Advanced Synthesis of Ethyl 1-oxo-4-azaspiro[5.5]undecane-9-carboxylate for Commercial Scale-up
Advanced Synthesis of Ethyl 1-oxo-4-azaspiro[5.5]undecane-9-carboxylate for Commercial Scale-up
The pharmaceutical industry continuously demands more efficient routes for complex heterocyclic scaffolds, particularly spiro-piperidine derivatives which serve as critical building blocks for various therapeutic agents. Patent CN115785019A introduces a groundbreaking methodology for the synthesis of ethyl-1-oxo-4-azaspiro[5.5]undecane-9-carboxylate, addressing significant bottlenecks found in legacy production methods. This technical disclosure outlines a streamlined pathway that replaces inefficient multi-step sequences with a high-yielding reductive amination and cyclization strategy. For R&D directors and procurement specialists, this innovation represents a pivotal shift towards cost-effective and scalable manufacturing of high-purity pharmaceutical intermediates. By leveraging readily available starting materials and robust reaction conditions, this process ensures supply chain stability while drastically reducing waste generation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of related spiro-morpholine structures relied on convoluted pathways that suffered from severe efficiency losses. As documented in earlier literature such as CN202010428362.6, the traditional approach involved constructing the spiro-center through a series of functional group transformations that included chlorination and iodination steps before a final cyclization. 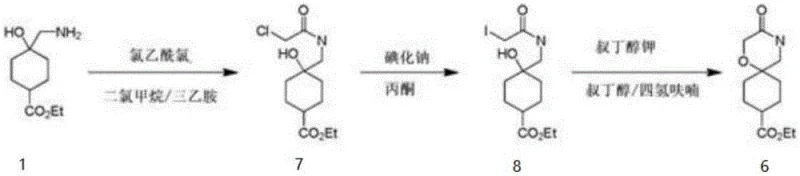 This legacy route resulted in a cyclization yield of merely 17%, creating a massive material bottleneck. Furthermore, attempts to convert the resulting ketone intermediate into the desired amine via reduction were plagued by reproducibility issues, with yields plummeting to between 1% and 5%. Such poor performance renders these conventional methods economically unviable for commercial scale-up, as the accumulation of losses at each step drives up the cost of goods sold exponentially.
This legacy route resulted in a cyclization yield of merely 17%, creating a massive material bottleneck. Furthermore, attempts to convert the resulting ketone intermediate into the desired amine via reduction were plagued by reproducibility issues, with yields plummeting to between 1% and 5%. Such poor performance renders these conventional methods economically unviable for commercial scale-up, as the accumulation of losses at each step drives up the cost of goods sold exponentially.
The Novel Approach
The patented methodology revolutionizes this landscape by introducing a direct and convergent synthetic strategy that bypasses the problematic oxidative steps entirely. Instead of building the ring system through halogenated intermediates, the new process utilizes a reductive amination between a cyclohexane derivative and glycolaldehyde dimer to install the necessary carbon-nitrogen framework in a single operation. This is immediately followed by a highly efficient intramolecular cyclization mediated by p-toluenesulfonyl chloride and potassium tert-butoxide. 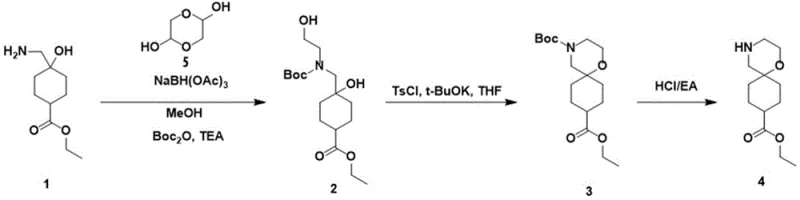 This strategic redesign elevates the cyclization yield to over 74% and the final deprotection step to nearly 99%, representing a quantum leap in process efficiency. For a reliable pharmaceutical intermediate supplier, adopting this route means transforming a marginal laboratory curiosity into a robust industrial process capable of meeting global demand without the burden of excessive raw material consumption.
This strategic redesign elevates the cyclization yield to over 74% and the final deprotection step to nearly 99%, representing a quantum leap in process efficiency. For a reliable pharmaceutical intermediate supplier, adopting this route means transforming a marginal laboratory curiosity into a robust industrial process capable of meeting global demand without the burden of excessive raw material consumption.
Mechanistic Insights into Reductive Amination and Base-Mediated Cyclization
The core of this technological advancement lies in the precise control of chemoselectivity during the formation of the morpholine ring. The initial step involves the condensation of the primary amine on the cyclohexane scaffold with glycolaldehyde dimer, facilitated by a mild reducing agent such as sodium triacetoxyborohydride. This reductive amination is crucial as it installs the hydroxyethyl side chain necessary for the subsequent ring closure while simultaneously protecting the nitrogen with a Boc group to prevent polymerization. The choice of solvent, typically methanol or dichloromethane, and the stoichiometric balance of the reducing agent are critical parameters that ensure the reaction proceeds to completion without over-reduction or side reactions. This careful orchestration results in the stable linear precursor, compound 2, which serves as the perfect substrate for the cyclization event.
The cyclization mechanism itself is a masterpiece of base-mediated nucleophilic substitution. Upon treatment with potassium tert-butoxide in tetrahydrofuran, the hydroxyl group of the side chain is activated, likely forming an alkoxide species that attacks the sulfonate ester generated in situ from the reaction with p-toluenesulfonyl chloride. This intramolecular SN2 reaction closes the six-membered morpholine ring with high stereoelectronic favorability due to the pre-organization of the chain. The use of mild heating at 50°C accelerates this kinetics without compromising the integrity of the sensitive ester functionality on the cyclohexane ring. This mechanistic clarity allows for tight impurity control, as the reaction conditions are specific enough to avoid epimerization or hydrolysis, ensuring that the final spiro-piperidine product maintains the rigorous purity specifications required for downstream API synthesis.
How to Synthesize Ethyl 1-oxo-4-azaspiro[5.5]undecane-9-carboxylate Efficiently
Implementing this synthesis requires strict adherence to the optimized conditions detailed in the patent to maximize yield and safety. The process begins with the preparation of the Boc-protected amino-alcohol intermediate, followed by the critical ring-closing step and final acid deprotection. Each stage has been fine-tuned to balance reaction rate with product quality, ensuring that the process is not only chemically sound but also operationally simple for plant personnel. The following guide summarizes the critical operational parameters derived from the experimental examples, providing a roadmap for technical teams to replicate this success in their own facilities. Detailed standardized synthesis steps are provided in the section below.
- Perform reductive amination of the starting amine with glycolaldehyde dimer using sodium triacetoxyborohydride, followed by in-situ Boc protection to form the linear precursor.
- Execute intramolecular cyclization using p-toluenesulfonyl chloride and potassium tert-butoxide in tetrahydrofuran at 50°C to close the morpholine ring.
- Remove the Boc protecting group under acidic conditions using hydrochloric acid in ethyl acetate to yield the final spiro-piperidine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers profound strategic benefits that extend far beyond simple chemistry. The primary advantage lies in the drastic simplification of the manufacturing workflow, which directly translates to reduced operational expenditures and enhanced supply security. By eliminating the need for hazardous halogenating agents and low-yielding reduction steps, the process minimizes the handling of dangerous chemicals and reduces the volume of waste streams requiring disposal. This alignment with green chemistry principles not only lowers environmental compliance costs but also future-proofs the supply chain against increasingly stringent regulatory pressures regarding solvent usage and waste generation in fine chemical manufacturing.
- Cost Reduction in Manufacturing: The economic impact of improving the cyclization yield from 17% to over 74% cannot be overstated, as it effectively quadruples the output from the same amount of starting materials. This substantial increase in material throughput significantly lowers the unit cost of the intermediate, allowing for more competitive pricing in the final API market. Furthermore, the elimination of expensive transition metal catalysts and the use of commodity reagents like p-toluenesulfonyl chloride and potassium tert-butoxide ensure that raw material costs remain stable and predictable. The simplified workup procedures, which rely on standard extractions and filtrations rather than complex chromatographic separations, further reduce processing time and labor costs, driving down the overall cost of goods sold.
- Enhanced Supply Chain Reliability: The reliance on readily available and stable starting materials, such as glycolaldehyde dimer and common cyclohexane derivatives, mitigates the risk of supply disruptions often associated with specialized or custom-synthesized reagents. The robustness of the reaction conditions, which tolerate ambient temperatures and standard atmospheric pressure, means that production can be easily scaled across multiple manufacturing sites without the need for specialized cryogenic or high-pressure equipment. This flexibility ensures continuous supply continuity even in the face of regional logistical challenges, providing pharmaceutical partners with a dependable source of critical intermediates that supports their own production schedules without delay.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing solvents like methanol and ethyl acetate that are easily recovered and recycled in industrial distillation units. The absence of heavy metal contaminants simplifies the purification process, reducing the burden on wastewater treatment facilities and ensuring that the final product meets strict residual solvent and heavy metal guidelines. This environmental compatibility facilitates faster regulatory approvals and reduces the carbon footprint of the manufacturing process, aligning with the sustainability goals of modern pharmaceutical companies. The ability to produce high-purity material with minimal environmental impact positions this synthesis route as a preferred choice for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and process descriptions within the patent, offering clarity on how this method compares to existing alternatives. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their specific pipeline requirements. The answers provided reflect the proven capabilities of the method as demonstrated in the disclosed examples.
Q: How does this new synthesis method improve yield compared to prior art?
A: The novel route bypasses the low-yielding oxidation-reduction sequence of previous methods, achieving a cyclization yield of over 74% compared to the historical 17%, significantly boosting overall material throughput.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes common solvents like methanol and THF, avoids cryogenic conditions, and features simple aqueous workups, making it highly robust for multi-kilogram to ton-scale manufacturing.
Q: What is the purity profile of the final intermediate?
A: The final deprotection step yields the product as an HCl salt with demonstrated purity exceeding 99.0%, minimizing the need for extensive downstream purification before use in API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethyl 1-oxo-4-azaspiro[5.5]undecane-9-carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful translation of a patent concept into commercial reality requires more than just chemical knowledge; it demands engineering excellence and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields observed in the laboratory are faithfully reproduced on the plant floor. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of ethyl-1-oxo-4-azaspiro[5.5]undecane-9-carboxylate meets the exacting standards required for pharmaceutical applications. Our dedication to process optimization means we continuously refine our manufacturing protocols to enhance efficiency and sustainability.
We invite you to collaborate with us to leverage this advanced synthesis technology for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this superior route for your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in complex spiro-cycle manufacturing can accelerate your timeline to market while optimizing your budget.
