Scalable Synthesis of Chiral Ferrocene Phosphine Ligands for Advanced Catalytic Applications
Scalable Synthesis of Chiral Ferrocene Phosphine Ligands for Advanced Catalytic Applications
The landscape of asymmetric catalysis is continually evolving, driven by the demand for highly efficient and tunable chiral ligands that can accelerate complex pharmaceutical transformations. Patent CN103193830A introduces a robust and versatile methodology for the preparation of chiral ferrocene phosphine ligands, specifically targeting the PPFA (1-(2-diphenylphosphino)ferrocenyl-N,N-dimethylethylamine) class of compounds. This technology addresses critical bottlenecks in ligand synthesis by utilizing phosphorus trichloride as a central building block, thereby bypassing the need for scarce and expensive diarylphosphine chlorides. For R&D directors and procurement specialists, this represents a significant opportunity to diversify the library of available catalysts while simultaneously optimizing the cost structure of fine chemical manufacturing. The ability to finely tune the electronic and steric environment around the phosphorus center opens new avenues for optimizing enantioselectivity in hydrogenation and coupling reactions.
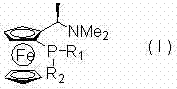
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of PPFA-type ligands has relied heavily on the direct reaction of lithiated Ugi's amine with pre-synthesized diaryl(alkyl)phosphine chlorides. While effective for standard structures, this approach suffers from severe supply chain constraints because many specialized phosphine chlorides are not commercially available or are prohibitively expensive to source in bulk quantities. Furthermore, the structural diversity is inherently limited by the availability of these specific chlorinating agents, restricting chemists to a narrow subset of possible ligand architectures. This dependency creates a vulnerability in the supply chain for high-value asymmetric intermediates, as any disruption in the supply of niche phosphine chlorides can halt production lines. Additionally, the purification of byproducts from these direct couplings can be challenging, often requiring extensive chromatography which reduces overall process efficiency and increases waste generation significantly.
The Novel Approach
The innovative strategy disclosed in the patent fundamentally shifts the synthetic paradigm by introducing phosphorus substituents sequentially onto a reactive dichloride intermediate derived from inexpensive PCl3. By first generating a ferrocenyl-phosphorus dichloride species through ortho-lithiation and subsequent phosphorylation, the method creates a highly reactive hub that can accept a wide variety of nucleophiles. This allows for the introduction of diverse alkyl, aryl, heteroaryl, and cycloalkyl groups using standard organolithium or Grignard reagents, which are commodity chemicals with stable global supply chains. The flexibility of this approach means that ligand properties can be customized on demand without waiting for specialized reagent synthesis, drastically reducing lead times for new catalyst development. Moreover, the stepwise nature of the substitution allows for the precise construction of P-chiral centers, a feature that is difficult to achieve with high fidelity using conventional one-pot coupling methods.
Mechanistic Insights into Stepwise Phosphorus Substitution
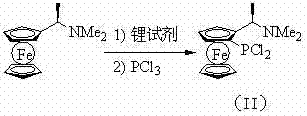
The core of this technology lies in the controlled activation of the ferrocene backbone and the subsequent manipulation of the phosphorus oxidation state. The process initiates with the diastereoselective lithiation of Ugi's amine, directed by the dimethylamino group to the ortho-position of the cyclopentadienyl ring. This lithiated species is then reacted with phosphorus trichloride at cryogenic temperatures ranging from -120°C to -60°C to prevent side reactions and ensure the formation of the dichloride intermediate (II). Maintaining strict temperature control during this exothermic phosphorylation step is critical for maximizing yield and minimizing the formation of polymeric phosphorus byproducts. The resulting dichloride serves as a versatile electrophile, ready to undergo nucleophilic attack by organometallic reagents to install the desired organic substituents on the phosphorus atom.
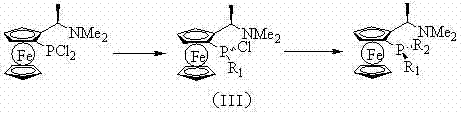
For the creation of P-chiral ligands, the patent describes a sophisticated sequential substitution mechanism that leverages the difference in reactivity between the two chlorine atoms on the phosphorus center. By adding exactly one equivalent of a first organometallic reagent (R1Li or R1MgX) at low temperatures, a monochloride intermediate (III) is formed with high selectivity. This intermediate is then subjected to a second substitution with a different organometallic reagent (R2Li or R2MgX) to complete the ligand structure with two distinct groups attached to the phosphorus. This stepwise protocol ensures that the stereochemical integrity of the ferrocene backbone is preserved while establishing a new chiral center at the phosphorus atom. The ability to mix and match R1 and R2 groups from a vast array of commercially available halides provides an almost unlimited combinatorial space for ligand optimization.
How to Synthesize Chiral Ferrocene Phosphine Ligands Efficiently
Implementing this synthesis route requires careful attention to thermal management and stoichiometry, particularly during the lithiation and phosphorylation stages. The process begins with the dissolution of Ugi's amine in ethers such as methyl tert-butyl ether or THF, followed by the slow addition of sec-butyllithium to generate the ortho-lithiated species. Once the lithiation is complete, the reaction mixture must be cooled significantly before the introduction of phosphorus trichloride to control the exotherm and prevent decomposition. Following the formation of the dichloride suspension, the choice of substitution strategy depends on whether a symmetric or P-chiral ligand is desired, with the latter requiring precise metering of organometallic reagents. Detailed standardized operating procedures for scaling this chemistry from gram to kilogram levels are essential to ensure reproducibility and safety.
- Activate Ugi's amine via ortho-lithiation using organolithium reagents like s-BuLi at controlled temperatures between -15°C and 35°C.
- React the lithiated intermediate with phosphorus trichloride (PCl3) at cryogenic conditions (-120°C to -60°C) to form the dichloride species.
- Perform nucleophilic substitution using organolithium or Grignard reagents, either in excess for symmetric ligands or stepwise for P-chiral variants.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology offers substantial benefits by decoupling ligand production from the volatile market of specialized phosphine chlorides. By relying on phosphorus trichloride and common organometallic reagents, manufacturers can leverage existing bulk purchasing agreements for commodity chemicals, leading to significant cost reduction in pharmaceutical intermediate manufacturing. The simplification of the raw material portfolio reduces the administrative burden of managing numerous niche suppliers and mitigates the risk of supply disruptions caused by the discontinuation of specific reagents. Furthermore, the modular nature of the synthesis allows for rapid adaptation to changing market demands, enabling the production of custom ligand variants without the need for extensive retooling or new supplier qualification processes.
- Cost Reduction in Manufacturing: The elimination of expensive, pre-functionalized phosphine chlorides from the bill of materials directly lowers the variable cost per kilogram of the final ligand. Since PCl3 and simple aryl or alkyl halides are produced on a massive industrial scale, their prices are stable and predictable compared to fine chemical intermediates. Additionally, the high atom economy of the substitution reactions minimizes waste disposal costs, contributing to a leaner and more economically efficient production process. The ability to synthesize a wide range of ligands from a single common intermediate further amortizes the fixed costs of production across multiple product SKUs.
- Enhanced Supply Chain Reliability: Utilizing commodity-grade starting materials ensures a robust and resilient supply chain that is less susceptible to geopolitical or logistical shocks. Unlike specialized reagents that may have single-source suppliers with long lead times, the key inputs for this process are available from multiple global vendors, ensuring continuity of supply. This reliability is crucial for maintaining consistent production schedules for downstream API manufacturing, where delays in catalyst availability can have cascading effects on drug launch timelines. The simplified logistics also reduce the complexity of inventory management and quality control testing for incoming raw materials.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are compatible with standard stainless steel reactors equipped with low-temperature cooling capabilities, facilitating straightforward scale-up from pilot plant to commercial production. The use of common solvents like ethers and hydrocarbons simplifies solvent recovery and recycling operations, aligning with modern green chemistry principles and environmental regulations. By avoiding the use of heavy metal catalysts or toxic reagents in the ligand synthesis itself, the process generates a cleaner waste stream that is easier and cheaper to treat. This environmental profile supports sustainability goals and reduces the regulatory burden associated with hazardous chemical handling.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral ligand synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is vital for R&D teams evaluating the feasibility of adopting this route for their specific catalytic applications. It is important to note that while the general method is robust, specific reaction parameters may require optimization depending on the steric and electronic nature of the chosen substituents.
Q: What are the advantages of this PCl3-based route over traditional methods?
A: Traditional methods often rely on pre-formed diarylphosphine chlorides which are expensive and structurally limited. This patent utilizes cheap PCl3 and versatile organometallic reagents, allowing for infinite structural tuning.
Q: Can this method produce P-chiral ligands specifically?
A: Yes, by employing a stepwise substitution strategy where different organometallic reagents are added sequentially to the dichloride intermediate, distinct P-chiral centers can be established with high stereocontrol.
Q: Is this process suitable for large-scale industrial production?
A: The process uses standard low-temperature reactor capabilities and commodity chemicals like PCl3 and Grignard reagents, making it highly amenable to commercial scale-up without requiring exotic catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Ferrocene Phosphine Ligand Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging deep expertise in organometallic chemistry to deliver high-performance chiral ligands for the global pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of ligand meets the exacting standards required for asymmetric catalysis. Our commitment to quality and consistency makes us a trusted partner for companies seeking to secure their supply of critical catalytic materials.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into the potential economic benefits of switching to this PCl3-based methodology for your ligand requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will empower you to make informed decisions about your catalyst sourcing strategy. Let us help you optimize your supply chain and accelerate your drug development timeline with our superior chemical solutions.
