Scalable Synthesis of Jaspine B Isomers: A Novel L-Serine Route for Commercial API Production
The pharmaceutical industry continuously seeks efficient pathways for synthesizing bioactive natural products, particularly those exhibiting potent anticancer properties. Patent CN103601706A introduces a groundbreaking method for synthesizing isomers of Jaspine B, also known as Pachastrissamine, a furan-containing sphingosine compound first isolated from marine sponges. This molecule is renowned for its significant inhibitory effects on various cancer cell lines such as P388 and HT29 at nanogram levels, making it a high-value target for drug discovery. The disclosed technology leverages cheap and accessible L-serine as the initial chiral pool material, bypassing the limitations of traditional sugar-based syntheses. By integrating methyl ester protection, Corey-Fuchs reaction, and a novel silver nitrate catalyzed cyclization, this route achieves the total synthesis of the target molecule with three continuous chiral centers in a syn-configuration. This approach not only establishes a firm foundation for the complete synthesis of natural Jaspine B but also offers a robust platform for generating diverse analogs for structure-activity relationship studies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Jaspine B and its analogs has relied heavily on carbohydrate starting materials such as D-xylose, D-ribose, D-glucose, or Garner’s aldehyde. While these chiral pools provide the necessary stereochemical framework, the resulting synthetic routes are often excessively long and convoluted. Conventional methodologies frequently suffer from slow reaction rates and low overall yields due to the accumulation of losses over numerous steps. Furthermore, the manipulation of multiple hydroxyl groups in sugar derivatives often requires extensive protecting group strategies, which adds to the cost and complexity of the process. A critical drawback in these traditional approaches is the propensity for chiral centers to undergo racemization under harsh reaction conditions, compromising the optical purity of the final API intermediate. These factors collectively hinder the commercial viability of sugar-based routes, creating a significant bottleneck for researchers aiming to produce sufficient quantities for preclinical and clinical evaluation.
The Novel Approach
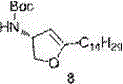
In stark contrast, the method described in patent CN103601706A utilizes L-serine, an abundant and inexpensive amino acid, to construct the sphingosine backbone with superior efficiency. This novel approach streamlines the synthesis by employing a strategic sequence of methyl esterification, tert-butoxycarbonyl (Boc) protection, and propylidene protection to establish the core stereocenters early in the sequence. The integration of the Corey-Fuchs reaction allows for the precise installation of the terminal alkyne functionality, which serves as a versatile handle for subsequent chain elongation. Most notably, the route features a unique metal-catalyzed cyclization step using silver nitrate to construct the tetrahydrofuran ring, a transformation that is both rapid and highly selective. This design results in a single product structure with minimal side reactions, effectively overcoming the yield and purity issues associated with older methods while significantly reducing the operational complexity for manufacturing teams.
Mechanistic Insights into Silver Nitrate Catalyzed Cyclization
The cornerstone of this synthetic innovation lies in the silver nitrate catalyzed construction of the furan ring, which transforms the acyclic alkyne precursor into the cyclic core of the Jaspine B isomer. In this critical step, the terminal alkyne moiety, generated via the Corey-Fuchs protocol and subsequent alkylation, undergoes an intramolecular cyclization facilitated by silver ions. The reaction is conducted in anhydrous tetrahydrofuran with the addition of activated 4A molecular sieves to maintain strictly dry conditions, which is essential for preventing catalyst deactivation. Upon heating to reflux, the silver catalyst activates the alkyne towards nucleophilic attack by the proximal hydroxyl group, driving the formation of the five-membered heterocycle with high regioselectivity. This mechanistic pathway avoids the use of toxic transition metals often found in alternative cyclization methods, thereby simplifying the downstream purification process and ensuring the final product meets stringent heavy metal specifications required for pharmaceutical applications.
Beyond the primary cyclization event, the entire synthetic sequence is meticulously designed to preserve the integrity of the three consecutive chiral centers located at the C-2 to C-4 positions. The use of mild reaction conditions, such as the low-temperature DIBAL-H reduction at -78°C and the ambient temperature deprotection steps, minimizes the thermal stress on the stereocenters. This careful control prevents epimerization or racemization, ensuring that the final isomer retains the desired syn-configuration essential for biological activity. Furthermore, the selection of robust protecting groups like the Boc and propylidene moieties provides orthogonal stability, allowing for selective deprotection without affecting other sensitive functionalities. This high level of stereochemical fidelity translates directly into a cleaner impurity profile, reducing the burden on quality control laboratories and facilitating faster regulatory approval for the resulting pharmaceutical intermediates.
How to Synthesize Jaspine B Isomer Efficiently
The execution of this synthesis begins with the conversion of L-serine into a protected oxazolidine intermediate, setting the stage for chain elongation. Following the reduction to the aldehyde and conversion to the terminal alkyne, the carbon chain is extended via alkylation with iodotetradecane to mimic the natural lipid tail. The pivotal silver-catalyzed cyclization then closes the ring, after which hydroboration-oxidation and Swern oxidation install the remaining hydroxyl functionalities. The detailed standardized synthesis steps, including specific reagent quantities, temperature profiles, and workup procedures for each transformation from Formula 1 to Formula 10, are outlined in the guide below.
- Protect L-Serine via methyl esterification and Boc-protection, followed by propylidene protection to form the oxazolidine intermediate.
- Perform DIBAL-H reduction and Corey-Fuchs reaction to generate the terminal alkyne, followed by alkylation with iodotetradecane.
- Execute the key silver nitrate catalyzed cyclization to construct the furan ring, followed by hydroboration and deprotection to yield the target isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from complex sugar-based syntheses to this L-serine driven route represents a substantial opportunity for cost optimization and risk mitigation. The reliance on L-serine, a commodity chemical produced on a massive scale for the food and feed industries, ensures a stable and predictable raw material supply chain that is immune to the agricultural fluctuations affecting sugar feedstocks. Additionally, the reduction in the total number of synthetic steps directly correlates to lower consumption of solvents, reagents, and labor hours, driving down the variable cost of goods sold. The simplified operational technique, characterized by standard unit operations like extraction and column chromatography rather than specialized equipment, enhances the flexibility of manufacturing facilities to scale production up or down based on market demand without significant capital expenditure.
- Cost Reduction in Manufacturing: The elimination of expensive and scarce chiral sugars in favor of L-serine drastically lowers the starting material costs, which is a primary driver of overall manufacturing expenses. Furthermore, the high yield of the key silver-catalyzed cyclization step reduces the amount of wasted material, improving the atom economy of the process. By avoiding the need for complex resolution steps to correct racemization, the process saves significant resources on chiral separation technologies and associated consumables. These cumulative efficiencies result in a leaner cost structure that allows for more competitive pricing of the final high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Sourcing L-serine is far more reliable than procuring specialized chiral sugars, as it is produced by multiple global suppliers with consistent quality standards. The robustness of the synthetic route, with its tolerance for standard laboratory conditions and lack of ultra-sensitive reagents, minimizes the risk of batch failures due to minor environmental variations. This reliability ensures consistent delivery schedules, which is critical for maintaining the continuity of downstream drug development programs. Consequently, partners can reduce safety stock levels and optimize inventory turnover, knowing that the supply of this critical intermediate is secure and resilient against external disruptions.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing common organic solvents like dichloromethane, methanol, and tetrahydrofuran that are easily recovered and recycled in industrial settings. The absence of highly toxic heavy metal catalysts, relying instead on silver nitrate which can be recovered, simplifies waste treatment and ensures compliance with increasingly stringent environmental regulations. The mild reaction conditions also reduce energy consumption for heating and cooling, contributing to a lower carbon footprint for the manufacturing process. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the scalability, purity, and specific reaction conditions of this patented synthesis method. Understanding these details is crucial for R&D teams evaluating the feasibility of adopting this route for their own pipeline projects. The answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a transparent view of the technology's capabilities and limitations.
Q: What are the advantages of using L-Serine over sugar-based starting materials for Jaspine B synthesis?
A: Using L-Serine significantly shortens the synthetic route compared to traditional sugar-based methods like D-xylose or D-ribose. It offers better control over the three consecutive chiral centers, reducing the risk of racemization and simplifying purification processes.
Q: How does the silver nitrate catalyzed step improve the overall yield?
A: The silver nitrate catalyzed cyclization efficiently constructs the critical furan ring under mild reflux conditions. This specific metal catalysis minimizes side reactions and ensures a single product structure, leading to higher isolated yields of the key intermediate.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the route utilizes cheap and accessible raw materials like L-Serine and employs standard organic transformations. The mild reaction conditions and simple operating techniques make it highly adaptable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Jaspine B Isomer Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient synthetic routes like the one described in CN103601706A for accelerating drug discovery timelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis, including low-temperature reactions and moisture-sensitive catalytic steps, while our rigorous QC labs enforce stringent purity specifications to guarantee the quality of every batch. We are committed to delivering high-purity Jaspine B isomers that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with our technical procurement team to explore how this advanced synthesis can optimize your supply chain and reduce your overall project costs. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to support your next breakthrough in oncology research.
