Transforming Byproduct Light Oil into High-Value o-Chlorocyclohexanone: A Technical & Commercial Analysis
Introduction: Unlocking Value from Cyclohexanone Byproducts
The chemical industry is constantly seeking pathways to transform low-value byproducts into high-performance intermediates, and patent CN107540531B presents a compelling solution for the synthesis of o-chlorocyclohexanone. This critical intermediate serves as a precursor for catechol, furocoumarin compounds, and various agrochemical and pharmaceutical agents, yet its traditional production has often been plagued by inefficiency. The disclosed technology leverages light oil, a specific byproduct generated during the industrial oxidation of cyclohexane, which typically contains 30-40% cyclohexene oxide. By capitalizing on this abundant resource, the method not only addresses environmental concerns regarding waste disposal but also establishes a robust, cost-effective supply chain for high-purity organic synthesis. This report analyzes the technical merits and commercial viability of this novel approach, demonstrating how it outperforms legacy methodologies in both yield and operational simplicity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of o-chlorocyclohexanone has relied on several established routes, each carrying significant drawbacks that hinder large-scale efficiency. As illustrated in the comparative reaction schemes below, methods such as the direct chlorination of cyclohexanone often suffer from poor selectivity, leading to the formation of poly-chlorinated byproducts that are difficult to separate. Similarly, the cyclohexene method and enol method frequently require harsh reaction conditions, including extreme temperatures or the use of hazardous reagents, which complicate safety protocols and increase energy consumption. Furthermore, the epoxycyclohexane method, while chemically sound, traditionally necessitates the use of additional solvents and rigorous purification steps to achieve acceptable purity levels. These cumulative inefficiencies result in higher production costs and inconsistent product quality, creating a bottleneck for manufacturers requiring reliable supplies of this key intermediate for downstream applications.
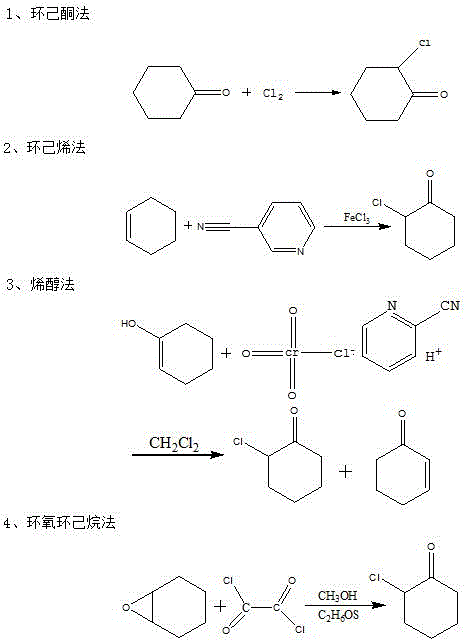
The Novel Approach
In stark contrast to these legacy processes, the method described in CN107540531B introduces a streamlined pathway that bypasses many of these inherent obstacles by utilizing light oil directly. This innovative route eliminates the need for expensive, pure starting materials by valorizing a waste stream rich in cyclohexene oxide. The process operates under remarkably mild conditions, with the ring-opening reaction occurring effectively between 10-60°C, thereby reducing thermal stress on the equipment and minimizing energy expenditure. Crucially, the inclusion of specific auxiliary agents enhances the reaction kinetics, allowing for high conversion rates without the need for complex solvent systems in the initial stage. This results in a simplified workflow where the crude product can be easily separated and purified through standard distillation techniques, yielding o-chlorocyclohexanone with purity exceeding 99% and yields consistently above 90%, marking a substantial improvement over conventional industrial standards.
Mechanistic Insights into Additive-Assisted Ring Opening and Oxidation
The core of this technological advancement lies in the precise control of the ring-opening reaction of cyclohexene oxide in the presence of hydrochloric acid and a carefully selected auxiliary agent. The mechanism involves the protonation of the epoxide oxygen, followed by a nucleophilic attack by the chloride ion to form 2-chlorocyclohexanol. The addition of salts such as sodium chloride, zinc chloride, or calcium chloride plays a pivotal role in this transformation; these additives likely function by modifying the ionic strength of the reaction medium or by coordinating with the intermediate species to stabilize the transition state. This stabilization effect significantly boosts the selectivity towards the desired mono-chlorinated alcohol, suppressing competing side reactions that typically lead to impurity formation. By optimizing the molar ratio of cyclohexene oxide to hydrogen chloride to between 1:1 and 1:2, the process ensures complete consumption of the epoxide while minimizing the presence of unreacted acid in the final mixture, setting the stage for a clean subsequent oxidation step.
Following the isolation of high-purity 2-chlorocyclohexanol, the second critical phase involves selective oxidation to the ketone. The patent highlights the efficacy of using Dess-Martin periodinane (DMP) or tetramethyl piperidine as the oxidant, operating at moderate temperatures of 20-40°C. This choice of oxidant is strategic, as it offers high chemoselectivity, converting the secondary alcohol to the ketone without affecting other sensitive functional groups or causing over-oxidation to carboxylic acids. The mild nature of this oxidation step preserves the structural integrity of the cyclohexane ring, ensuring that the final o-chlorocyclohexanone retains its utility for further synthetic elaboration. Post-reaction washing with aqueous solutions of sodium carbonate or sodium thiosulfate effectively removes acidic byproducts and residual iodine species from the DMP, facilitating a straightforward rectification process that delivers the final product with exceptional purity suitable for sensitive pharmaceutical applications.
How to Synthesize o-Chlorocyclohexanone Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible framework for manufacturing o-chlorocyclohexanone with industrial relevance. The process begins with the charging of light oil into a reactor, followed by the controlled addition of hydrochloric acid and the auxiliary agent under stirring. Temperature control is maintained strictly within the 10-60°C range to optimize the ring-opening kinetics, after which the mixture is allowed to settle for phase separation. The resulting oil phase undergoes distillation to remove volatile components, isolating the 2-chlorocyclohexanol intermediate which is then subjected to oxidation. For R&D teams looking to implement this, the detailed standardized synthesis steps are provided in the guide below to ensure consistent replication of the high yields and purity reported in the patent examples.
- React cyclohexene oxide-containing light oil with hydrochloric acid solution in the presence of an auxiliary agent like sodium chloride at 10-60°C to form 2-chlorocyclohexanol.
- Separate the oil phase and remove low-boiling components via distillation to isolate high-purity 2-chlorocyclohexanol.
- Oxidize the 2-chlorocyclohexanol using Dess-Martin periodinane or tetramethyl piperidine at 20-40°C, followed by washing and rectification to collect o-chlorocyclohexanone.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers transformative benefits that directly impact the bottom line and operational resilience. By shifting the raw material base from refined cyclohexanone or cyclohexene to light oil—a byproduct of existing large-scale cyclohexane oxidation plants—manufacturers can drastically reduce raw material acquisition costs. This strategy not only lowers the direct cost of goods sold but also insulates the supply chain from volatility in the pricing of primary petrochemical feedstocks. The ability to source raw materials from waste streams creates a circular economy model that is increasingly favored in modern chemical manufacturing, providing a dual advantage of cost efficiency and enhanced sustainability credentials which are vital for meeting corporate ESG goals.
- Cost Reduction in Manufacturing: The elimination of expensive solvents in the initial ring-opening step represents a significant saving in both material costs and waste disposal fees. Traditional methods often require vast quantities of organic solvents to manage exothermic reactions or solubilize reagents, whereas this novel approach utilizes the light oil matrix itself effectively. Furthermore, the high selectivity achieved through the use of auxiliary agents minimizes the formation of hard-to-remove impurities, reducing the load on downstream purification units and lowering the overall energy consumption required for distillation and refining. These cumulative efficiencies translate into a leaner manufacturing process with a substantially lower cost basis per kilogram of finished product.
- Enhanced Supply Chain Reliability: Sourcing cyclohexene oxide from light oil, a ubiquitous byproduct of the nylon and phenol industries, ensures a stable and abundant supply of raw materials. Unlike specialized reagents that may be subject to geopolitical supply disruptions or limited vendor availability, light oil is generated continuously in major chemical hubs globally. This abundance allows for the establishment of long-term supply contracts with favorable terms, reducing the risk of production stoppages due to raw material shortages. Additionally, the mild reaction conditions reduce the dependency on specialized high-pressure or cryogenic equipment, making the technology easier to deploy across diverse manufacturing sites and enhancing overall supply chain flexibility.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations such as stirred tank reactors and standard distillation columns that are commonplace in fine chemical facilities. The absence of heavy metal catalysts and the use of relatively benign oxidants simplify the regulatory compliance landscape, particularly concerning residue limits in pharmaceutical intermediates. Waste generation is minimized through the efficient conversion of byproducts and the recycling of aqueous wash layers, aligning with stringent environmental regulations. This ease of scale-up from pilot to commercial production ensures that demand surges can be met rapidly without the need for extensive capital investment in new, specialized infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this light oil-based synthesis route. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on reaction parameters, impurity profiles, and scalability potential. Understanding these details is crucial for technical directors and procurement specialists evaluating the feasibility of integrating this method into their existing production portfolios.
Q: What are the primary advantages of using light oil over traditional cyclohexanone chlorination?
A: The light oil method utilizes a waste byproduct containing cyclohexene oxide, significantly reducing raw material costs compared to purchasing pure cyclohexanone. Furthermore, it avoids the harsh reaction conditions and complex purification steps associated with direct chlorination, leading to higher selectivity and fewer poly-chlorinated impurities.
Q: How does the addition of auxiliary agents like NaCl improve the reaction?
A: Auxiliary agents such as sodium chloride, zinc chloride, or calcium chloride act to enhance the selectivity and conversion rate of the ring-opening reaction. They help manage the reaction environment, ensuring that the cyclohexene oxide converts efficiently to 2-chlorocyclohexanol without excessive side reactions, thereby simplifying downstream purification.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process is designed for industrial scalability. It operates under mild temperatures (10-60°C) and atmospheric or reduced pressure distillation, avoiding extreme pressures or cryogenic conditions. The use of common reagents like HCl and standard oxidants makes it highly adaptable for large-scale manufacturing of fine chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable o-Chlorocyclohexanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the global pharmaceutical and agrochemical supply chains. Our team of expert chemists has extensively analyzed the pathway described in CN107540531B and possesses the technical capability to adapt and optimize this route for commercial-scale production. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial reality is seamless. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, delivering o-chlorocyclohexanone that meets the exacting standards required for API synthesis and fine chemical applications.
We invite you to collaborate with us to leverage this cost-effective technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this novel route can improve your margins. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can support your long-term supply chain objectives.
