Advanced Chiral Synthesis of Nebivolol Hydrochloride for Commercial Scale-Up
Advanced Chiral Synthesis of Nebivolol Hydrochloride for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for complex cardiovascular agents, particularly those possessing multiple chiral centers like Nebivolol. Patent CN102344431B discloses a highly efficient method for preparing Nebivolol Hydrochloride, addressing critical bottlenecks in stereochemical control and raw material costs. This technology enables the production of the active SRRR and RSSS isomers through a convergent strategy that leverages cheap, achiral starting materials alongside a single chiral source. By optimizing reaction conditions and eliminating hazardous reagents, this process represents a significant advancement for any reliable nebivolol intermediate supplier aiming to secure supply chains for beta-blocker manufacturing. The structural complexity of Nebivolol, featuring four chiral carbon atoms and ten possible isomers, demands precise synthetic control to ensure therapeutic efficacy.
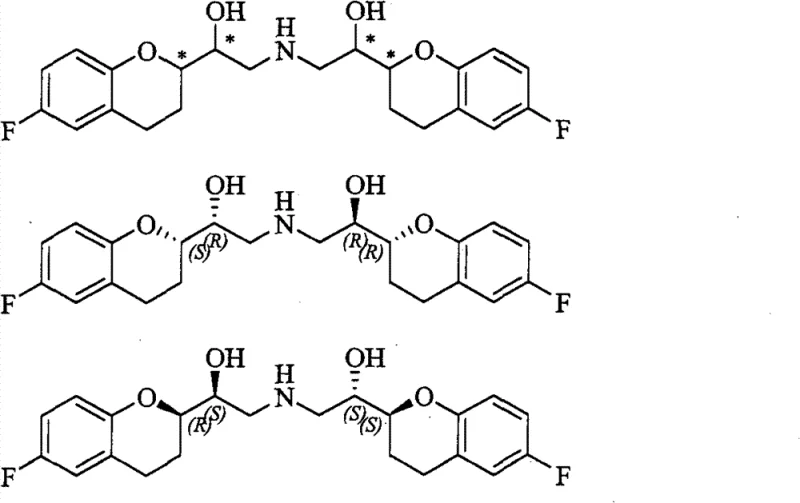
Nebivolol functions as a third-generation beta-blocker with unique nitric oxide-mediated vasodilatory properties, making its high-purity synthesis paramount for patient safety. The disclosed invention achieves the target molecule through a sequence involving bromination, Wittig olefination, reduction, hydrolysis, condensation, chiral inversion, ring-opening addition, and hydrogenation. Unlike traditional routes that struggle with racemization or require prohibitively expensive chiral auxiliaries, this method utilizes D-glyceraldehyde acetonide to introduce chirality early in the synthesis. This strategic choice not only lowers the entry barrier for production but also enhances the overall stereochemical integrity of the final API, ensuring that the resulting hydrochloride salt meets stringent pharmacopeial standards for enantiomeric purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing Nebivolol have historically been plagued by excessive step counts, harsh reaction conditions, and reliance on unstable chiral building blocks. For instance, earlier patents such as CN1834093A and CN1978442A describe routes that, while conceptually sound, often result in partial racemization of the glycerol chiral center during key transformation steps. As illustrated in existing literature, attempts to replicate these methods frequently yield mixtures of diastereomers rather than the desired optically pure intermediates, necessitating difficult and costly separation processes. Furthermore, other documented approaches, such as those disclosed in WO2004/041805, require up to 30 synthetic steps to reach the racemic mixture, involving extreme temperatures ranging from 0°C to 145°C.
These conventional pathways often depend on L-glyceraldehyde acetonide, a reagent known for its poor stability and high market price, which drastically inflates the cost of goods sold (COGS). The necessity for multiple column chromatography purifications in these older routes further hinders their applicability to large-scale manufacturing, as chromatography is notoriously difficult to scale and generates significant solvent waste. Additionally, the use of toxic solvents and reagents in some legacy processes poses environmental compliance challenges, making them less attractive for modern green chemistry initiatives. The cumulative effect of these limitations is a supply chain that is fragile, expensive, and prone to delays, failing to meet the demands of a cost reduction in pharmaceutical intermediates manufacturing strategy.
The Novel Approach
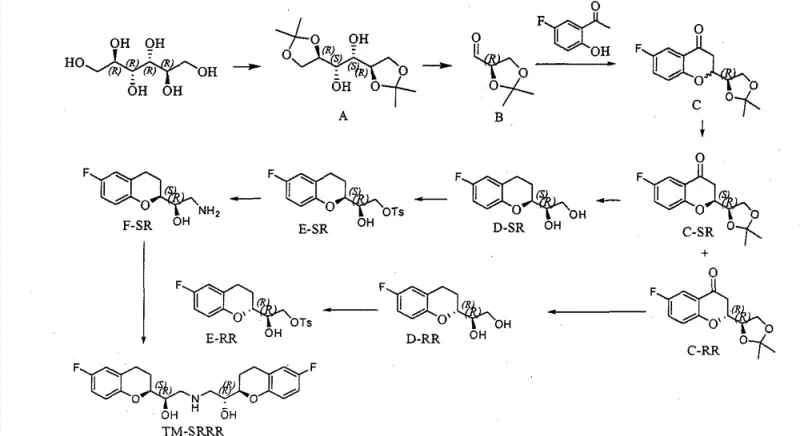
The innovative process described in CN102344431B overcomes these hurdles by streamlining the synthesis into approximately 17 to 21 steps, utilizing mild reaction conditions that are inherently safer and easier to control. A pivotal improvement is the substitution of expensive L-glyceraldehyde acetonide with the commercially available and stable D-glyceraldehyde acetonide. This switch allows for the introduction of chirality at an early stage without compromising the stereochemical outcome of subsequent reactions. The route begins with the bromination of 5-fluoro-2-hydroxyacetophenone, followed by a Wittig reaction to construct the carbon skeleton, as depicted in the initial stages of the new synthetic pathway.
By employing a chiral inversion strategy and selective epoxide ring-opening reactions, the process effectively differentiates between the required SRRR and RSSS isomers without the need for extensive chromatographic purification. The reaction conditions are notably gentle, avoiding the extreme thermal stresses seen in prior art, which minimizes degradation and byproduct formation. This approach not only simplifies the operational workflow but also significantly reduces the environmental footprint by eliminating toxic waste streams associated with complex purification steps. For procurement teams, this translates to a more predictable supply of high-purity nebivolol intermediates with a lower risk of batch failure due to process variability.
Mechanistic Insights into Stereoselective Epoxide Formation and Coupling
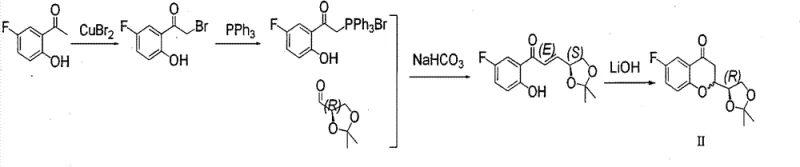
The core of this synthetic achievement lies in the precise manipulation of stereochemistry during the formation of the chroman ring and the subsequent epoxide intermediates. The process initiates with the cyclization of the Wittig product to form the fluorochromanone derivative (Formula II), where the chiral center from the glyceraldehyde acetonide dictates the stereochemistry at the C2 position. Subsequent reduction of the ketone and functional group manipulations lead to the formation of key epoxy intermediates (Formula IVa, IVb, IVc, IVd). The stereochemical outcome of these epoxides is critical, as they serve as the electrophilic partners in the final coupling reaction. The use of Mitsunobu conditions or base-mediated cyclization allows for the inversion or retention of configuration as needed to access all four required stereoisomers.
Impurity control is rigorously managed through the selection of specific reagents and conditions that favor the desired diastereomer. For example, the ring-opening of the epoxide by the amine nucleophile proceeds with high regioselectivity and stereospecificity, ensuring that the secondary alcohol and the amine linkage are formed with the correct spatial arrangement. The final debenzylation step using palladium on carbon under hydrogen pressure cleanly removes the protecting groups without affecting the sensitive ether linkages or the fluorine substituent. This mechanistic precision ensures that the final product mixture contains predominantly the therapeutically active SRRR and RSSS isomers, minimizing the burden on downstream purification and ensuring consistent quality for the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Nebivolol Hydrochloride Efficiently
The synthesis of Nebivolol Hydrochloride via this patented route involves a logical sequence of transformations designed for maximum efficiency and yield. Starting from simple achiral precursors and a single chiral source, the process builds molecular complexity step-by-step while maintaining strict control over stereochemistry. The detailed standardized synthesis steps involve specific protocols for bromination, Wittig olefination, cyclization, and the critical epoxide coupling, all of which are optimized for industrial feasibility. For a comprehensive guide on executing these reactions with precise stoichiometry and workup procedures, refer to the structured protocol below.
- Prepare the chromanone intermediate (Formula II) via bromination of 5-fluoro-2-hydroxyacetophenone followed by a Wittig reaction with D-glyceraldehyde acetonide and subsequent cyclization.
- Convert the chromanone to chiral epoxides (Formula IVa-d) through reduction, protection/deprotection sequences, and Mitsunobu or base-mediated cyclization to establish the required stereochemistry.
- Perform the convergent coupling of the chiral epoxides with benzylamine derivatives, followed by catalytic hydrogenation for debenzylation and salt formation to yield Nebivolol Hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis route offers transformative benefits for supply chain stability and cost management. By eliminating the dependency on unstable and expensive L-glyceraldehyde acetonide, manufacturers can secure raw material sourcing at a fraction of the historical cost. The reduction in total synthetic steps from nearly 30 to roughly 20 directly correlates with improved throughput and reduced labor costs, allowing for faster turnaround times on large orders. Furthermore, the avoidance of extreme temperature requirements (-20°C or 145°C) means that standard reactor infrastructure can be utilized without specialized cooling or heating investments, lowering capital expenditure barriers for production facilities.
- Cost Reduction in Manufacturing: The substitution of high-cost chiral reagents with affordable alternatives significantly lowers the raw material bill of materials. Additionally, the reduction in purification steps, specifically the minimization of column chromatography in favor of crystallization and filtration, drastically cuts solvent consumption and waste disposal costs. This leaner process architecture ensures that the final API intermediate is produced with a much lower cost basis, enhancing margin potential for downstream drug manufacturers.
- Enhanced Supply Chain Reliability: Utilizing commercially available and stable starting materials mitigates the risk of supply disruptions caused by the scarcity of specialized chiral pools. The robustness of the reaction conditions ensures high batch-to-batch consistency, reducing the likelihood of production delays due to failed batches or out-of-specification results. This reliability is crucial for maintaining continuous supply lines for essential cardiovascular medications, ensuring that reducing lead time for high-purity pharmaceutical intermediates becomes a tangible reality.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, avoiding toxic solvents and reagents that complicate regulatory approval and waste management. The mild reaction conditions and simplified workup procedures make the technology highly scalable from pilot plant to multi-ton commercial production. This environmental compatibility not only reduces the ecological footprint but also streamlines the regulatory filing process, accelerating time-to-market for generic versions of Nebivolol.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Nebivolol Hydrochloride using this advanced methodology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and chemical nuances of the process for industry stakeholders. Understanding these details is essential for evaluating the feasibility of adopting this route for large-scale manufacturing.
Q: Why is this synthesis route preferred over previous methods for Nebivolol?
A: This route avoids the use of expensive and unstable L-glyceraldehyde acetonide, relying instead on cheap and stable D-glyceraldehyde acetonide. It also eliminates the need for extreme reaction conditions like -20°C or 145°C found in prior art, making it safer and more suitable for industrial production.
Q: How does the process control the four chiral centers in Nebivolol?
A: The process utilizes a combination of chiral pool synthesis starting from D-glyceraldehyde acetonide and stereoselective reactions such as epoxide ring-opening. Specific intermediates like Formula IVa, IVb, IVc, and IVd are prepared with defined stereochemistry to ensure the final product contains the correct SRRR and RSSS isomers.
Q: Is this method scalable for commercial API manufacturing?
A: Yes, the method is designed for industrialization. It reduces the total number of steps to approximately 17-21, avoids column chromatography in favor of crystallization where possible, and uses environmentally friendly solvents and reagents, addressing key scalability and cost concerns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nebivolol Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for life-saving cardiovascular drugs. Our team of expert chemists has extensively analyzed the technological breakthroughs presented in CN102344431B and possesses the capability to implement this advanced chiral synthesis at an industrial level. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Nebivolol intermediate meets the highest global standards.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthesis technology for your API requirements. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating exactly how this route can optimize your procurement budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the global pharmaceutical market.
