Industrial Scale Synthesis of Cis-Capsaicin via Mild Wittig Reaction Route
Industrial Scale Synthesis of Cis-Capsaicin via Mild Wittig Reaction Route
The global demand for high-purity capsaicin and its analogs continues to surge across the pharmaceutical, agrochemical, and flavor industries, driven by their potent biological activities and sensory properties. Patent CN1865234A introduces a groundbreaking chemical synthesis and purification method that addresses the critical limitations of traditional extraction techniques. This technology enables the production of specific cis-isomers of capsaicin through a robust five-step sequence, starting from readily available commodity chemicals like vanillin and isobutyraldehyde. By shifting from agricultural dependency to precise organic synthesis, manufacturers can achieve unprecedented control over stereochemistry and impurity profiles. This report analyzes the technical merits of this Wittig-based approach, highlighting its potential to redefine supply chain reliability and cost structures for reliable flavor & fragrance intermediates supplier networks globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the procurement of capsaicin has relied heavily on the extraction from Capsicum fruits, a process fraught with significant inefficiencies and variability. Natural capsaicin exists as a complex mixture of alkaloids, including dihydrocapsaicin, nordihydrocapsaicin, and homocapsaicin, with the target compound often comprising only a fraction of the total extract. The content of capsaicinoids in pepper fruits is notoriously low, typically ranging from 0.1% to 0.4%, necessitating the processing of massive volumes of biomass to obtain commercially viable quantities. Furthermore, the composition of the extract is highly susceptible to variations in pepper variety, harvesting time, and storage conditions, leading to inconsistent batch quality. Separating high-purity cis-capsaicin from this complex matrix is technically challenging and economically burdensome, often requiring multiple rounds of chromatography that drastically reduce overall yield. These inherent bottlenecks make natural extraction unsuitable for applications demanding strict stereochemical purity and consistent supply continuity.
The Novel Approach
The synthetic methodology disclosed in CN1865234A offers a transformative alternative by constructing the capsaicin skeleton through rational organic synthesis rather than isolation. The core innovation lies in the use of a Wittig reaction to establish the crucial carbon-carbon double bond with high stereoselectivity, specifically favoring the formation of the cis-isomer which is difficult to obtain via other synthetic routes. By reacting vanillylamine with a specifically designed acyl chloride derived from isobutyraldehyde, the process bypasses the impurity issues associated with natural sources entirely. This approach allows for the modular synthesis of various capsaicin analogs by simply varying the acylating agent, providing a versatile platform for developing new bioactive compounds. The reaction conditions are notably mild, utilizing common solvents and reagents that are compatible with large-scale industrial reactors, thereby facilitating a seamless transition from laboratory discovery to commercial manufacturing without the need for exotic catalysts or extreme pressures.
Mechanistic Insights into Wittig Olefination and Acylation
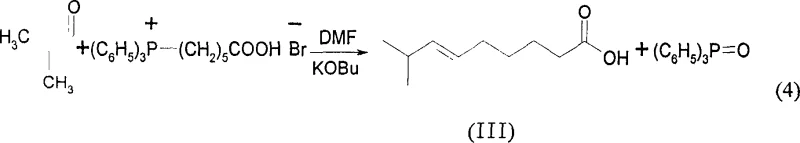
The heart of this synthetic strategy is the Wittig olefination step, which constructs the unsaturated fatty acid chain with precise geometric control. In this critical transformation, (6-carboxyhexyl)triphenylphosphine bromide serves as the phosphonium salt precursor, which is deprotonated by a strong base, preferably potassium tert-butoxide, in an aprotic solvent such as DMF. The resulting phosphorus ylide reacts with isobutyraldehyde to form the alkene linkage. The choice of base and solvent is paramount; the patent specifies that using potassium tert-butoxide in DMF at room temperature promotes the formation of the cis-alkene while minimizing side reactions. The mechanism proceeds through a four-membered oxaphosphetane intermediate, which collapses to release triphenylphosphine oxide and the desired cis-8-methyl-6-nonenoic acid. This step is particularly elegant because it avoids the use of heavy metal catalysts often required for selective hydrogenation of alkynes, thus eliminating the risk of toxic metal residues in the final product.
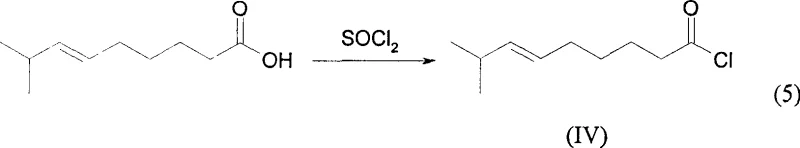
Following the formation of the carboxylic acid, the pathway requires activation to enable the final amide bond formation with vanillylamine. The patent details the conversion of cis-8-methyl-6-nonenoic acid into its corresponding acid chloride using thionyl chloride (SOCl2). This activation step is chosen for its efficiency and the ease of removing excess reagent; thionyl chloride decomposes into gaseous by-products (SO2 and HCl) upon heating, which can be easily vented or scrubbed, driving the reaction to completion without leaving non-volatile residues. The resulting acid chloride is highly reactive towards nucleophilic attack by the amine group of vanillylamine. The final acylation is conducted in anhydrous ether under reflux, ensuring that the moisture-sensitive acid chloride reacts exclusively with the amine to form the target vanillyl amide. This sequence ensures that the sensitive phenolic hydroxyl group on the vanillyl ring remains intact, preserving the biological activity of the molecule.
How to Synthesize Cis-Capsaicin Efficiently
The implementation of this synthesis route requires careful attention to stoichiometry and purification protocols to maximize yield and purity. The process begins with the preparation of vanillylamine via the Leuckart reaction, followed by the synthesis of the phosphonium salt. The subsequent Wittig reaction must be performed under strictly anhydrous conditions to prevent ylide decomposition. Detailed standard operating procedures for each reaction step, including specific temperatures, molar ratios, and workup techniques, are essential for reproducibility. For comprehensive technical guidance on executing this synthesis, please refer to the standardized protocol below.
- Synthesize vanillylamine from vanillin and ammonium formate via Leuckart reaction.
- Prepare (6-carboxyhexyl)triphenylphosphine bromide from 6-bromohexanoic acid and triphenylphosphine.
- Perform Wittig reaction between the phosphonium salt and isobutyraldehyde using potassium tert-butoxide in DMF to form cis-8-methyl-6-nonenoic acid.
- Activate the carboxylic acid using thionyl chloride to generate the corresponding acid chloride intermediate.
- React the acid chloride with vanillylamine in anhydrous ether to form cis-capsaicin, followed by purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from extraction to the synthetic route described in CN1865234A represents a strategic opportunity to mitigate risk and optimize costs. The reliance on agricultural raw materials introduces volatility due to weather patterns, crop diseases, and geopolitical factors affecting spice-producing regions. By adopting this chemical synthesis method, companies can secure a supply of capsaicin that is independent of harvest seasons and geographic limitations. The raw materials, such as vanillin, isobutyraldehyde, and triphenylphosphine, are commodity chemicals produced on a multi-ton scale globally, ensuring a stable and competitive pricing structure. This decoupling from agriculture significantly enhances supply chain resilience, allowing for consistent production schedules and reliable delivery timelines regardless of external environmental factors.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive and low-yield extraction processes that require processing tons of biomass to recover small amounts of product. By utilizing high-efficiency reactions like the Wittig olefination and acylation, the overall material throughput is significantly improved. The removal of transition metal catalysts from the process flow means that costly steps for heavy metal scavenging and validation are no longer required, further reducing operational expenses. Additionally, the ability to recycle solvents like DMF and ether in a closed-loop system contributes to substantial long-term cost savings in utility and waste management.
- Enhanced Supply Chain Reliability: One of the most critical advantages of this technology is the stabilization of lead times. Natural capsaicin supplies are often subject to long lead times dictated by the annual harvest cycle of peppers. In contrast, the chemical synthesis described here can be operated continuously throughout the year, provided that basic chemical feedstocks are available. This continuous manufacturing capability allows suppliers to maintain safety stock levels more effectively and respond rapidly to sudden spikes in market demand. It effectively transforms capsaicin from a seasonal agricultural commodity into a predictable industrial chemical, greatly simplifying inventory planning for downstream users.
- Scalability and Environmental Compliance: The reaction conditions specified in the patent are inherently scalable, utilizing standard glass-lined or stainless steel reactors common in fine chemical plants. The use of thionyl chloride for activation generates gaseous by-products that are easily managed with standard scrubbing systems, avoiding the generation of large volumes of liquid acidic waste associated with other activation methods. Furthermore, the purification steps involving column chromatography and recrystallization are well-understood unit operations that can be adapted for multi-kilogram or ton-scale production. This scalability ensures that the process meets the rigorous environmental and safety standards required for modern chemical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of cis-capsaicin based on the patented technology. These insights are derived directly from the experimental data and background analysis provided in the patent documentation, offering clarity on purity, scalability, and regulatory considerations. Understanding these details is crucial for R&D teams evaluating this route for potential integration into their existing manufacturing portfolios.
Q: Why is chemical synthesis preferred over natural extraction for high-purity capsaicin?
A: Natural extraction yields a complex mixture of capsaicinoids with low content (0.1%-0.4%), making high-purity isolation difficult and costly. Chemical synthesis allows for the specific production of the cis-isomer with controlled purity and eliminates seasonal supply dependencies.
Q: What are the key advantages of the Wittig reaction conditions described in CN1865234A?
A: The patent utilizes mild reaction conditions, specifically using potassium tert-butoxide in DMF at room temperature for the Wittig step. This avoids harsh thermal conditions, reduces energy consumption, and simplifies the removal of by-products like triphenylphosphine oxide.
Q: How does this synthetic route impact supply chain stability for flavor manufacturers?
A: By relying on commodity chemicals like vanillin and isobutyraldehyde rather than agricultural crops, this route decouples production from harvest cycles and weather events. This ensures consistent availability and stable pricing for large-scale flavor and fragrance manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cis-Capsaicin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of next-generation flavor and pharmaceutical products. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN1865234A can be translated into robust industrial processes. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify the stereochemical integrity of every batch. Our commitment to quality assurance means that clients receive cis-capsaicin that meets the highest standards for consistency and performance, free from the impurities commonly found in natural extracts.
We invite global partners to collaborate with us to leverage this advanced synthetic technology for their specific applications. Whether you require custom synthesis of capsaicin analogs or large-scale supply of the standard cis-isomer, our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
