Advanced Ethylenediamine NHC Precursors for Commercial Chiral Synthesis
The pharmaceutical and fine chemical industries are constantly seeking robust catalytic systems that can deliver high enantioselectivity without compromising on cost or scalability. Patent CN102153557A introduces a significant advancement in this domain by disclosing a class of polychiral center nitrogen heterocyclic carbene (NHC) precursor salts featuring an ethylenediamine skeleton. This technology represents a strategic shift from complex, expensive fused-ring systems to more accessible, modular architectures derived from cheap chiral substituted diamines. For R&D directors and procurement managers, this patent offers a pathway to synthesize high-value chiral benzopyrone compounds through polarity inversion reactions with improved efficiency. The core innovation lies in the structural flexibility of the ethylenediamine backbone, which allows for extensive tuning of the steric and electronic environment around the catalytic center. This adaptability is crucial for optimizing reaction outcomes in complex organic syntheses, ensuring that manufacturers can achieve stringent purity specifications required for active pharmaceutical ingredients. By leveraging this technology, companies can secure a reliable supply chain for critical chiral intermediates while mitigating the risks associated with proprietary catalyst dependency.
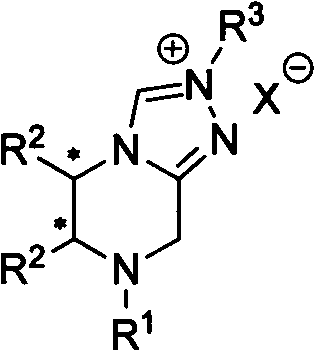
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional nitrogen heterocyclic carbene catalysts have predominantly relied on fused-ring triazolium structures, which, while effective, often present significant challenges in terms of synthetic accessibility and cost. The preparation of these conventional catalysts frequently involves multi-step sequences using expensive starting materials and harsh reaction conditions that are difficult to control on a large scale. Furthermore, the structural rigidity of fused-ring systems limits the ability to fine-tune the chiral environment, often resulting in suboptimal enantioselectivity for specific substrate classes. For procurement managers, this translates to higher raw material costs and longer lead times due to the complexity of the supply chain for these specialized reagents. Additionally, the removal of residual metals or by-products from traditional catalytic systems can add substantial downstream processing costs, impacting the overall economic viability of the manufacturing process. These limitations create a bottleneck for companies aiming to scale up the production of chiral intermediates efficiently.
The Novel Approach
In contrast, the novel approach detailed in CN102153557A utilizes a three-step synthesis starting from commercially cheap and easy-to-obtain optically pure substituted ethylenediamines. This method drastically simplifies the production workflow by avoiding the need for complex fused-ring construction, thereby reducing both time and resource expenditure. The ethylenediamine skeleton provides a robust chiral environment that can be easily modified through the selection of different R groups, allowing for precise optimization of catalytic activity and selectivity. This modularity means that manufacturers can adapt the catalyst to specific reaction requirements without overhauling the entire synthetic route. From a supply chain perspective, the use of readily available diamines ensures a stable and continuous supply of raw materials, reducing the risk of production delays. The mild reaction conditions described in the patent further enhance the safety and environmental profile of the process, aligning with modern green chemistry principles and regulatory compliance standards.

Mechanistic Insights into Ethylenediamine-Based NHC Catalysis
The catalytic efficacy of these precursor salts stems from the unique electronic and steric properties imparted by the ethylenediamine backbone. Upon deprotonation, the precursor generates a free carbene species that acts as a potent nucleophile, facilitating the polarity inversion of aldehyde substrates. The chiral centers located on the ethylenediamine ring exert a strong influence on the approach of the substrate, effectively discriminating between enantiotopic faces to ensure high enantiomeric excess in the final product. This mechanism is particularly effective for the synthesis of chiral benzopyrone compounds, where the stereochemical integrity of the molecule is paramount for its biological activity. The presence of various substituents (R1, R2, R3) allows chemists to modulate the electron density at the carbene carbon, fine-tuning the reactivity to match the specific electronic demands of the aldehyde substrate. Such precise control over the catalytic cycle minimizes side reactions and by-product formation, leading to cleaner reaction profiles and simplified purification processes.
Impurity control is another critical aspect where this technology excels, as the well-defined structure of the catalyst reduces the likelihood of generating hard-to-remove impurities. The patent specifies that by-products can be successfully separated using standard techniques like column chromatography or recrystallization, indicating a high level of process robustness. For quality control teams, this means that achieving stringent purity specifications is more attainable, reducing the risk of batch rejection. The stability of the precursor salts also contributes to consistent performance over time, ensuring that the catalytic activity remains stable throughout the reaction duration. This reliability is essential for maintaining product quality in commercial manufacturing, where batch-to-batch consistency is a key requirement for regulatory approval. By understanding these mechanistic details, R&D teams can better leverage this technology to develop scalable and efficient synthetic routes for high-value intermediates.
How to Synthesize Chiral NHC Precursor Salts Efficiently
The synthesis of these valuable catalyst precursors follows a logical and scalable three-step sequence that begins with the cyclization of a chiral diamine. This initial step establishes the core ethylenediamine skeleton, setting the stereochemical foundation for the entire molecule. Subsequent functionalization of the nitrogen atom allows for the introduction of diverse protecting groups, which play a crucial role in modulating the solubility and reactivity of the intermediate. The final ring-closing step involves condensation with hydrazine and an orthoformate, a transformation that is both high-yielding and operationally simple. Detailed standardized synthetic steps see the guide below.
- Cyclization of chiral substituted diamine with ethyl bromoacetate to form the lactam skeleton.
- Protection of the lactam nitrogen using sulfonyl or acyl chlorides to establish the R1 group.
- Condensation with hydrazine and orthoformate under reflux to close the triazolium ring and form the salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain leaders, the adoption of this ethylenediamine-based NHC technology offers substantial strategic benefits that extend beyond mere technical performance. The primary advantage lies in the significant cost reduction potential driven by the use of cheap and readily available starting materials. Unlike traditional catalysts that rely on scarce or expensive chiral pools, this method utilizes substituted ethylenediamines that are produced in large volumes for various industrial applications. This abundance ensures a stable pricing structure and mitigates the volatility often associated with specialized chemical reagents. Furthermore, the simplified three-step synthesis reduces the overall manufacturing footprint, lowering energy consumption and waste generation. These efficiencies translate directly into lower production costs, allowing companies to improve their profit margins or offer more competitive pricing to their customers without sacrificing quality.
- Cost Reduction in Manufacturing: The elimination of complex fused-ring synthesis steps significantly lowers the operational expenditure associated with catalyst production. By avoiding the need for specialized reagents and harsh conditions, manufacturers can utilize standard equipment and reduce utility costs. The high yields reported in the patent examples suggest that raw material utilization is efficient, minimizing waste and maximizing output per batch. Additionally, the ease of purification reduces the consumption of solvents and chromatography media, further driving down the cost of goods sold. This economic efficiency makes the technology highly attractive for large-scale commercial applications where cost competitiveness is a decisive factor.
- Enhanced Supply Chain Reliability: Sourcing raw materials from established suppliers of chiral diamines ensures a robust and resilient supply chain. These materials are not subject to the same geopolitical or production bottlenecks as exotic catalysts, providing greater security of supply. The modular nature of the synthesis also allows for flexibility in sourcing; if one specific diamine is unavailable, alternatives with similar steric properties can often be substituted with minimal process adjustment. This adaptability reduces the risk of production stoppages due to raw material shortages. For supply chain heads, this reliability is crucial for maintaining continuous operations and meeting delivery commitments to downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The reaction conditions described, ranging from room temperature to moderate reflux, are inherently safer and easier to scale than processes requiring high pressure or cryogenic temperatures. This safety profile reduces the need for specialized containment systems and lowers insurance and compliance costs. Moreover, the use of common organic solvents facilitates solvent recovery and recycling, aligning with sustainability goals and environmental regulations. The ability to scale from gram to kilogram quantities without significant process re-engineering accelerates the time to market for new products. This scalability ensures that the technology can grow with the business, supporting increased demand without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this NHC precursor technology. These answers are derived directly from the patent specifications and are intended to clarify the operational benefits and application scope for potential adopters. Understanding these details is essential for making informed decisions about integrating this catalytic system into existing manufacturing workflows.
Q: What are the primary advantages of ethylenediamine-based NHC precursors?
A: These precursors offer a versatile chiral environment derived from cheap, commercially available diamines, providing excellent enantioselectivity in polarity inversion reactions compared to traditional fused-ring triazolium catalysts.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the patent describes mild reaction conditions (0°C to 150°C) and uses common organic solvents, making the three-step process highly suitable for commercial scale-up without requiring extreme pressure or specialized equipment.
Q: What specific applications do these catalysts support?
A: They are specifically designed for catalyzing the polarity inversion of aldehyde compounds, enabling the efficient preparation of chiral benzopyrone compounds which are valuable scaffolds in pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral NHC Precursor Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced catalytic technologies play in driving innovation within the pharmaceutical and fine chemical sectors. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to plant is seamless and efficient. We are committed to delivering high-purity Chiral NHC Precursor Salts that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to quality ensures that every batch we supply performs consistently, enabling you to maintain the highest standards in your own production processes.
We invite you to collaborate with us to unlock the full potential of this ethylenediamine-based catalytic system for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your production volume and requirements. Please contact us to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain for greater efficiency and profitability.
