Advanced N,N-Coordinated Palladium Carborane Catalyst for Efficient Arylamine Formylation in Pharma Manufacturing
Advanced N,N-Coordinated Palladium Carborane Catalyst for Efficient Arylamine Formylation in Pharma Manufacturing
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more robust, selective, and environmentally benign catalytic systems. A significant breakthrough in this domain is documented in Chinese Patent CN110372755B, which discloses a novel N,N-coordinated palladium complex featuring a meta-carborane ligand. This innovation addresses critical pain points in the production of arylamine formamides, which are pivotal intermediates in the synthesis of pharmaceuticals, agrochemicals, and functional materials. Unlike traditional catalysts that often suffer from thermal degradation or limited substrate tolerance, this new complex leverages the unique icosahedral geometry of the carborane cage to impart exceptional stability and catalytic activity. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, understanding the mechanistic advantages of this technology is essential for optimizing supply chains and reducing manufacturing costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the formylation of amines has been achieved using a variety of reagents such as formic acid, chloral, ammonium formate, or various formate esters. While these methods are established, they are fraught with significant operational and safety drawbacks that hinder large-scale cost reduction in fine chemical manufacturing. Many conventional protocols require harsh reaction conditions, including high temperatures or strongly acidic environments, which can lead to the decomposition of sensitive functional groups on the substrate. Furthermore, reagents like chloral are highly toxic and pose severe environmental and health risks, necessitating expensive waste treatment protocols. The use of multiple reagents often complicates the downstream purification process, leading to lower overall yields and increased generation of hazardous waste residues. Additionally, traditional catalysts may lack the necessary thermal stability, decomposing under reaction conditions and requiring frequent replenishment, which disrupts production continuity and inflates operational expenditures.
The Novel Approach
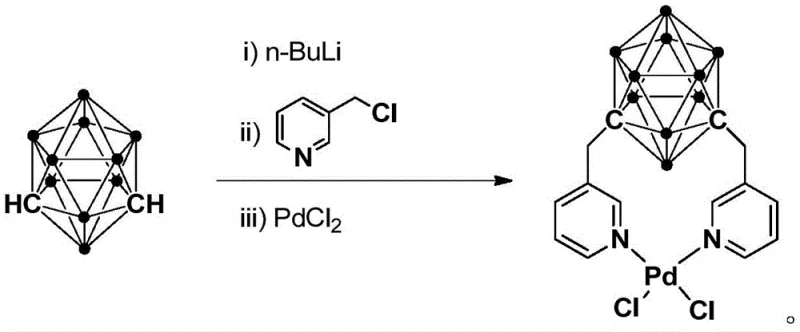
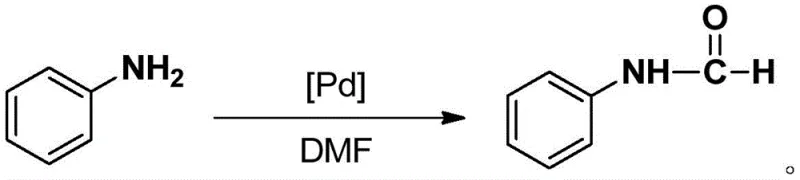
The technology outlined in patent CN110372755B presents a paradigm shift by introducing a highly stable palladium catalyst that operates under mild, green conditions. The core of this innovation lies in the synthesis of an N,N-coordinated divalent palladium complex where the ligand is derived from meta-carborane. This specific architecture allows the catalyst to remain active and structurally intact even at elevated temperatures up to 300°C. The synthesis of the catalyst itself is streamlined, involving a one-pot reaction sequence starting from meta-carborane, n-butyllithium, 3-chloromethylpyridine, and palladium chloride. This straightforward preparation minimizes unit operations and solvent usage. In application, the catalyst enables the use of N,N-dimethylformamide (DMF) as a dual-purpose solvent and formylating agent. This eliminates the need for external, toxic formylating reagents, drastically simplifying the reaction setup and workup. The result is a process that delivers high-purity arylamine formamides with excellent selectivity and yields ranging from 82% to 95%, representing a substantial improvement over legacy methods.
Mechanistic Insights into N,N-Coordinated Palladium Carborane Catalysis
The superior performance of this catalyst can be attributed to the unique electronic and steric properties of the meta-carborane ligand. The carborane cage is an electron-withdrawing, three-dimensional aromatic system that stabilizes the metal center against oxidation and aggregation. When coordinated to the palladium center via the nitrogen atoms of the pyridine rings attached to the carborane framework, it forms a rigid chelate structure. This N,N-coordination mode prevents the dissociation of the ligand under thermal stress, which is a common failure mode for many homogeneous catalysts. The robustness of the carbon-boron cluster ensures that the catalyst maintains its integrity throughout the catalytic cycle, allowing for consistent turnover numbers. Furthermore, the steric bulk provided by the carborane cage likely creates a specific microenvironment around the active site that favors the approach of the amine substrate while inhibiting side reactions, thereby enhancing selectivity. This mechanistic stability is crucial for commercial scale-up of complex palladium catalysts, as it ensures batch-to-batch reproducibility and reduces the risk of catalyst deactivation during prolonged reaction times.
In terms of impurity control, the use of DMF as both solvent and reagent significantly reduces the complexity of the impurity profile. Since no external formylating agents are added, there are fewer opportunities for the formation of byproducts derived from reagent decomposition or side reactions with impurities in the reagent stream. The high thermal stability of the catalyst also means that it does not degrade into palladium black or other inactive species that could contaminate the final product with heavy metal residues. This is particularly important for pharmaceutical applications where strict limits on residual metals are enforced. The reaction proceeds smoothly at temperatures between 60°C and 100°C, which is mild enough to preserve sensitive functional groups on the aromatic ring, such as methoxy or nitro groups, as demonstrated in the patent examples. This broad functional group tolerance expands the utility of the process for synthesizing diverse drug intermediates without the need for extensive protecting group strategies.
How to Synthesize N,N-Coordinated Palladium Carborane Complex Efficiently
The preparation of this advanced catalyst is designed for operational simplicity, utilizing readily available starting materials and standard laboratory techniques. The process begins with the lithiation of meta-carborane, followed by alkylation with 3-chloromethylpyridine to generate the bidentate ligand in situ, which is then immediately complexed with palladium chloride. This telescoped approach minimizes isolation steps and exposure to air-sensitive intermediates. The detailed standardized synthesis steps, including precise molar ratios, temperature controls, and purification parameters, are critical for achieving the reported high yields and stability. For process chemists looking to implement this technology, adhering to the specific stoichiometry and reaction times outlined in the patent is essential to maximize the quality of the final catalyst.
- Lithiation of meta-carborane: Add n-BuLi solution to a meta-carborane tetrahydrofuran solution at low temperature (-80°C to -75°C), then warm to room temperature and react for 30-60 minutes.
- Ligand Formation: Introduce 3-chloromethyl pyridine to the reaction mixture and stir at room temperature for 3-5 hours to form the functionalized ligand.
- Complexation: Add PdCl2 to the system and react at room temperature for 2-5 hours, followed by filtration and column chromatography purification to isolate the final palladium complex.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the adoption of this meta-carborane palladium catalyst offers compelling economic and logistical benefits. The primary driver for cost reduction in pharmaceutical intermediate manufacturing is the simplification of the raw material bill of materials. By utilizing DMF, a commodity chemical, as the sole source of the formyl group, the process eliminates the need for purchasing and storing specialized, often expensive, formylating agents. This consolidation of reagents not only lowers direct material costs but also reduces the inventory footprint and associated handling risks. Furthermore, the high catalytic efficiency and stability mean that lower catalyst loadings can potentially be used to achieve the same conversion, further driving down the cost per kilogram of the final product. The robustness of the catalyst also implies a longer shelf life, reducing waste due to expiration and allowing for larger, less frequent procurement batches which optimizes cash flow.
- Cost Reduction in Manufacturing: The elimination of toxic and expensive formylating reagents in favor of DMF significantly lowers the variable cost of production. The high yields reported (82-95%) minimize the loss of valuable amine substrates, ensuring that raw material investment is maximized in the final product. Additionally, the simplified workup procedure, which avoids complex neutralization or extraction steps associated with acidic formylation methods, reduces labor hours and utility consumption. The ability to operate at moderate temperatures (60-100°C) also translates to lower energy costs compared to processes requiring reflux or high-pressure conditions. Overall, the process economics are favorable due to the convergence of high efficiency, low-cost reagents, and reduced waste disposal fees.
- Enhanced Supply Chain Reliability: The insensitivity of the catalyst to air and water is a critical factor for supply chain resilience. Unlike many organometallic catalysts that require inert atmosphere handling and specialized shipping containers, this complex can be handled with greater ease, reducing the risk of spoilage during transit and storage. The use of common solvents like THF, petroleum ether, and ethyl acetate for synthesis and purification ensures that supply disruptions for exotic chemicals are unlikely to impact production schedules. The broad substrate scope means that a single catalyst inventory can support the production of multiple different formamide intermediates, allowing for greater flexibility in production planning and reducing the need for multiple specialized catalysts. This versatility strengthens the supply chain against demand fluctuations for specific end-products.
- Scalability and Environmental Compliance: The green chemistry attributes of this process align perfectly with modern environmental regulations and corporate sustainability goals. The reduction in hazardous waste generation, owing to the absence of toxic reagents and high atom economy, simplifies compliance with environmental discharge standards. The thermal stability of the catalyst facilitates safe scale-up from gram to tonne quantities, as the risk of runaway exotherms or catalyst decomposition is minimized. The straightforward purification via column chromatography or crystallization (depending on scale optimization) ensures that the final product meets stringent purity specifications required for regulatory filings. This ease of scale-up accelerates the time-to-market for new drug candidates relying on these intermediates, providing a competitive advantage in the fast-paced pharmaceutical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-carborane technology. These insights are derived directly from the experimental data and claims presented in patent CN110372755B, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this catalyst into their existing manufacturing workflows.
Q: What are the thermal stability characteristics of this palladium catalyst?
A: According to patent CN110372755B, the N,N-coordinated palladium complex exhibits exceptional thermal stability, remaining stable at temperatures up to 300°C, which ensures robustness during storage and handling.
Q: Can this catalyst be used for substrates with electron-withdrawing groups?
A: Yes, the catalyst demonstrates broad substrate scope, successfully catalyzing the formylation of 4-nitroaniline (containing a strong electron-withdrawing nitro group) with high efficiency and yield.
Q: What solvent system is utilized for the formylation reaction?
A: The process utilizes N,N-dimethylformamide (DMF) which serves a dual role as both the reaction solvent and the formylation reagent, simplifying the workup procedure and reducing waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Palladium Carborane Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the N,N-coordinated palladium carborane complex in driving efficiency and innovation in chemical synthesis. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such patented methodologies into robust, commercial-scale processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We maintain stringent purity specifications across all our outputs, supported by state-of-the-art rigorous QC labs equipped to analyze complex organometallic species and ensure the absence of detrimental impurities. Whether you require the catalyst itself or the downstream arylamine formamide intermediates, our commitment to quality and consistency makes us the ideal partner for your long-term supply needs.
We invite you to explore how this technology can optimize your specific production lines. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your current manufacturing bottlenecks. We encourage you to contact our technical procurement team to request specific COA data for related intermediates or to discuss route feasibility assessments for your target molecules. By leveraging our deep understanding of catalytic chemistry and supply chain dynamics, we can help you secure a competitive edge in the global market for high-value pharmaceutical intermediates.
