Advanced Pd-Catalyzed Selective Alkenylation for High-Value Pharmaceutical Intermediate Manufacturing
Advanced Pd-Catalyzed Selective Alkenylation for High-Value Pharmaceutical Intermediate Manufacturing
The landscape of organic synthesis for high-value pharmaceutical intermediates is constantly evolving, driven by the need for more efficient, selective, and environmentally benign methodologies. A significant breakthrough in this domain is documented in Chinese Patent CN110642752A, which details a novel synthetic method for the alkenylation of allyl alcohol carbamate compounds. This technology addresses a long-standing challenge in the field: the selective activation and functionalization of vinylic carbon-hydrogen bonds in allylic systems. By leveraging a palladium-catalyzed system with a carbamate directing group, this invention enables the precise construction of conjugated diene derivatives with exceptional stereocontrol. For R&D directors and process chemists, this represents a pivotal shift from traditional, less selective methods to a robust, directing-group-mediated approach that can be seamlessly integrated into the manufacturing of complex active pharmaceutical ingredients (APIs) and natural product analogs.
The significance of this patent extends beyond mere academic interest; it offers a tangible solution for the commercial scale-up of complex pharmaceutical intermediates. The method utilizes readily available starting materials, such as allyl alcohols and cyclohexen-3-ols, which are abundant and cost-effective feedstocks in the fine chemical industry. The ability to transform these simple precursors into highly functionalized alkenyl products with yields ranging from 45% to 98% underscores the efficiency and reliability of the process. Furthermore, the reaction conditions are remarkably mild, typically operating at 80°C in trifluoroethanol, which reduces energy consumption and minimizes the thermal degradation of sensitive functional groups often present in drug candidates. This aligns perfectly with the industry's push towards greener chemistry and sustainable manufacturing practices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct functionalization of allyl alcohols has been fraught with challenges regarding regioselectivity and chemoselectivity. Traditional methods often rely on harsh acidic conditions or stoichiometric amounts of toxic reagents, which can lead to undesirable side reactions such as polymerization, isomerization, or the formation of complex mixtures of regioisomers. For instance, previous attempts at allylic oxidation or alkenylation frequently struggled to distinguish between the multiple reactive sites on the allylic system, particularly when the double bond was internal rather than terminal. This lack of precision necessitates extensive purification steps, driving up production costs and reducing overall throughput. Moreover, many conventional catalytic systems require expensive and air-sensitive ligands or high temperatures that are incompatible with thermally labile substrates, limiting their utility in the synthesis of complex drug molecules.
The Novel Approach
In stark contrast, the methodology disclosed in CN110642752A introduces a paradigm shift by utilizing the carbamate moiety not just as a protecting group, but as a strategic directing group for C-H activation. This approach allows for the selective cleavage of the vinylic C-H bond at the same carbon position as the oxygen atom, a transformation that was previously difficult to achieve with high fidelity. The reaction employs a palladium catalytic system supported by an amino acid ligand, such as N-acetylglycine, and silver carbonate as a terminal oxidant. This combination facilitates a smooth catalytic cycle that tolerates a wide range of functional groups. As illustrated in the general reaction scheme below, this method is versatile enough to accommodate both acyclic allyl alcohols and cyclic cyclohexen-3-ol derivatives, producing conjugated diene structures with defined Z,E configurations.
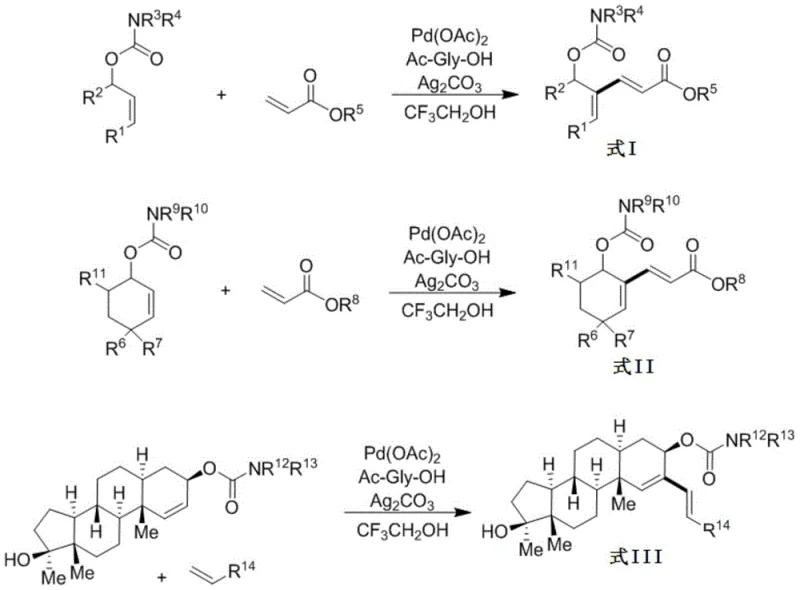
The versatility of this novel approach is further highlighted by its compatibility with various electron-deficient olefins, including acrylates, alkenyl sulfones, and alkenyl phosphonates. This broad substrate scope means that procurement managers can source diverse coupling partners without needing to redesign the core synthetic route for each new target molecule. The use of trifluoroethanol as a solvent also enhances the solubility of polar intermediates and stabilizes the cationic palladium species, contributing to the high yields observed across different substrate classes. By overcoming the regioselectivity hurdles of the past, this technology provides a reliable pathway for the cost reduction in pharmaceutical intermediate manufacturing, allowing companies to produce high-purity building blocks with fewer impurities and reduced waste generation.
Mechanistic Insights into Pd-Catalyzed C-H Alkenylation
To fully appreciate the robustness of this synthetic method, one must delve into the mechanistic underpinnings that drive its high selectivity and efficiency. The reaction initiates with the coordination of the carbamate oxygen to the palladium(II) center, forming a pre-reactive complex. This coordination is the key step that directs the metal to the specific vinylic C-H bond adjacent to the carbamate group. Following this coordination, the palladium species undergoes oxidative addition into the C-H bond, generating a six-membered palladacycle transition state. This cyclic intermediate is crucial as it locks the conformation of the substrate, ensuring that the subsequent steps occur with high stereochemical control. The formation of this stable metallacycle explains why the reaction is so selective for the homobenzylic or vinylic position over other potential sites of activation on the molecule.
Once the palladacycle is formed, the electron-deficient olefin coordinates to the palladium center and inserts into the Pd-C bond. This migratory insertion step extends the carbon skeleton and sets the stage for the final product formation. The cycle is completed by a beta-hydride elimination, which releases the alkenylated product and generates a palladium-hydride species. Finally, the silver carbonate oxidant plays a dual role: it reoxidizes the reduced palladium(0) species back to the active palladium(II) state, allowing the catalytic cycle to continue, and it acts as a base to neutralize the acid generated during the process. The efficacy of this mechanism on complex scaffolds is demonstrated in the patent's examples involving steroid derivatives, where the catalyst successfully navigates the steric bulk of the polycyclic framework to deliver the desired functionalization.
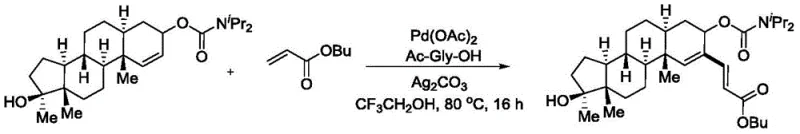
Understanding this mechanism is vital for R&D teams aiming to optimize the process for specific targets. The reliance on a six-membered transition state implies that substrates capable of forming such rings will react most efficiently. Furthermore, the choice of the amino acid ligand, specifically N-acetylglycine, is not arbitrary; it likely assists in stabilizing the palladium center and facilitating the C-H activation step through hydrogen bonding or secondary coordination interactions. This level of mechanistic detail assures quality control teams that the process is well-understood and controllable, minimizing the risk of batch-to-batch variability. The ability to predict the outcome based on the electronic and steric properties of the substrates allows for rational design of experiments, accelerating the development timeline for new drug candidates.
How to Synthesize Alkenylated Allyl Alcohol Carbamates Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to ensure optimal results. The patent outlines a straightforward procedure that begins with the preparation of the catalytic system in an oven-dried vessel under an inert atmosphere, typically argon or nitrogen, to prevent catalyst deactivation by oxygen. The precise stoichiometry of the reagents is critical, with the patent recommending a molar ratio of carbamate to olefin of approximately 1:2, and catalyst loading around 10 mol%. The reaction is heated to 80°C, a temperature that balances reaction rate with safety and energy efficiency. Detailed standardized synthesis steps for scaling this reaction from gram to kilogram scale are provided in the guide below.
- Prepare the reaction mixture by combining palladium acetate catalyst, N-acetylglycine ligand, and silver carbonate oxidant in trifluoroethanol solvent under inert atmosphere.
- Add the allyl alcohol carbamate substrate and the electron-deficient olefin coupling partner to the reaction vessel in the specified molar ratios.
- Heat the sealed reaction mixture to 80°C for 16 hours, then filter, concentrate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic advantages that go beyond simple yield metrics. The primary benefit lies in the simplification of the supply chain for raw materials. Since the method utilizes allyl alcohols and cyclohexen-3-ols, which are commodity chemicals produced on a massive scale globally, there is minimal risk of supply disruption or price volatility associated with exotic starting materials. Additionally, the catalyst system relies on palladium acetate and silver carbonate, which, while precious metals, are used in catalytic quantities and can potentially be recovered and recycled, further mitigating cost concerns. The operational simplicity of the reaction—requiring only heating and stirring without the need for specialized high-pressure equipment—lowers the barrier to entry for contract manufacturing organizations (CMOs).
- Cost Reduction in Manufacturing: The economic viability of this process is driven by its high atom economy and the elimination of multi-step protection-deprotection sequences that are common in traditional synthesis. By using the carbamate as a traceless directing group that can be easily transformed or retained, the overall step count is reduced. Fewer steps mean less solvent consumption, lower labor costs, and reduced waste disposal fees. Although specific percentage savings depend on the specific target molecule, the qualitative reduction in process complexity invariably leads to a lower cost of goods sold (COGS). The high yields reported, reaching up to 98% in optimized cases, ensure that raw material utilization is maximized, minimizing the financial loss associated with unreacted starting materials.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply chain reliability. The tolerance of the method to various functional groups means that the same protocol can be applied to a library of analogs without extensive re-optimization. This flexibility allows manufacturers to respond quickly to changes in demand for different API intermediates. Furthermore, the use of stable reagents like silver carbonate and amino acid ligands ensures that the supply of critical reaction components is secure, as these are standard catalog items available from multiple global suppliers. This redundancy in the supply base protects against single-source risks and ensures continuous production capabilities even during market fluctuations.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method offers a cleaner alternative to older technologies. The avoidance of harsh acids or toxic stoichiometric oxidants reduces the generation of hazardous waste streams, simplifying compliance with increasingly stringent environmental regulations. The reaction produces water and reduced silver salts as byproducts, which are easier to manage than heavy metal sludge from other processes. Scalability is also a key strength; the patent demonstrates success on substrates ranging from simple chains to complex steroids, indicating that the chemistry holds up well as molecular weight and complexity increase. This makes it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates, ensuring a smooth transition from lab bench to industrial reactor.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific applications, we have compiled a set of frequently asked questions based on the detailed technical disclosures within the patent documentation. These answers address common concerns regarding substrate compatibility, reaction scalability, and product purity. Understanding these nuances is essential for integrating this synthetic route into existing manufacturing workflows and for assessing its fit for specific project requirements. The following insights are derived directly from the experimental data and embodiments provided in the source intellectual property.
Q: What is the primary advantage of using carbamate as a directing group in this alkenylation reaction?
A: The carbamate group acts as a powerful directing group that coordinates with the palladium catalyst, enabling highly selective activation of the vinylic C-H bond at the same carbon position, which overcomes the regioselectivity issues common in traditional allylic functionalization.
Q: Can this synthesis method be applied to complex steroid skeletons?
A: Yes, the patent explicitly demonstrates the successful application of this method on complex steroid derivatives, such as methyl testosterone analogs, achieving high yields and maintaining stereochemical integrity, making it ideal for late-stage functionalization of bioactive molecules.
Q: What are the typical reaction conditions required for this transformation?
A: The reaction typically proceeds under mild heating conditions at 80°C using trifluoroethanol as the solvent, with palladium acetate as the catalyst, an amino acid derivative as the ligand, and silver carbonate as the oxidant, ensuring operational simplicity and safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Allyl Alcohol Carbamates Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies like the one described in CN110642752A for the next generation of pharmaceutical manufacturing. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale discoveries are translated into robust industrial processes. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs dedicated to verifying the identity and purity of every batch of high-purity pharmaceutical intermediates we produce. We understand that consistency is key in the supply of critical drug substances, and our infrastructure is designed to meet the exacting standards of global regulatory bodies.
We invite you to collaborate with us to leverage this cutting-edge alkenylation technology for your pipeline projects. Whether you require custom synthesis of complex steroid derivatives or large-scale production of conjugated diene intermediates, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to not just a supplier, but a strategic partner dedicated to optimizing your supply chain and accelerating your time to market with reliable, high-quality chemical solutions.
