Advanced Electrochemical Manufacturing of High-Purity Indoloquinoline Pharmaceutical Intermediates
Introduction to Next-Generation Indoloquinoline Synthesis
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally sustainable pathways for constructing complex heterocyclic scaffolds, particularly those with significant biological activity such as indoloquinolines. A groundbreaking advancement in this domain is detailed in Chinese Patent CN114232013B, which discloses a novel method for synthesizing indoloquinoline compounds under electrochemical conditions. This technology represents a paradigm shift from traditional transition-metal catalysis to organic electrochemical synthesis, utilizing electric current as a clean redox reagent to drive the formation of the core carbon-nitrogen bonds. For R&D directors and procurement specialists, this patent offers a compelling alternative that bypasses the need for expensive noble metal catalysts and stoichiometric oxidants, thereby addressing critical pain points regarding cost, purity, and environmental compliance in the manufacturing of high-value pharmaceutical intermediates.
The significance of this electrochemical approach lies in its ability to generate imine nitrogen radicals through a catalytic decarboxylation process initiated by n-tetrabutylammonium bromide. This mechanism facilitates an intramolecular carbon-nitrogen bond coupling reaction that constructs the rigid indoloquinoline framework with high efficiency. Unlike conventional methods that often suffer from harsh reaction conditions and poor reproducibility, this electrochemical protocol operates under remarkably mild conditions, typically at room temperature. This not only enhances the safety profile of the operation but also expands the functional group tolerance, allowing for the synthesis of diverse derivatives that are crucial for structure-activity relationship (SAR) studies in drug discovery programs targeting cancer and malaria.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indoloquinoline skeleton has relied heavily on transition-metal catalyzed processes, which present substantial challenges for large-scale commercial production. Literature precedents describe methods involving gold-catalyzed cyclization of acyclic alkynes, copper-catalyzed tandem reactions, and palladium-catalyzed Suzuki couplings or isonitrile insertions. While chemically elegant, these routes are plagued by the necessity of using precious metals such as Palladium, Gold, Rhodium, and Manganese, which are not only costly but also pose severe contamination risks in active pharmaceutical ingredients (APIs). The removal of trace metal residues to meet stringent regulatory limits (often in the ppm range) requires additional purification steps, such as scavenging or recrystallization, which inevitably reduce overall yield and increase manufacturing costs. Furthermore, many of these traditional protocols require harsh oxidants or high temperatures, leading to safety hazards and the generation of significant chemical waste that complicates environmental management.
The Novel Approach
In stark contrast, the electrochemical method disclosed in CN114232013B offers a streamlined, metal-free alternative that fundamentally changes the economics of indoloquinoline manufacturing. By employing electricity as the primary driving force for oxidation, the process eliminates the requirement for external chemical oxidants and expensive metal catalysts entirely. The reaction utilizes a simple catalytic system involving n-tetrabutylammonium bromide, which circulates within the reaction medium to facilitate the decarboxylation of the substrate. This results in a cleaner reaction profile with benign byproducts like carbon dioxide and acetone, adhering strictly to the principles of green chemistry. The operational simplicity is another major advantage; the reaction proceeds in an undivided cell at room temperature with constant current, removing the need for specialized high-pressure reactors or cryogenic cooling systems often associated with sensitive organometallic chemistry.
Mechanistic Insights into Electrochemical Decarboxylative Cyclization
The core of this innovative synthesis lies in the anodic oxidation mechanism that triggers a cascade of radical transformations. The process begins with the electrochemical oxidation of the bromide ion from the n-tetrabutylammonium bromide catalyst at the anode surface. This generated active bromine species then mediates the decarboxylation of the oxime ester substrate, a critical step that leads to the homolytic cleavage of the nitrogen-oxygen bond. This cleavage generates a highly reactive imine nitrogen radical intermediate, which is the key species responsible for the subsequent bond-forming event. The radical nature of this intermediate allows for rapid intramolecular attack on the adjacent aromatic ring, forming the new carbon-nitrogen bond that closes the quinoline ring system. This radical pathway is distinct from ionic mechanisms found in acid-catalyzed cyclizations, offering unique selectivity and avoiding the formation of polymeric byproducts often seen in harsh acidic conditions.
From an impurity control perspective, this electrochemical mechanism offers superior predictability and cleanliness. Because the reaction is driven by electron transfer rather than stoichiometric reagents, the concentration of reactive intermediates can be precisely controlled by adjusting the current intensity (typically 2-5 mA in the patent examples). This control minimizes side reactions such as over-oxidation or non-selective radical coupling, which are common pitfalls in chemical oxidation methods. Furthermore, the absence of transition metals means there is no risk of metal-catalyzed decomposition pathways or the formation of metal-organic complexes that are difficult to separate. The resulting crude product typically exhibits a high purity profile, significantly reducing the burden on downstream purification processes like column chromatography or preparative HPLC, which is a critical factor for cost reduction in pharmaceutical intermediate manufacturing.
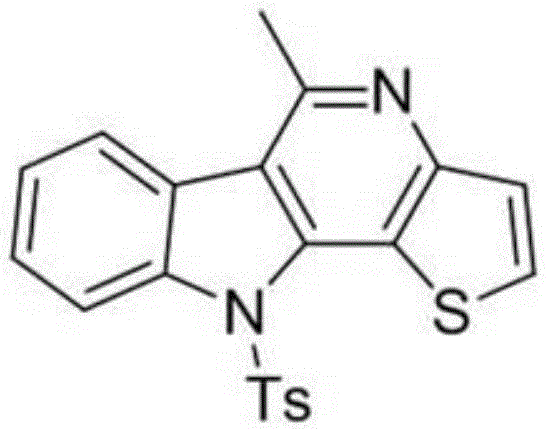
![Chemical structure of 6-methyl-11-tosyl-11H-indolo[3,2-c]quinoline representing the core scaffold synthesized via electrochemical method](/insights/img/indoloquinoline-electrochemical-synthesis-pharma-supplier-20260304141313-01.png)
How to Synthesize Indoloquinoline Derivatives Efficiently
The practical implementation of this electrochemical synthesis involves a multi-step sequence that begins with the preparation of the requisite oxime ether substrate from readily available 2-phenylindoles. The substrate is then subjected to electrolysis in a mixture of hexafluoroisopropanol (HFIP) and trifluoroethanol (TFE), solvents chosen for their ability to stabilize radical intermediates and enhance conductivity. The detailed standardized synthetic steps, including specific molar ratios, electrode configurations, and workup procedures, are outlined in the guide below to ensure reproducibility and safety during scale-up operations.
- Prepare the oxime ether substrate by reacting substituted 2-phenyl-3-acetylindole with 2-(aminooxy)-2-methylpropionate in ethanol with sodium acetate.
- Set up an undivided electrochemical cell with an RVC anode and platinum cathode, dissolving the substrate in HFIP/TFE solvent mixture with n-Bu4NBF4 electrolyte.
- Apply a constant current of 2-5 mA at room temperature for 5-9 hours to induce decarboxylative radical cyclization, followed by standard extraction and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical technology translates into tangible strategic advantages beyond mere technical novelty. The primary benefit is the drastic simplification of the supply chain for raw materials; by removing the dependency on volatile and expensive precious metal catalysts like Palladium or Gold, manufacturers can insulate themselves from market price fluctuations and supply shortages of these critical resources. Additionally, the elimination of stoichiometric oxidants reduces the inventory of hazardous chemicals required on-site, lowering storage costs and regulatory compliance burdens associated with dangerous goods. The mild reaction conditions also imply lower energy consumption for heating or cooling, contributing to a reduced carbon footprint and aligning with corporate sustainability goals which are increasingly important for multinational pharmaceutical partners.
- Cost Reduction in Manufacturing: The most significant economic driver for this technology is the complete removal of expensive transition metal catalysts from the bill of materials. In traditional synthesis, the cost of palladium or rhodium catalysts, combined with the ligands required to stabilize them, can constitute a substantial portion of the total production cost. By replacing these with inexpensive n-tetrabutylammonium bromide and electricity, the direct material cost is significantly lowered. Furthermore, the simplified purification process resulting from the absence of metal residues reduces the consumption of silica gel, solvents, and labor hours required for chromatography, leading to substantial overall cost savings in the production of high-purity indoloquinoline intermediates.
- Enhanced Supply Chain Reliability: Relying on electrochemical synthesis diversifies the sourcing strategy away from specialized catalyst suppliers who may have long lead times or geopolitical supply risks. The reagents used in this process, such as tetrabutylammonium salts and common solvents like ethanol and ethyl acetate, are commodity chemicals available from multiple global vendors. This commoditization of inputs ensures a more resilient supply chain, reducing the risk of production stoppages due to raw material shortages. Moreover, the robustness of the electrochemical method across various substituted substrates means that a single production line can be adapted to manufacture a wide library of derivatives, enhancing flexibility in responding to changing customer demands.
- Scalability and Environmental Compliance: Scaling electrochemical reactions is inherently straightforward as it often involves increasing electrode surface area or using flow chemistry setups, avoiding the heat transfer limitations common in exothermic chemical oxidations. The process generates minimal hazardous waste, with the primary byproducts being gaseous carbon dioxide and acetone, which are easy to manage and dispose of. This clean profile simplifies the environmental permitting process for new manufacturing facilities and reduces the costs associated with wastewater treatment and hazardous waste disposal, making it an ideal candidate for green manufacturing initiatives and regulatory approval in strict jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of indoloquinolines, based on the data provided in patent CN114232013B. These answers are designed to clarify the feasibility, safety, and quality implications of adopting this technology for commercial production, providing decision-makers with the confidence to integrate this method into their supply chains.
Q: Why is electrochemical synthesis preferred over traditional metal catalysis for indoloquinolines?
A: Traditional methods rely on expensive and toxic transition metals like Palladium, Gold, or Rhodium, which require complex removal steps to meet pharmaceutical purity standards. The electrochemical method described in CN114232013B utilizes electrons as clean reagents, eliminating heavy metal contamination risks and significantly simplifying downstream purification.
Q: What are the scalability advantages of this electrochemical protocol?
A: The process operates under mild conditions (room temperature, ambient pressure) and uses simple undivided cells with commercially available electrodes like RVC and Platinum. This avoids the harsh conditions and specialized high-pressure equipment often needed for traditional cyclization, facilitating easier scale-up from gram to tonnage production.
Q: How does this method impact the environmental footprint of API intermediate manufacturing?
A: By replacing stoichiometric chemical oxidants with electricity, the process generates benign byproducts such as carbon dioxide and acetone. This aligns with green chemistry principles, reducing hazardous waste disposal costs and improving the overall E-factor of the synthesis compared to conventional oxidative cyclization methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indoloquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in the production of complex pharmaceutical intermediates like indoloquinolines. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the laboratory success of patent CN114232013B can be seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped to handle electrochemical processes safely and efficiently, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required for clinical and commercial drug substance manufacturing.
We invite global pharmaceutical partners to collaborate with us to leverage this cost-effective and green technology for their pipeline projects. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific target molecules. We are prepared to provide specific COA data from our pilot runs and comprehensive route feasibility assessments to demonstrate how switching to this electrochemical method can optimize your supply chain, reduce costs, and accelerate your time to market for next-generation therapeutic agents.