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of TMPTA has been plagued by significant environmental and operational challenges that hinder efficient commercial scale-up of complex polymer additives. Traditional methods often relied on benzene or toluene as azeotropic agents to remove water, introducing severe toxicity risks to personnel and creating substantial regulatory burdens for waste disposal. Furthermore, alternative routes utilizing acryloyl chloride generated copious amounts of hydrogen chloride gas, leading to severe equipment corrosion and necessitating expensive scrubbing systems to meet environmental compliance standards. These legacy processes typically operated at higher temperatures, often exceeding 110°C, which increased energy consumption and promoted thermal degradation of the sensitive acrylic double bonds, ultimately capping the achievable purity and yield. The reliance on liquid strong acids as catalysts also complicated the downstream purification process, requiring extensive neutralization and washing steps that generated large volumes of saline wastewater.
The Novel Approach
The methodology presented in CN103664599A represents a paradigm shift by substituting toxic aromatics with safer C6-C7 alkanes such as cyclohexane, n-hexane, or petroleum ether. This substitution not only mitigates health hazards but also optimizes the boiling point range for effective water removal at lower temperatures, specifically between 80°C and 100°C. By employing solid acid catalysts like PEP-15u molecular sieves or specific metal methanesulfonates, the reaction achieves high conversion rates without the corrosive aftermath associated with mineral acids. The integration of effective polymerization inhibitors, such as thiodiphenylamine, directly into the reaction matrix ensures that the acrylic double bonds remain intact throughout the heating cycle. This holistic approach simplifies the workup procedure, eliminates the generation of acidic gases, and aligns perfectly with the principles of green chemistry, thereby reducing lead time for high-purity coating intermediates by streamlining the purification workflow.
Mechanistic Insights into Green Esterification Catalysis
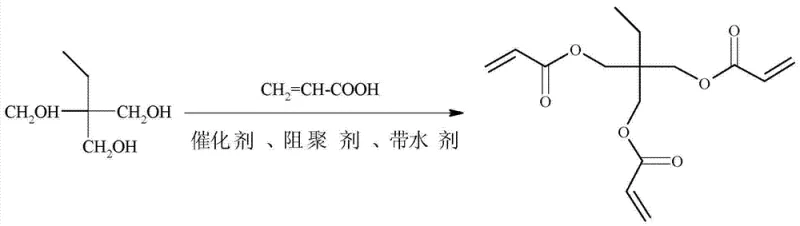
The core of this technological breakthrough lies in the manipulation of the esterification equilibrium through efficient water removal and selective catalysis. In the reaction vessel, trimethylolpropane reacts with three equivalents of acrylic acid in a reversible condensation reaction. The addition of the alkane water carrier forms a low-boiling azeotrope with the water produced, allowing it to be continuously distilled off at temperatures significantly lower than the boiling point of acrylic acid itself. This continuous removal of the byproduct drives the equilibrium towards the formation of the tri-ester, pushing the reaction to completion without the need for a vast excess of reagents. The use of molecular sieve catalysts provides a high surface area with specific acidic sites that activate the carboxylic acid group of the acrylic acid, facilitating nucleophilic attack by the hydroxyl groups of the TMP while minimizing side reactions such as etherification or Michael addition.
Controlling the impurity profile is equally critical, particularly the suppression of acrylic acid polymerization which can occur readily at elevated temperatures. The patent specifies the use of phenolic or amine-based inhibitors like thiodiphenylamine, which act as radical scavengers to terminate any propagating polymer chains before they can grow into insoluble gels. This mechanism ensures that the final product remains a clear, low-viscosity liquid suitable for precise application in UV curing formulations. Furthermore, the mild reaction conditions (80-100°C) prevent the thermal decomposition of the inhibitor and the monomer, preserving the color and stability of the final product. The subsequent purification steps, involving alkaline washing to remove unreacted acid and activated carbon treatment to remove colored impurities, leverage the chemical stability imparted by this gentle synthesis route to achieve the reported >98% purity levels essential for demanding electronic and coating applications.
How to Synthesize Trimethylolpropane Triacrylate Efficiently
The synthesis protocol outlined in the patent provides a reproducible framework for manufacturing TMPTA that balances reaction kinetics with safety and environmental considerations. The process begins with the precise charging of trimethylolpropane, acrylic acid, the selected alkane solvent, catalyst, and inhibitor into a reactor equipped with a water separator. The mixture is then heated to reflux, maintaining a temperature window of 80-100°C, until the theoretical amount of water has been collected, indicating the completion of the esterification. Following the reaction, the solvent is removed under reduced pressure, and the crude product undergoes a series of washes and treatments to isolate the pure triacrylate. For a comprehensive, step-by-step technical guide including specific molar ratios and equipment specifications, please refer to the standardized synthesis instructions below.
- Mix trimethylolpropane, acrylic acid, alkane water carrier (e.g., cyclohexane), catalyst (e.g., PEP-15u molecular sieve), and polymerization inhibitor in a reactor.
- Heat the mixture to 80-100°C under atmospheric pressure and reflux until no more water is separated in the water trap.
- Remove the water carrier under reduced pressure, wash the organic phase with alkali and water, decolorize with activated carbon, and dry to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement strategists and supply chain directors, the adoption of this green esterification technology offers profound advantages that extend beyond mere technical specifications. By eliminating the need for hazardous benzene and corrosive acid chlorides, manufacturers can drastically simplify their safety protocols and reduce the capital expenditure associated with specialized corrosion-resistant equipment and toxic waste treatment facilities. The shift to readily available alkanes and solid catalysts creates a more resilient supply chain, less susceptible to the regulatory volatility often associated with aromatic solvents. Moreover, the lower operating temperatures translate directly into reduced energy consumption per batch, contributing to a lower overall cost of goods sold (COGS) and a smaller carbon footprint, which is increasingly a prerequisite for doing business with major multinational corporations focused on sustainability goals.
- Cost Reduction in Manufacturing: The elimination of expensive acid chloride reagents and the associated scrubbing systems for hydrogen chloride gas removal results in significant operational savings. Additionally, the use of recoverable alkane solvents and heterogeneous catalysts reduces raw material consumption and waste disposal costs, allowing for a more lean and cost-effective production model that enhances margin potential without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sourcing C6-C7 alkanes and molecular sieve catalysts is generally more stable and less regulated than procuring benzene or acryloyl chloride, which are often subject to strict transport and storage restrictions. This accessibility ensures a smoother flow of raw materials, minimizing the risk of production stoppages due to supply shortages or regulatory compliance issues, thereby guaranteeing consistent delivery schedules for downstream customers.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, operating at atmospheric pressure with moderate thermal requirements that are easily managed in large-scale reactors. The absence of toxic byproducts and the use of environmentally benign solvents facilitate easier permitting and compliance with increasingly stringent global environmental regulations, future-proofing the manufacturing asset against evolving legislative landscapes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-purity TMPTA synthesis method. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the technology's capabilities and limitations for potential partners and technical evaluators.
Q: What are the advantages of using alkanes instead of benzene in TMPTA synthesis?
A: Using C6-C7 alkanes like cyclohexane or n-hexane replaces toxic benzene and toluene, significantly reducing environmental hazards and operator health risks while maintaining effective water removal during esterification.
Q: How does the novel catalyst system improve product purity?
A: The use of solid acid catalysts like PEP-15u molecular sieves or specific metal salts allows for milder reaction conditions (80-100°C) compared to traditional methods, minimizing side reactions and thermal degradation, resulting in purity exceeding 98%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process operates at atmospheric pressure with moderate temperatures and avoids corrosive byproducts like hydrogen chloride gas, making it highly scalable, energy-efficient, and compliant with green chemistry standards for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trimethylolpropane Triacrylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of consistent quality and supply continuity for high-value monomers like Trimethylolpropane Triacrylate. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch meets the exacting standards required for photo-cured coatings and advanced polymer applications, giving our partners confidence in the reliability of our supply.
We invite you to engage with our technical procurement team to discuss how this innovative green synthesis route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this cleaner process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that demonstrate our commitment to delivering superior chemical solutions.
