Advanced One-Pot Synthesis of Multi-Substituted Acenes for High-Performance Organic Electronics
Advanced One-Pot Synthesis of Multi-Substituted Acenes for High-Performance Organic Electronics
The rapid evolution of the organic electronics sector demands materials that combine exceptional charge carrier mobility with thermal stability and processability. Patent CN101279890A introduces a groundbreaking methodology for the preparation of multi-substituted acene derivatives, a class of compounds critical for next-generation organic electroluminescent devices, organic field-effect transistors (OFETs), and organic photovoltaics. Unlike conventional multi-step syntheses that rely on costly transition metals and unstable intermediates, this invention leverages a streamlined one-pot reductive coupling strategy. By utilizing accessible 1,2-disubstituted acetylene derivatives and polyhalogenated benzenes in the presence of alkali metal reducing agents, the process achieves high structural diversity under mild conditions. For R&D directors and procurement specialists seeking a reliable OLED material supplier, this technology represents a pivotal shift towards more sustainable and economically viable manufacturing of high-purity organic semiconductors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted acenes has been plagued by significant operational hurdles that hinder commercial scalability. Traditional routes often involve the cycloaddition of aryne derivatives with furan derivatives followed by reduction, a sequence that is inherently step-intensive and generates substantial chemical waste. Alternatively, methods employing copper-catalyzed coupling of cyclopentadienyl zirconocene derivatives with polyhalobenzenes have been reported, but these suffer from severe economic and practical drawbacks. The prerequisite use of zirconocene dichloride is not only expensive but also necessitates the in-situ generation of unstable cyclopentadienyl zirconocene intermediates, which complicates reactor management and increases safety risks. Furthermore, these legacy processes often lack the flexibility to easily tune the substitution pattern, limiting the ability to optimize the electronic properties of the final material for specific device architectures.
The Novel Approach
The methodology disclosed in CN101279890A circumvents these bottlenecks through a direct, one-pot reductive coupling mechanism. By reacting 1,2-disubstituted acetylene derivatives with a reducing agent—such as lithium, sodium, potassium, or specific organometallic intermediates like lithium naphthalenide—followed by the addition of a polyhalogenated benzene derivative, the target acene skeleton is constructed efficiently. This approach operates effectively at temperatures ranging from -78°C to 50°C, with a preferred window of -20°C to 30°C, eliminating the need for extreme cryogenic conditions often associated with organolithium chemistry. The simplicity of mixing reagents in common ether solvents like tetrahydrofuran (THF) drastically reduces processing time and equipment complexity. This innovation directly addresses the need for cost reduction in electronic chemical manufacturing by minimizing unit operations and maximizing atom economy relative to older transition-metal catalyzed routes.
Mechanistic Insights into Reductive Coupling Cyclization
The core of this synthetic breakthrough lies in the generation of highly reactive organometallic species that facilitate the construction of the fused aromatic ring system. The process initiates with the reduction of the 1,2-disubstituted acetylene by the alkali metal or organometallic reducing agent. This step likely involves a single-electron transfer (SET) mechanism, generating radical anion intermediates that are poised for nucleophilic attack. The choice of reducing agent is critical; the patent highlights the efficacy of lithium naphthalenide or lithium biphenylide solutions, which offer a controlled reduction potential compared to bulk metal. These intermediates subsequently react with the polyhalogenated benzene derivative, where the halogen atoms (preferably fluorine) act as leaving groups in a nucleophilic aromatic substitution or elimination-addition sequence. This cascade reaction effectively stitches the acetylene units onto the benzene core, forming the extended pi-conjugated acene structure in a single vessel.
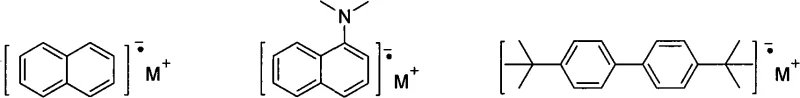
Understanding the impurity profile is essential for R&D teams focusing on device performance. The one-pot nature of the reaction minimizes the exposure of reactive intermediates to atmospheric moisture and oxygen, which are common sources of oxidation byproducts in acene chemistry. Furthermore, the ability to control the stoichiometry between the acetylene and the polyhalobenzene allows for the selective formation of either symmetric or asymmetric products. For instance, maintaining a specific molar ratio ensures that the substitution pattern remains uniform, thereby reducing the formation of regioisomers that are difficult to separate and can act as trap states in organic thin-film transistors. This level of control over the杂质谱 (impurity profile) is vital for achieving the high purity specifications required in commercial OLED production.
How to Synthesize Multi-Substituted Acene Derivatives Efficiently
Implementing this synthesis route requires strict adherence to anhydrous and oxygen-free conditions to preserve the activity of the reducing agents and prevent side reactions. The general protocol involves preparing the reducing agent solution first, followed by the sequential addition of the alkyne and the halogenated aromatic substrate. The reaction progress is monitored to ensure complete consumption of the starting materials before quenching with a mild proton source such as saturated ammonium chloride. Detailed standard operating procedures regarding specific molar ratios, solvent volumes, and workup techniques are critical for reproducibility. For a comprehensive guide on executing this transformation with optimal yield and purity, please refer to the standardized synthesis steps outlined below.
- Prepare the reducing agent by reacting lithium metal with naphthalene or a biphenyl derivative in an anhydrous ether solvent like THF under inert atmosphere.
- Add the 1,2-disubstituted acetylene derivative to the reducing agent solution and stir to form the organometallic intermediate.
- Introduce the polyhalogenated benzene derivative to the mixture, allow the coupling reaction to proceed, and finally quench with aqueous ammonium chloride followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the transition to this one-pot methodology offers profound logistical and economic benefits. The elimination of expensive transition metal catalysts like zirconium complexes removes a significant cost driver from the bill of materials. Moreover, the reliance on commodity chemicals such as lithium metal, naphthalene, and substituted acetylenes ensures a robust and continuous supply chain, mitigating the risk of raw material shortages that often plague specialty chemical manufacturing. The simplified workflow reduces the demand for specialized reactor configurations, allowing for production in standard glass-lined or stainless steel vessels equipped with basic cooling capabilities. This adaptability facilitates easier technology transfer from laboratory scale to pilot and commercial plants, ensuring consistent supply continuity for downstream device manufacturers.
- Cost Reduction in Manufacturing: The primary economic advantage stems from the drastic simplification of the process flow. By consolidating multiple reaction steps into a single pot, the requirement for intermediate isolation, drying, and purification is removed. This consolidation leads to substantial savings in solvent consumption, energy usage for heating and cooling cycles, and labor hours. Additionally, the avoidance of precious or rare earth metal catalysts eliminates the need for costly metal scavenging steps to meet residual metal specifications, further driving down the overall cost of goods sold (COGS) for high-value electronic materials.
- Enhanced Supply Chain Reliability: The raw materials utilized in this process, including various substituted acetylenes and polyfluorinated benzenes, are widely available from global chemical suppliers. Unlike proprietary catalysts that may be sourced from a single vendor, these commoditized inputs offer procurement managers greater negotiating power and supply security. The robustness of the reaction conditions, which tolerate a range of temperatures and do not require ultra-high vacuum or exotic atmospheres beyond standard inert gas blanketing, further enhances operational reliability. This resilience ensures that production schedules can be maintained even under fluctuating market conditions, securing the supply of critical OLED materials.
- Scalability and Environmental Compliance: Scaling this chemistry is straightforward due to the homogeneous nature of the reaction mixture and the absence of heterogeneous catalysts that can cause fouling or filtration issues on a large scale. The use of ether solvents like THF, which are well-understood in industrial settings, allows for efficient recovery and recycling, aligning with green chemistry principles. The reduction in waste generation, particularly the absence of heavy metal waste streams, simplifies environmental compliance and waste disposal protocols. This eco-friendly profile not only reduces regulatory burdens but also aligns with the sustainability goals of major electronics corporations seeking greener supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and scope of the method. Understanding these details is crucial for technical teams evaluating the integration of these materials into their product pipelines.
Q: What are the primary advantages of this one-pot method over traditional zirconocene-mediated synthesis?
A: The traditional method often requires expensive zirconocene dichloride and involves multiple steps to generate unstable intermediates. This novel one-pot approach utilizes readily available lithium-based reducing agents, operates under milder conditions (-20 to 30°C), and eliminates the need for isolating sensitive intermediates, significantly simplifying the operational complexity.
Q: Can the symmetry of the final acene product be controlled during synthesis?
A: Yes, the patent explicitly demonstrates that product symmetry is tunable by adjusting the molar ratio of reactants. A ratio of polyhalobenzene to acetylene greater than or equal to 1:2 favors asymmetric products, while a ratio less than or equal to 1:4 predominantly yields symmetric structures, offering flexibility for specific electronic property tuning.
Q: What types of substituents are compatible with this reductive coupling methodology?
A: The process is highly versatile, accommodating a wide range of substituents including halogens (specifically fluorine), alkyl groups (C1-C22), aryl groups (phenyl, naphthyl, etc.), and trialkylsilyl groups. This allows for the precise engineering of HOMO-LUMO levels and solid-state packing for OLED and OFET applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Multi-Substituted Acene Supplier
As the demand for high-performance organic semiconductors continues to surge, partnering with an experienced CDMO is essential for navigating the complexities of scale-up. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of analyzing trace impurities that could affect device performance. We understand that the consistency of electronic materials is paramount, and our quality management systems are designed to deliver batch-to-batch reproducibility that meets the exacting standards of the display and photovoltaic industries.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific application needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this one-pot method can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data for our acene derivatives and to discuss route feasibility assessments for your custom projects. Together, we can accelerate the development of next-generation organic electronic devices.
