Advanced Catalytic Synthesis of Sulfamate-Functionalized Butenolide Intermediates for Scalable Production
Introduction to Next-Generation Butenolide Chemistry
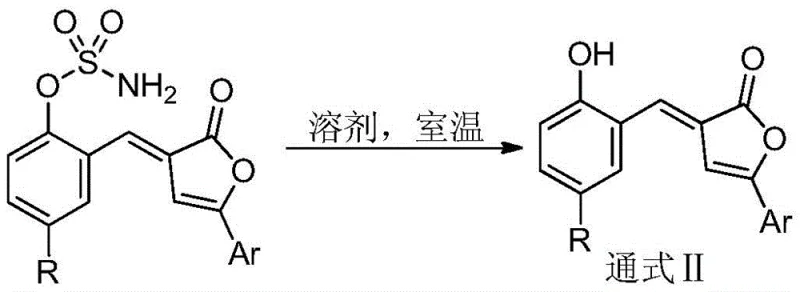
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more efficient and sustainable synthetic pathways. A significant breakthrough in this domain is documented in patent CN108440466B, which introduces a novel class of 5-aryl-3-(2-sulfamate-5-substituted benzylidene)butenolide compounds. These structures represent a critical advancement in the field of heterocyclic chemistry, offering a robust platform for the development of pharmaceutical intermediates and agrochemical agents. The core innovation lies in the strategic incorporation of a sulfamate moiety onto the butenolide scaffold, a modification that not only enhances the chemical versatility of the molecule but also streamlines the synthetic process. By leveraging a catalytic condensation reaction between benzo[1,2,3]oxathiazine 2,2-dioxide and 5-arylbutenolides, this technology enables the construction of complex molecular architectures under exceptionally mild conditions. For R&D directors and procurement specialists alike, this represents a shift towards high-atom-economy processes that minimize waste while maximizing yield potential.
From a commercial perspective, the implications of this patent extend far beyond the laboratory bench. The ability to synthesize these specialized intermediates at room temperature using environmentally benign solvents addresses two of the most pressing challenges in modern chemical supply chains: energy efficiency and regulatory compliance. Traditional methods for functionalizing butenolide rings often require harsh reagents, elevated temperatures, or toxic heavy metal catalysts, all of which introduce significant cost burdens and safety risks. In contrast, the methodology outlined in CN108440466B utilizes simple organic bases and common solvents, creating a pathway that is inherently safer and more cost-effective. This report delves deep into the technical nuances of this synthesis, evaluating its feasibility for commercial scale-up and its potential to serve as a reliable source for high-purity pharmaceutical intermediates in a competitive global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of functionalized butenolide derivatives has been plagued by inefficiencies that hinder large-scale adoption. Conventional routes often rely on multi-step sequences involving protecting group manipulations, stoichiometric amounts of hazardous reagents, and rigorous temperature control. For instance, introducing oxygen or nitrogen functionality at the ortho-position of a phenolic ring typically necessitates the use of strong electrophiles or transition metal catalysts that are difficult to remove from the final product. These residual metals can be detrimental in pharmaceutical applications, requiring additional purification steps that drive up costs and reduce overall throughput. Furthermore, many traditional protocols demand anhydrous conditions and inert atmospheres, adding layers of operational complexity that are ill-suited for continuous manufacturing environments. The cumulative effect of these limitations is a supply chain that is fragile, expensive, and prone to bottlenecks, particularly when scaling from gram to kilogram quantities.
The Novel Approach
The technology described in patent CN108440466B offers a paradigm shift by simplifying the synthetic logic into a direct, catalytic coupling. The core reaction involves the condensation of benzo[1,2,3]oxathiazine 2,2-dioxide with 5-arylbutenolides in the presence of a basic catalyst. This approach bypasses the need for pre-functionalization or harsh activation steps, allowing the reaction to proceed smoothly at room temperature. The use of benzo[1,2,3]oxathiazine 2,2-dioxide as a reagent is particularly ingenious, as it serves as a masked source of the sulfamate group, which is installed directly onto the aromatic ring during the bond-forming event. This atom-economic strategy ensures that the majority of the starting material mass is incorporated into the final product, drastically reducing chemical waste. Moreover, the tolerance for diverse solvents, including water and alcohols, underscores the green chemistry credentials of this method, making it an attractive option for manufacturers aiming to reduce their environmental footprint while maintaining high productivity standards.
![General reaction scheme showing the catalytic synthesis of 5-aryl-3-(2-sulfamate-5-substituted benzylidene)butenolide from benzo[1,2,3]oxathiazine 2,2-dioxide and 5-arylbutenolide](/insights/img/sulfamate-butenolide-synthesis-pharma-supplier-20260307071913-01.webp)
Mechanistic Insights into Organocatalytic Condensation
Understanding the mechanistic underpinnings of this transformation is crucial for optimizing process parameters and ensuring consistent quality. The reaction likely proceeds through a nucleophilic attack mechanism where the basic catalyst activates the 5-arylbutenolide, enhancing its nucleophilicity at the gamma-position. This activated species then attacks the electrophilic center of the benzo[1,2,3]oxathiazine 2,2-dioxide. The unique electronic structure of the oxathiazine dioxide facilitates the cleavage of the N-O bond, leading to the formation of the new carbon-carbon double bond and the simultaneous installation of the sulfamate group on the adjacent phenolic oxygen. This concerted or stepwise process is highly sensitive to the electronic nature of the substituents on both the butenolide and the oxathiazine rings. Electron-withdrawing groups on the butenolide may enhance acidity and facilitate enolate formation, while electron-donating groups on the oxathiazine could stabilize the transition state. The result is a highly selective formation of the Z-isomer or E-isomer depending on steric factors, although the patent indicates a high degree of stereocontrol favoring the thermodynamically stable configuration.
Impurity control is another critical aspect where this mechanism offers distinct advantages. Because the reaction occurs under mild, neutral-to-basic conditions without strong oxidants or reductants, the formation of side products such as over-oxidized species or polymerization byproducts is minimized. The primary impurities are likely to be unreacted starting materials or hydrolysis products of the sulfamate group, both of which are easily removed during the aqueous workup. The stability of the sulfamate ester under the reaction conditions is remarkable, preventing premature degradation before the final isolation step. This inherent stability simplifies the purification process, often allowing for direct crystallization or simple flash chromatography. For quality assurance teams, this means a cleaner crude profile and a more predictable impurity spectrum, which is essential for meeting the stringent specifications required for API intermediates. The mechanistic clarity also allows for rational tuning of the catalyst loading and solvent choice to further suppress minor byproducts.
How to Synthesize 5-Aryl-3-benzylidene Butenolide Efficiently
Implementing this synthesis in a production setting requires adherence to specific operational protocols to maximize yield and safety. The process begins with the precise weighing of benzo[1,2,3]oxathiazine 2,2-dioxide and the chosen 5-arylbutenolide derivative. These solids are introduced into a reaction vessel equipped with efficient stirring capabilities. A solvent system is then selected based on solubility profiles; while water is preferred for its green credentials, mixtures of water with co-solvents like acetonitrile or THF may be employed to improve homogeneity. Once the substrates are dissolved or suspended, the catalyst, typically triethylamine or DABCO, is added. The reaction mixture is maintained at ambient temperature, eliminating the need for external heating or cooling loops. Monitoring is conducted via thin-layer chromatography (TLC) to track the consumption of the starting material. Upon completion, the reaction is quenched, and the product is extracted into an organic phase, dried, and purified. Detailed standardized operating procedures for this synthesis are provided below to ensure reproducibility across different batches and facilities.
- Mix benzo[1,2,3]oxathiazine 2,2-dioxide and 5-arylbutenolide in a reactor with a suitable solvent such as water or dichloromethane.
- Add an organic base catalyst like triethylamine or DABCO and stir the mixture at room temperature for 1 to 3 hours.
- Upon completion confirmed by TLC, perform extraction, separation, and purification via column chromatography to isolate the target sulfamate compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology translates into tangible economic and logistical benefits. The most immediate advantage is the drastic reduction in energy costs associated with the elimination of heating and cooling cycles. Running reactions at room temperature significantly lowers the utility load on manufacturing plants, contributing to a lower cost of goods sold (COGS). Additionally, the use of non-halogenated and potentially aqueous solvent systems reduces the expense and regulatory burden associated with solvent recovery and disposal. This aligns perfectly with global trends towards sustainable manufacturing, potentially qualifying the process for green chemistry incentives or preferential sourcing status from environmentally conscious clients. The simplicity of the workup procedure also means faster turnaround times between batches, increasing the overall capacity utilization of existing reactor infrastructure without the need for capital-intensive equipment upgrades.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of commodity amine bases like triethylamine significantly lower raw material costs. Furthermore, the high atom economy of the reaction ensures that less waste is generated per kilogram of product, reducing waste treatment fees. The mild conditions also extend the lifespan of reactor vessels and seals by avoiding corrosive reagents and extreme thermal stress, leading to lower maintenance overheads over the long term.
- Enhanced Supply Chain Reliability: The starting materials, benzo[1,2,3]oxathiazine 2,2-dioxide and various 5-arylbutenolides, are either commercially available or can be synthesized via established, robust routes. This reduces the risk of supply disruptions caused by reliance on exotic or single-source reagents. The broad substrate scope demonstrated in the patent, accommodating various halogen and alkyl substituents, allows for flexible sourcing strategies where alternative substituted precursors can be utilized if specific grades become unavailable, ensuring continuity of supply for downstream customers.
- Scalability and Environmental Compliance: The exothermic profile of the reaction is manageable at room temperature, mitigating the risk of thermal runaway during scale-up. This safety margin allows for larger batch sizes and the potential transition to continuous flow processing. From an environmental standpoint, the avoidance of heavy metals and chlorinated solvents simplifies the permitting process for new manufacturing lines and ensures compliance with increasingly strict international environmental regulations, safeguarding the supply chain against future regulatory shocks.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical and commercial viability of this process, we have compiled a set of frequently asked questions based on the patent data. These answers provide clarity on the operational flexibility and application potential of the sulfamate butenolide intermediates. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their existing portfolios. The following section covers key aspects ranging from reaction optimization to downstream utility, ensuring that all technical concerns are proactively managed.
Q: What are the primary advantages of this room-temperature synthesis route?
A: The process operates at room temperature using benign solvents like water or ethanol, significantly reducing energy consumption and eliminating the need for cryogenic cooling or high-pressure equipment often required in traditional heterocyclic synthesis.
Q: Can this intermediate be converted into other valuable scaffolds?
A: Yes, the sulfamate group serves as a versatile leaving group that can be readily displaced under mild solvolysis conditions to generate 5-aryl-3-o-hydroxybenzylidene butenolides, which are key precursors for bioactive molecules.
Q: Is this methodology suitable for large-scale manufacturing?
A: The protocol utilizes commercially available starting materials and simple workup procedures involving standard extraction and chromatography, making it highly adaptable for kilogram-to-ton scale production without complex safety hazards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Aryl-3-benzylidene Butenolide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN108440466B for producing high-value pharmaceutical intermediates. Our team of process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering these complex sulfamate-functionalized butenolides with stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our infrastructure is designed to handle the specific solvent and catalyst requirements of this green chemistry protocol, guaranteeing a consistent supply of material that meets your exacting needs for drug development and commercial manufacturing.
We invite you to collaborate with us to leverage this advanced technology for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain with a reliable partner dedicated to innovation, quality, and sustainable growth in the fine chemical sector.
