Advanced Green Oxidative Synthesis of N-Substituted 2H-Indazole Compounds for Commercial Scale-Up
Advanced Green Oxidative Synthesis of N-Substituted 2H-Indazole Compounds for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust, scalable, and environmentally benign pathways for constructing complex heterocyclic scaffolds essential for modern drug discovery. A significant breakthrough in this domain is documented in Chinese Patent CN112574112B, which discloses a novel method for the green oxidative synthesis of N-substituted 2H-indazole compounds. This technology addresses critical bottlenecks in the manufacturing of indazole-based active pharmaceutical ingredients (APIs), such as tyrosine kinase inhibitors and anti-cancer agents. By leveraging a persulfate-mediated oxidation strategy, this process eliminates the reliance on precious metal catalysts and harsh reaction conditions, offering a streamlined route that aligns perfectly with the principles of green chemistry. For procurement managers and R&D directors alike, this represents a pivotal shift towards more sustainable and cost-efficient production of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of N-substituted indazole frameworks has relied heavily on transition-metal catalysis or emerging but problematic technologies like visible-light photoredox catalysis. While photoredox methods offer unique reactivity, they are plagued by significant commercial drawbacks, including the necessity for toxic and expensive photocatalytic reagents that complicate downstream purification and increase raw material costs. Furthermore, these photochemical processes often suffer from prolonged reaction times, frequently extending up to 24 hours, which severely limits throughput in a manufacturing setting. Similarly, electrochemical approaches, though promising, often require expensive cathode materials such as platinum, creating a barrier to entry for large-scale adoption due to high capital expenditure and maintenance costs associated with specialized electrochemical reactors.
The Novel Approach
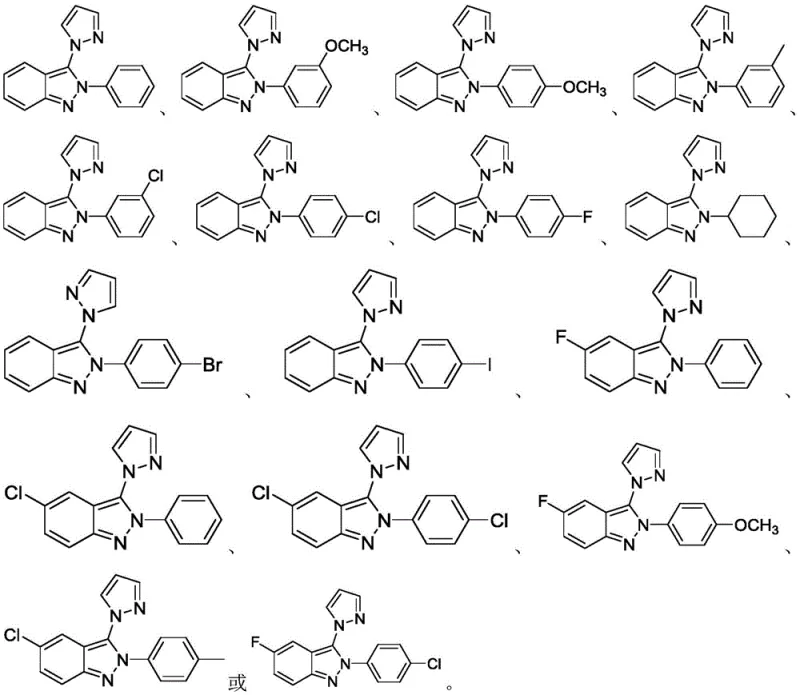
In stark contrast, the methodology outlined in patent CN112574112B introduces a paradigm shift by utilizing inexpensive and readily available inorganic oxidants, specifically sodium persulfate, potassium persulfate, or ammonium persulfate. This approach operates under mild thermal conditions, typically between 60°C and 100°C, and achieves completion within a concise 10 to 12-hour window. The reaction exhibits exceptional substrate tolerance, accommodating a wide range of substituents on the indazole nitrogen, including phenyl, substituted phenyl, and cyclohexyl groups. This operational simplicity translates directly into reduced process complexity, allowing manufacturers to utilize standard reactor setups without the need for specialized light sources or electrochemical cells, thereby significantly lowering the barrier for commercial implementation.

Mechanistic Insights into Persulfate-Mediated Oxidative Coupling
The core of this innovation lies in the efficient generation of radical species facilitated by the thermal decomposition of persulfate salts. Under the specified reaction conditions, the persulfate oxidant acts as a single-electron transfer agent, initiating a radical cascade that promotes the direct C-H amination of the indazole core with pyrazole derivatives. This mechanism bypasses the need for pre-functionalized substrates, such as halogenated precursors, which are often required in traditional cross-coupling reactions. The radical pathway ensures high atom economy and minimizes the formation of stoichiometric salt byproducts, a common issue in classical nucleophilic substitution reactions. From a process chemistry perspective, this radical mechanism is highly advantageous as it proceeds cleanly in polar solvents, including water, which further enhances the environmental profile of the synthesis.
Impurity control is another critical aspect where this method excels. The use of well-defined inorganic oxidants reduces the risk of metal contamination, a stringent requirement for API manufacturing. The reaction profile is clean, with the primary byproduct being the reduced sulfate species, which are easily removed during the aqueous workup phase. The patent data indicates that the process tolerates various electronic environments on the aromatic rings, whether electron-donating groups like methoxy and methyl or electron-withdrawing groups like fluoro and chloro. This robustness ensures consistent quality and yield across different batches, providing R&D teams with a reliable platform for generating diverse libraries of indazole analogs for biological screening without the variability often seen in metal-catalyzed systems.
How to Synthesize N-Substituted 2H-Indazole Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and thermal profiles to maximize yield and purity. The process begins with the dissolution of the 2H-indazole starting material and pyrazole in a selected solvent system, followed by the controlled addition of the persulfate oxidant. Maintaining the reaction temperature within the optimal range of 60°C to 100°C is crucial for sustaining the radical flux necessary for coupling. Detailed standardized operating procedures regarding mixing rates, addition sequences, and quenching protocols are essential for reproducibility. For a comprehensive breakdown of the specific experimental steps and conditions validated in the patent examples, please refer to the technical guide below.
- Dissolve the 2H-indazole compound (Formula II) and pyrazole in a suitable solvent such as water or 1,2-dichloroethane.
- Add an oxidizing agent, preferably potassium persulfate, maintaining a molar ratio of indazole to pyrazole to oxidant of approximately 1: 1.5:3.
- Heat the reaction mixture to 60-100°C for 10-12 hours, then perform extraction and column chromatography to isolate the pure N-substituted product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this persulfate-based oxidative synthesis offers tangible strategic benefits that extend beyond mere chemical efficiency. The primary driver for cost reduction lies in the substitution of high-value catalytic systems with commodity chemicals. Potassium persulfate is a bulk industrial chemical with a stable supply chain and low price volatility, unlike specialized photocatalysts or platinum electrodes which are subject to market fluctuations and geopolitical supply risks. Furthermore, the ability to use water as a primary solvent drastically reduces the cost of goods sold (COGS) associated with solvent purchase, recovery, and disposal. This shift not only lowers direct material costs but also simplifies the waste management infrastructure required at the manufacturing site.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and specialized equipment creates a leaner cost structure. By avoiding the need for rigorous metal scavenging steps to meet regulatory limits on residual metals in APIs, manufacturers can reduce both processing time and the consumption of auxiliary purification materials. The high yields reported in the patent examples, often exceeding 80% and reaching up to 90% for specific substrates, mean that less raw material is wasted, directly improving the overall mass balance and economic efficiency of the production line.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis—indazoles, pyrazoles, and persulfates—are widely available from multiple global suppliers, reducing the risk of single-source dependency. This diversification strengthens supply chain resilience against disruptions. Additionally, the simplified reaction setup allows for production in multipurpose facilities without the need for dedicated photochemical or electrochemical suites, increasing manufacturing flexibility and capacity utilization rates across the plant.
- Scalability and Environmental Compliance: The use of water and the generation of benign sulfate byproducts align with increasingly stringent environmental regulations. This green profile facilitates easier permitting for new production lines and reduces the carbon footprint of the manufacturing process. The straightforward workup procedure involving extraction and crystallization is inherently scalable, allowing for seamless transition from kilogram-scale pilot runs to multi-ton commercial production without significant process re-engineering.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its integration into their supply chains. The following questions address common inquiries regarding the practical application, scope, and benefits of this patented technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What are the primary advantages of this oxidative synthesis method over photoredox catalysis?
A: Unlike photoredox methods which require expensive, toxic photocatalysts and long reaction times (up to 24 hours), this method utilizes cheap, non-toxic potassium persulfate and completes in 10-12 hours with high yields.
Q: Is this process scalable for industrial production of API intermediates?
A: Yes, the process uses common solvents like water and standard heating equipment, avoiding specialized electrochemical cells or light sources, making it highly suitable for commercial scale-up.
Q: What is the substrate scope for the R1 group in this synthesis?
A: The method demonstrates broad applicability, successfully synthesizing derivatives where R1 is phenyl, substituted phenyl (with methyl, methoxy, or halogens like F, Cl, Br, I), or cyclohexyl.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Substituted 2H-Indazole Supplier
As the demand for complex heterocyclic intermediates continues to surge in the oncology and anti-inflammatory sectors, having a manufacturing partner with deep technical expertise is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of oxidative synthesis, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We understand that consistency and quality are non-negotiable for your downstream drug development programs, and our team is dedicated to delivering intermediates that exceed these expectations.
We invite you to collaborate with us to leverage this innovative synthetic route for your specific project needs. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this green chemistry approach can optimize your budget. Please contact our technical procurement team today to request specific COA data for our indazole portfolio and to discuss route feasibility assessments for your custom synthesis projects. Together, we can accelerate your path to market with efficient, high-quality chemical solutions.
