Advanced Synthesis of Chiral Ugi's Amine for Scalable Ferrocene Ligand Production
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for the production of high-purity chiral intermediates, particularly those serving as precursors for advanced catalytic ligands. Patent CN107286203B, published on May 22, 2020, introduces a groundbreaking preparation method for chiral Ugi's amine and its derivatives, which are critical building blocks for Josiphos ferrocene diphosphine ligands. This technology addresses the longstanding challenges in asymmetric catalysis by providing a route that bypasses traditional resolution steps, thereby enhancing overall process efficiency. The invention utilizes chiral amines and ferrocene derivatives as initial raw materials, employing a sequence of reductive amination, alkylation, and substitution reactions to achieve high optical purity. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for metal-catalyzed asymmetric reactions, ensuring a reliable chiral Ugi's amine supplier for complex API intermediate manufacturing.
The demand for enantiomerically pure ferrocene compounds has surged due to their indispensable role in homogeneous catalysis within the pharmaceutical sector. Traditional methods often rely on the resolution of racemates via tartaric acid or enzymatic catalysis, which can be costly and limited in scale. The novel approach detailed in this patent leverages a chiral pool strategy, introducing the chiral center at the beginning of the synthesis using commercially available chiral amines. This strategic shift not only simplifies the purification process but also drastically improves the atom economy of the overall transformation. By eliminating the need for expensive chiral catalysts in the kilogram preparation stage, the method offers substantial cost savings and operational simplicity, making it an attractive option for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of chiral Ugi's amine has been fraught with inefficiencies, primarily stemming from the reliance on resolution techniques to separate enantiomers from racemic mixtures. Conventional processes often involve the resolution of racemic 1-ferrocenyl ethanol using (R)-(+)-tartaric acid, a method that inherently caps the maximum theoretical yield at 50% and requires cumbersome recrystallization steps to achieve high optical purity. Furthermore, enzymatic catalysis methods, while selective, often suffer from limitations in substrate scope and the high cost of biocatalysts, making them less viable for large-scale industrial applications. These traditional pathways frequently encounter issues with chiral purity consistency and low synthesis efficiency, creating bottlenecks in the supply chain for downstream ligand production. The need for multiple purification stages increases solvent consumption and waste generation, negatively impacting the environmental profile and overall manufacturing cost of the final chiral ferrocene derivatives.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes a direct synthetic route that constructs the chiral framework from readily available chiral amines and ferrocene ketones. This method employs a Lewis acid-catalyzed condensation followed by reduction, effectively transferring the chirality from the amine to the ferrocene backbone with high fidelity. By avoiding the resolution step entirely, the process achieves significantly higher overall yields and simplifies the operational workflow, reducing the number of unit operations required. The reaction conditions are mild, typically operating between 25 °C and 45 °C, which minimizes energy consumption and reduces the risk of thermal degradation of sensitive intermediates. This streamlined methodology not only enhances the economic feasibility of producing chiral Ugi's amine but also ensures a more consistent quality profile, which is paramount for reliable agrochemical intermediate and pharmaceutical supplier operations.
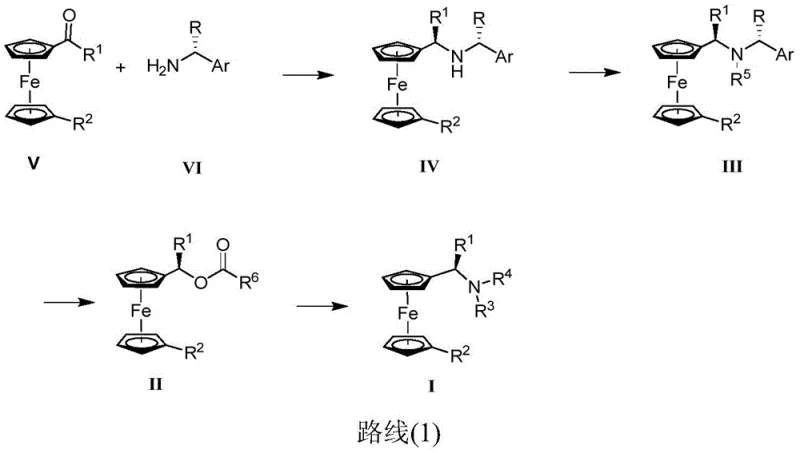
The mechanistic insights into this synthesis reveal a sophisticated interplay of Lewis acid catalysis and nucleophilic substitution that ensures high stereocontrol. The initial step involves the condensation of a ferrocene derivative (Formula V) with a chiral amine (Formula VI) in the presence of tetraisopropyl titanate, forming an imine intermediate that is subsequently reduced by sodium borohydride. This reductive amination is critical as it establishes the first chiral center adjacent to the ferrocene moiety, setting the stage for subsequent functionalization. The use of Ti(Oi-Pr)4 acts as a water scavenger and Lewis acid, driving the equilibrium towards imine formation while activating the carbonyl group for nucleophilic attack. Following this, the amino group of the resulting intermediate (Formula IV) undergoes alkylation or further reductive amination to introduce additional substituents, allowing for the diversification of the ligand structure to meet specific catalytic requirements.
Impurity control is meticulously managed through the selection of reagents and reaction conditions that favor the formation of the desired optical isomer. The patent specifies the use of specific molar ratios, such as 1: (1-3) for the Lewis acid and chiral amine relative to the ferrocene derivative, to minimize side reactions like over-alkylation or polymerization. The subsequent substitution reaction with acid anhydrides converts the amine into an acetate intermediate (Formula II), which is then displaced by a secondary amine to yield the final Ugi's amine (Formula I). This acetylation-substitution sequence is particularly advantageous as it allows for the inversion or retention of configuration depending on the specific conditions, providing flexibility in synthesizing different optical isomers. The rigorous control over reaction parameters ensures that the final product meets stringent purity specifications required for high-purity OLED material and electronic chemical applications.
How to Synthesize Chiral Ugi's Amine Efficiently
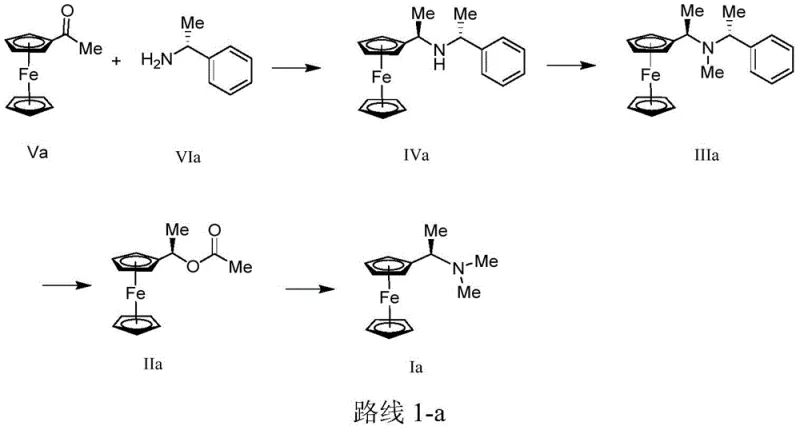
The synthesis of chiral Ugi's amine derivatives described in this patent offers a practical and scalable pathway for manufacturing facilities aiming to produce high-value ferrocene ligands. The process begins with the condensation of acetyl ferrocene and a chiral amine, followed by reduction to establish the core chiral amine structure. Subsequent steps involve methylation or alkylation to modify the nitrogen substituents, followed by acetylation and final amine substitution to complete the molecular architecture. This route is designed to be robust, utilizing common solvents like ethanol and methanol, and reagents that are easily sourced from global chemical suppliers. The detailed standardized synthetic steps see the guide below for specific operational parameters and safety considerations essential for laboratory and plant-scale execution.
- Perform reductive amination between a ferrocene derivative (Formula V) and a chiral amine (Formula VI) using Ti(Oi-Pr)4 and NaBH4 to obtain Formula IV.
- Conduct alkylation or reductive amination on the amino group of Formula IV to generate the intermediate Formula III.
- Execute substitution reaction with acid anhydride followed by amine substitution to yield the final chiral Ugi's amine (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers profound advantages for procurement managers and supply chain heads looking to optimize costs and ensure continuity. By eliminating the need for chiral resolution, the process inherently reduces raw material consumption and waste disposal costs, leading to significant cost reduction in fine chemical manufacturing. The use of commercially available chiral amines as starting materials mitigates the risk of supply chain disruptions associated with specialized catalysts or resolving agents. Furthermore, the mild reaction conditions and high yields contribute to a more predictable production schedule, reducing lead time for high-purity pharmaceutical intermediates and enhancing overall supply chain reliability. The scalability of the process allows for seamless transition from kilogram to multi-ton production, ensuring that manufacturers can meet fluctuating market demands without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The elimination of expensive chiral resolution agents and the associated loss of material during recrystallization results in a drastic simplification of the cost structure. By utilizing a chiral pool approach, the process avoids the 50% yield loss typical of resolution methods, effectively doubling the output from the same amount of starting ferrocene material. Additionally, the removal of transition metal catalysts in the early stages reduces the need for costly heavy metal scavenging steps, further lowering the operational expenditure. This qualitative improvement in process efficiency translates directly to substantial cost savings, making the final ligand more competitive in the global market without sacrificing purity or performance.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as acetyl ferrocene, sodium borohydride, and common chiral amines ensures a stable and diversified supply base. Unlike processes dependent on proprietary enzymes or rare earth catalysts, this method leverages widely available reagents, minimizing the risk of single-source bottlenecks. The robustness of the reaction conditions also means that production is less susceptible to variations in raw material quality, ensuring consistent output even when sourcing from different vendors. This resilience is crucial for maintaining the continuity of supply for critical API intermediates, allowing downstream customers to plan their production schedules with greater confidence and reduced inventory buffers.
- Scalability and Environmental Compliance: The synthetic route is designed with industrial scale-up in mind, utilizing solvents and reagents that are manageable in large-scale reactors. The absence of harsh conditions or hazardous byproducts simplifies waste treatment and aligns with increasingly stringent environmental regulations. The high atom economy of the reductive amination and substitution steps minimizes the generation of organic waste, reducing the environmental footprint of the manufacturing process. This compliance not only avoids potential regulatory fines but also enhances the corporate sustainability profile, which is becoming a key factor in supplier selection for multinational pharmaceutical and agrochemical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of chiral Ugi's amine derivatives. These answers are derived directly from the technical disclosures and experimental data within the patent, providing clarity on the feasibility and advantages of this novel method. Understanding these details is essential for stakeholders evaluating the integration of this technology into their existing manufacturing workflows or supply chains.
Q: How does this method improve optical purity compared to traditional resolution?
A: This method utilizes commercially available chiral amines to introduce chirality early in the synthesis, avoiding the cumbersome recrystallization and low efficiency associated with traditional racemate resolution using tartaric acid.
Q: What are the key reaction conditions for the reductive amination step?
A: The condensation reaction typically occurs at 35-45 °C using Lewis acids like Ti(Oi-Pr)4, followed by reduction with NaBH4 at 25 °C, ensuring mild conditions suitable for scale-up.
Q: Is this process suitable for industrial scale production of Josiphos ligands?
A: Yes, the patent explicitly states the method is suitable for industrial scale production due to easily obtained raw materials, simple synthetic routes, and high yields without expensive chiral catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Ugi's Amine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging advanced technologies like the one described in CN107286203B to deliver superior chiral intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from lab bench to full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral Ugi's amine meets the exacting standards required for asymmetric catalysis. Our commitment to technical excellence allows us to tackle complex synthetic challenges, providing our partners with a competitive edge in the development of next-generation pharmaceuticals and agrochemicals.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through innovative chemical solutions. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can enhance your project's success. Let us be your partner in engineering chemical excellence and driving your business forward with reliable, high-quality intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
