Advanced Crystallization Technology for High-Purity Gamma-ICD and Commercial Scalability
Advanced Crystallization Technology for High-Purity Gamma-ICD and Commercial Scalability
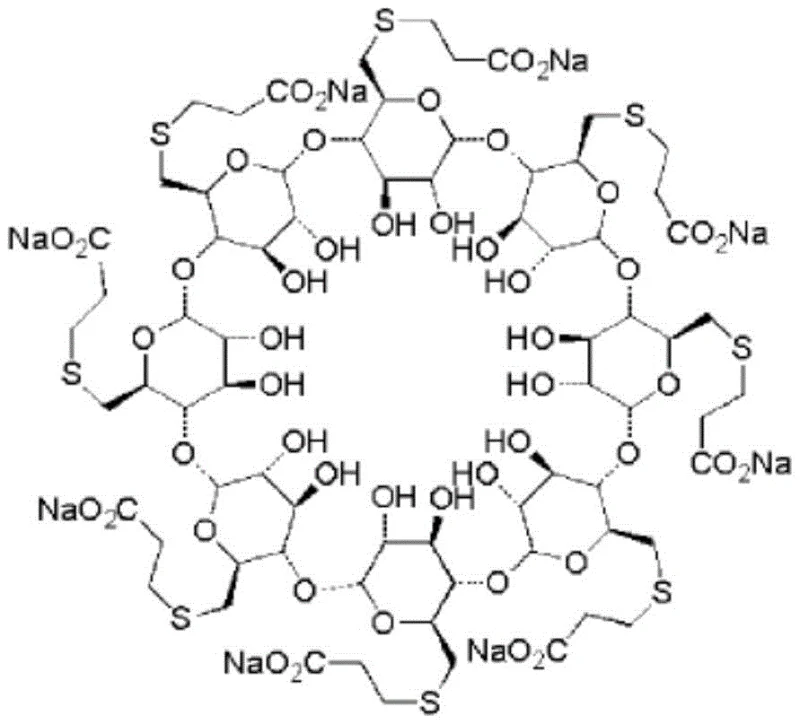
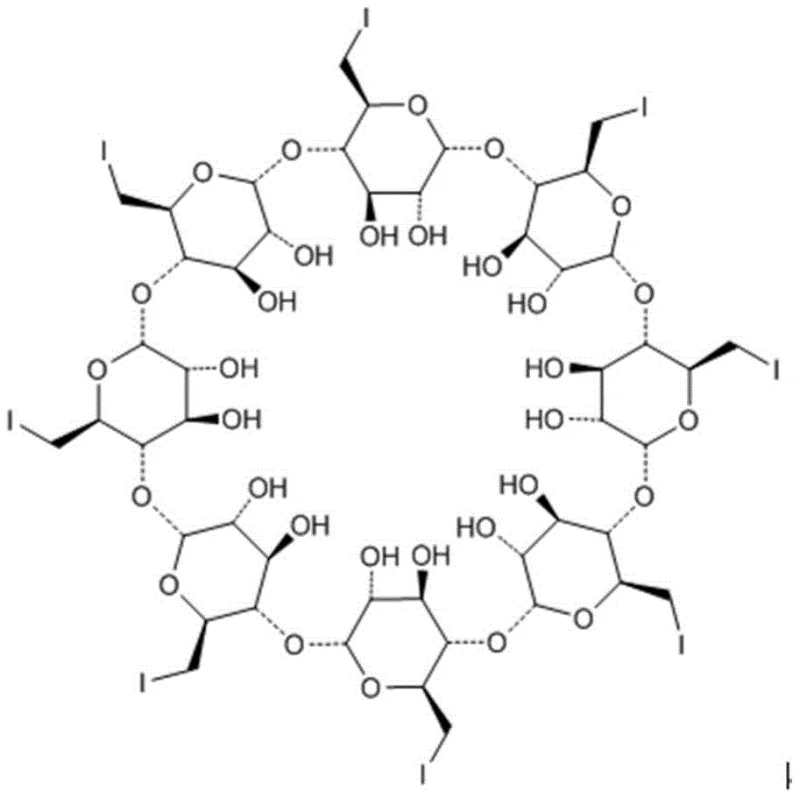
The pharmaceutical industry continuously seeks robust methodologies for synthesizing complex intermediates, particularly for novel muscle relaxant antagonists like sugammadex sodium. Patent CN109593143B introduces a groundbreaking purification method for preparing the critical intermediate gamma-ICD (6-per-deoxy-6-per-iodo-gamma-cyclodextrin) from sugammadex sodium precursors. This technology addresses the longstanding challenge of isolating high-purity cyclodextrin derivatives that possess unique solubility profiles, which have historically hindered efficient large-scale production. By utilizing a sophisticated two-step crystallization process involving DMF, water, and acetone, the invention successfully eliminates impurities with varying polarities that conventional single-solvent systems fail to remove. This advancement not only streamlines the synthetic route but also establishes a new benchmark for purity standards in the manufacturing of selective muscle relaxant antagonists, ensuring that downstream reactions proceed with maximum efficiency and minimal byproduct formation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of gamma-ICD has been plagued by significant technical hurdles due to its distinct physicochemical properties. The molecule exhibits high polarity yet demonstrates poor solubility in common protic solvents like water and methanol, while remaining soluble only in specific polar aprotic solvents such as DMF, DMSO, and formamide. Traditional purification techniques, including standard recrystallization or preparative High-Performance Liquid Chromatography (HPLC), often yield suboptimal results because they cannot simultaneously address the spectrum of impurities present in the crude reaction mixture. Specifically, impurities with larger polarity elute before the main peak, while those with smaller polarity elute after, creating a complex impurity profile that is resistant to simple separation. Furthermore, the structural complexity of the iodinated cyclodextrin core makes it susceptible to degradation under harsh purification conditions, leading to reduced overall yields and increased waste generation. These limitations result in elevated production costs and extended lead times, making the reliable supply of high-quality intermediates a persistent bottleneck for pharmaceutical manufacturers aiming to scale up sugammadex sodium production.
The Novel Approach
The innovative method disclosed in the patent overcomes these barriers through a strategic, sequential crystallization protocol that leverages the differential solubility of impurities in mixed solvent systems. The process begins by dissolving the crude gamma-ICD in DMF and introducing water dropwise, which selectively precipitates the target compound while retaining highly polar impurities in the supernatant. Following filtration, the wet product undergoes a second crystallization step using a DMF-acetone system, specifically designed to exclude less polar contaminants that survived the first stage. This dual-solvent strategy effectively fractionates the impurity profile, allowing for the isolation of gamma-ICD with exceptional purity levels, reported in examples to reach as high as 99.86%. By omitting complex chromatographic columns and reducing the reliance on expensive stationary phases, this approach simplifies the operational workflow and significantly enhances the throughput capacity of the manufacturing facility. The result is a streamlined, cost-effective purification pathway that maintains the structural integrity of the sensitive iodinated cyclodextrin while delivering the rigorous quality required for subsequent coupling reactions.
Mechanistic Insights into Solvent-Driven Fractional Crystallization
The core mechanism driving this purification success lies in the precise manipulation of solvent polarity and solubility parameters to control nucleation and crystal growth. In the first stage, the addition of water to the DMF solution drastically alters the dielectric constant of the medium, reducing the solvation energy available for the gamma-ICD molecules and forcing them out of the solution as a solid phase. However, impurities with even higher polarity remain solvated due to their stronger interactions with the aqueous component, effectively separating them from the precipitating product. The second stage utilizes acetone, a solvent with different hydrogen bonding capabilities and polarity compared to water, to further refine the crystal lattice. As the acetone is introduced to the DMF-dissolved intermediate, it creates an environment where low-polarity organic byproducts remain soluble, while the gamma-ICD, now freed from the bulk of polar contaminants, crystallizes with high fidelity. This mechanistic understanding allows process chemists to fine-tune parameters such as temperature (optimized between 50-100°C for dissolution and 25°C for crystallization) and solvent ratios to maximize recovery rates.
Furthermore, the control of impurity profiles is critical for the downstream synthesis of sugammadex sodium, where residual halides or organic byproducts could interfere with the thioetherification reaction. The described crystallization method ensures that the iodine content and substitution pattern on the cyclodextrin ring are preserved without dehalogenation or structural rearrangement. By maintaining the reaction temperature within a controlled range during the dissolution phases (preferably 70°C) and ensuring slow cooling rates, the process promotes the formation of large, well-defined crystals that trap fewer solvent inclusions and mother liquor impurities. This level of control is essential for meeting the stringent regulatory requirements for pharmaceutical intermediates, where the identity and quantity of every potential genotoxic impurity must be rigorously monitored. The ability to achieve such high purity through physical separation rather than chemical transformation underscores the elegance of this purification strategy, providing a robust platform for consistent batch-to-batch quality.
How to Synthesize Gamma-ICD Efficiently
The synthesis and purification of gamma-ICD require precise adherence to solvent ratios and thermal conditions to ensure optimal yield and purity. The patented process outlines a clear sequence starting with the Vilsmeier-Hack reaction to generate the crude iodinated species, followed immediately by the two-stage crystallization protocol. Operators must carefully manage the dropwise addition of anti-solvents to prevent oiling out, which can trap impurities within the solid matrix. The detailed standardized synthesis steps below provide a comprehensive guide for implementing this technology in a pilot or commercial setting, ensuring that the critical quality attributes of the intermediate are maintained throughout the production cycle.
- Dissolve crude gamma-ICD in DMF at 50-100°C, then add water dropwise to precipitate solids and remove high-polarity impurities.
- Filter the wet product and redissolve in a smaller amount of DMF at 50-100°C.
- Add acetone to the solution, cool to 25°C, stir for 2-5 hours, and filter to obtain pure gamma-ICD with removed low-polarity impurities.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this purification technology translates directly into enhanced operational efficiency and reduced total cost of ownership. By eliminating the need for complex and resource-intensive purification steps such as preparative HPLC or multiple column chromatographies, the manufacturing process becomes significantly more lean and agile. This simplification reduces the consumption of expensive stationary phases and large volumes of specialized solvents, leading to substantial cost savings in raw material procurement. Additionally, the streamlined workflow shortens the overall production cycle time, allowing for faster turnover of batches and improved responsiveness to market demand fluctuations. The robustness of the crystallization method also minimizes the risk of batch failures due to purification inefficiencies, thereby securing a more stable and predictable supply of this critical pharmaceutical intermediate for downstream API synthesis.
- Cost Reduction in Manufacturing: The transition from complex chromatographic separation to simple crystallization drastically lowers the operational expenditure associated with purification. Removing the dependency on expensive silica gels or resin columns eliminates a major cost driver, while the recycling potential of the DMF-acetone solvent system further enhances economic efficiency. This shift allows manufacturers to allocate resources more effectively, focusing on yield optimization rather than waste management, ultimately resulting in a more competitive pricing structure for the final API.
- Enhanced Supply Chain Reliability: The simplicity of the equipment required for this crystallization process—primarily standard reactors and filtration units—means that production can be easily scaled across multiple facilities without specialized infrastructure. This flexibility mitigates supply chain risks associated with bottlenecks in specialized purification capacity. Furthermore, the high purity achieved reduces the likelihood of downstream reaction failures, ensuring a continuous flow of materials for the synthesis of sugammadex sodium and preventing costly delays in the overall drug manufacturing timeline.
- Scalability and Environmental Compliance: From an environmental perspective, the reduction in solvent usage and the elimination of solid waste from chromatography columns align with green chemistry principles. The process generates less hazardous waste, simplifying disposal and compliance with increasingly strict environmental regulations. The scalability of the crystallization steps ensures that production can be ramped up from kilogram to tonne scale with minimal re-engineering, supporting the long-term commercial viability of the drug product while maintaining a sustainable manufacturing footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the purification of gamma-ICD, derived directly from the patent specifications and industry best practices. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into their existing production lines. The answers provided reflect the specific advantages and operational parameters defined in the intellectual property, offering clarity on how this technology resolves traditional purification bottlenecks.
Q: Why is gamma-ICD difficult to purify using standard methods?
A: Gamma-ICD exhibits complex solubility characteristics, being soluble in polar aprotic solvents like DMF and DMSO but insoluble in water and methanol. Its structural complexity and polarity profile make standard HPLC or single-solvent crystallization ineffective for removing both high and low polarity impurities simultaneously.
Q: What is the key advantage of the DMF-water-acetone system?
A: The dual-solvent system allows for selective precipitation. The initial DMF-water step targets high-polarity impurities, while the subsequent DMF-acetone step effectively removes low-polarity contaminants, achieving purity levels up to 99.86% without complex chromatography.
Q: How does this method impact industrial production costs?
A: By replacing complex purification processes like preparative HPLC with simple crystallization steps, the method significantly reduces material input, solvent consumption, and processing time, leading to substantial cost savings and easier scale-up for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gamma-ICD Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the successful development and commercialization of complex pharmaceutical agents like sugammadex sodium. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch against the highest global standards. Our capability to implement advanced crystallization technologies allows us to offer a reliable supply of gamma-ICD that supports your R&D and commercial manufacturing needs with unwavering consistency.
We invite you to collaborate with us to leverage these technological advancements for your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized purification processes can enhance your supply chain resilience and drive down your overall manufacturing costs.
