Advanced Synthesis of 1,1,2,2-Tetra[4-(1H-1,2,4-triazolyl-1-yl)phenyl]ethylene for Photoelectric Applications
The rapid evolution of the organic light-emitting diode (OLED) industry demands novel materials that combine high quantum efficiency with manufacturability. Patent CN103772304A introduces a groundbreaking approach to synthesizing 1,1,2,2-tetra[4-(1H-1,2,4-triazolyl-1-yl)phenyl]ethylene, a sophisticated tetraphenyl ethylene derivative designed specifically for photoelectric applications. This technology represents a significant leap forward by utilizing a robust copper-catalyzed coupling strategy that bypasses the traditional reliance on expensive palladium systems. For R&D directors and procurement specialists alike, this patent offers a compelling value proposition: a streamlined, one-pot synthetic route that delivers high-purity intermediates essential for next-generation display technologies. The structural complexity of the target molecule, featuring four triazole moieties attached to a central ethylene core, presents unique challenges in regioselectivity and purification, which this invention addresses through precise stoichiometric control and thermal management.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of C-N bonds in complex poly-heterocyclic systems like tetraphenyl ethylene derivatives has relied heavily on Buchwald-Hartwig amination protocols. These conventional methods typically necessitate the use of sophisticated ligand systems and precious metal catalysts such as palladium, which not only inflate the raw material costs but also introduce significant supply chain vulnerabilities due to the fluctuating market prices of noble metals. Furthermore, the removal of trace palladium residues from the final product is a notoriously difficult and costly downstream processing step, especially when targeting the ultra-high purity standards required for electronic materials. Multi-step syntheses often suffer from cumulative yield losses and generate substantial amounts of hazardous waste, creating environmental compliance burdens that can delay time-to-market for new photoelectric devices. The rigidity of these traditional pathways often limits the ability to rapidly scale production to meet the surging global demand for OLED components.
The Novel Approach
In stark contrast, the methodology disclosed in CN103772304A employs a modified Ullmann-type coupling reaction that utilizes earth-abundant copper oxide as the catalytic driver. This innovative route allows for the direct condensation of 1,1,2,2-tetra(4-bromophenyl)ethylene with triazole in a single reaction vessel, dramatically simplifying the operational workflow. By optimizing the molar ratios of the substrate, heterocycle, base, and catalyst, the inventors have achieved a process that operates effectively within a broad temperature window of 80°C to 200°C. The use of dimethylformamide (DMF) as a polar solvent facilitates the solubility of the bulky tetraphenyl ethylene precursor while promoting the catalytic cycle. This approach not only mitigates the cost pressures associated with precious metals but also simplifies the workup procedure to a straightforward filtration and water precipitation, thereby enhancing the overall process mass intensity and reducing the environmental footprint of the manufacturing process.
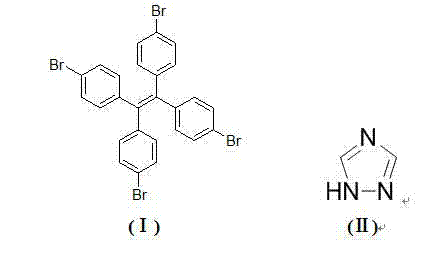
Mechanistic Insights into Cu-Catalyzed C-N Coupling
The core of this technological advancement lies in the efficient formation of four distinct C-N bonds between the phenyl rings and the triazole nitrogen atoms. The mechanism likely proceeds through a copper-mediated oxidative addition and reductive elimination cycle, where the copper oxide species activates the aryl bromide bonds on the tetraphenyl ethylene core. The presence of potassium carbonate serves a dual purpose: it acts as a base to deprotonate the triazole, generating the nucleophilic triazolyl anion, and it helps to neutralize the hydrogen bromide byproduct formed during the coupling. The specific stoichiometry identified in the patent, particularly the excess of triazole and base relative to the substrate, drives the equilibrium towards the fully substituted tetra-product, minimizing the formation of partially substituted intermediates which would act as difficult-to-remove impurities. This precise control over the reaction kinetics is crucial for ensuring the structural integrity and photophysical consistency of the final material.
![Chemical structure of 1,1,2,2-tetra[4-(1H-1,2,4-triazolyl-1-yl)phenyl]ethylene](/insights/img/tetraphenyl-ethylene-triazole-oled-supplier-20260304155640-01.png)
From an impurity control perspective, the choice of copper oxide over other copper salts appears to offer a cleaner reaction profile with fewer side reactions. The symmetrical nature of the starting material, 1,1,2,2-tetra(4-bromophenyl)ethylene, combined with the high reactivity of the copper catalyst system, ensures that the substitution occurs uniformly across all four positions of the molecule. This uniformity is vital for the electronic properties of the material, as asymmetric substitution could lead to unpredictable shifts in emission wavelengths or reduced charge transport mobility. The patent data indicates that the resulting product possesses a sharp melting point and consistent elemental analysis, confirming the high degree of chemical purity achievable through this route. For quality assurance teams, this implies a more robust specification sheet with tighter tolerances on impurity profiles, reducing the risk of batch rejection during the stringent QC testing phases typical of the electronic chemicals sector.
How to Synthesize 1,1,2,2-Tetra[4-(1H-1,2,4-triazolyl-1-yl)phenyl]ethylene Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing the importance of maintaining specific molar ratios to maximize yield. The process begins with the charging of the reactor with the aryl bromide precursor, triazole, potassium carbonate, and copper oxide, followed by the addition of DMF solvent. The mixture is then heated under reflux conditions, allowing the coupling reaction to proceed to completion over a period ranging from 12 to 120 hours depending on the specific temperature setpoint. Upon completion, the reaction mixture is cooled to room temperature, filtered to remove insoluble inorganic salts and catalyst residues, and the filtrate is poured into water to induce precipitation of the organic product. This simple isolation technique avoids the need for column chromatography, which is often a bottleneck in scaling up fine chemical synthesis. For detailed standardized operating procedures and safety guidelines, please refer to the step-by-step synthesis guide below.
- Charge a reactor with 1,1,2,2-tetra(4-bromophenyl)ethylene, triazole, potassium carbonate, and copper oxide in a specific molar ratio of 2: 10:30:1.
- Add DMF as the polar solvent and heat the mixture to a temperature range between 80°C and 200°C under stirring.
- Maintain reaction for 12 to 120 hours, then cool, filter, and precipitate the product by adding water to the filtrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this copper-catalyzed methodology offers profound strategic benefits that extend beyond simple unit price reductions. The elimination of palladium catalysts removes a major cost volatility factor from the bill of materials, allowing for more stable long-term pricing contracts with downstream OLED manufacturers. Additionally, the simplified workup procedure, which relies on basic filtration and precipitation rather than complex extraction or chromatographic purification, significantly reduces the consumption of organic solvents and the associated waste disposal costs. This leaner manufacturing process translates directly into improved margin potential and a more competitive positioning in the global market for specialty electronic chemicals. The robustness of the reaction conditions also suggests a high tolerance for minor variations in raw material quality, further securing the supply chain against upstream disruptions.
- Cost Reduction in Manufacturing: The substitution of expensive precious metal catalysts with inexpensive copper oxide results in a drastic reduction in catalytic costs, while the one-pot nature of the reaction minimizes labor and equipment usage time. By avoiding the need for specialized ligands and complex inert atmosphere techniques often required for palladium chemistry, the overall operational expenditure is significantly lowered. Furthermore, the high atom economy of the direct coupling reaction ensures that a greater proportion of the starting materials are converted into the valuable final product, reducing the effective cost per kilogram of the active pharmaceutical or electronic ingredient.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including 1,1,2,2-tetra(4-bromophenyl)ethylene and triazole, are commodity chemicals with well-established global supply networks, reducing the risk of single-source dependency. The use of common solvents like DMF and standard inorganic bases like potassium carbonate ensures that procurement teams can easily source all necessary inputs from multiple qualified vendors. This diversification of the supply base enhances resilience against geopolitical tensions or logistical bottlenecks, ensuring continuous production capability even during periods of market instability.
- Scalability and Environmental Compliance: The process is explicitly designed for large-scale industrial production, with a reaction profile that is amenable to standard stainless steel reactors used in fine chemical manufacturing. The reduction in hazardous waste generation, particularly the absence of heavy metal contamination in the product stream, simplifies the environmental permitting process and lowers the cost of wastewater treatment. This alignment with green chemistry principles not only satisfies regulatory requirements but also appeals to end-users who are increasingly prioritizing sustainability in their supplier selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this novel tetraphenyl ethylene derivative. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this material into your existing product development pipelines. We encourage technical teams to review these points carefully to assess the compatibility of this synthesis route with their specific quality and throughput requirements.
Q: What is the primary advantage of using Copper Oxide over Palladium catalysts in this synthesis?
A: The use of Copper Oxide (CuO) instead of precious metal catalysts like Palladium significantly reduces raw material costs and eliminates the need for complex heavy metal removal steps, which is critical for electronic grade purity.
Q: What are the optical properties of the synthesized tetraphenyl ethylene derivative?
A: The compound exhibits strong photoelectric properties with an excitation wavelength of 340 nm and an emission wavelength of 522 nm, making it highly suitable for luminescent material applications.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the 'one-pot' nature of the reaction, simple operational procedures, and low environmental pollution, all of which are key indicators for successful commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,1,2,2-Tetra[4-(1H-1,2,4-triazolyl-1-yl)phenyl]ethylene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance photoelectric materials play in the advancement of display technologies and optoelectronic devices. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume demands of the global electronics industry. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to replicate and optimize the copper-catalyzed synthesis described in CN103772304A allows us to offer a cost-effective and reliable supply of this key intermediate without compromising on quality or delivery timelines.
We invite you to collaborate with us to explore how this advanced material can enhance your product portfolio. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities align with your project goals. Let us be your partner in driving innovation and efficiency in the electronic materials sector.
