Advanced Synthesis of 4,4'-[1-(Trifluoromethyl)alkylidene]-bis-(2,6-diphenylphenols) for High-Performance Polymers
Advanced Synthesis of 4,4'-[1-(Trifluoromethyl)alkylidene]-bis-(2,6-diphenylphenols) for High-Performance Polymers
The chemical industry is constantly seeking more efficient and scalable routes for producing high-performance polymer precursors, and Patent CN101903320B presents a groundbreaking methodology for the production of 4,4'-[1-(trifluoromethyl)alkylidene]-bis-(2,6-diphenylphenols). These compounds serve as valuable raw materials for the synthesis of advanced polycarbonates and polyesters, offering superior thermal and mechanical properties. The patent details a novel multi-step process that begins with the self-condensation of cyclohexanone, followed by a sophisticated dehydrogenation step and a final coupling reaction with trifluoromethyl ketones. This approach represents a significant departure from traditional methods that often rely on hazardous gas-phase reactions or expensive starting materials. By shifting the paradigm to a condensed-phase liquid process, the technology offers enhanced safety profiles and improved control over reaction parameters, which is critical for maintaining high purity standards in fine chemical manufacturing.
![General chemical structure of 4,4'-[1-(trifluoromethyl)alkylidene]-bis-(2,6-diphenylphenols) showing the core bis-phenol framework](/insights/img/trifluoromethyl-bis-phenol-synthesis-pharma-supplier-20260313185247-01.webp)
Furthermore, the versatility of this synthesis allows for the production of various derivatives depending on the specific trifluoromethyl ketone employed, enabling customization for diverse application requirements in the pharmaceutical and agrochemical sectors. The core innovation lies in the efficient transformation of readily available cyclohexanone into complex tricyclic intermediates, which are then aromatized to form the crucial 2,6-diphenylphenol scaffold. This scaffold is subsequently functionalized to create the final bis-phenol product, ensuring a robust supply chain for these high-value intermediates. For R&D directors and procurement managers, understanding the nuances of this pathway is essential for evaluating potential suppliers who can deliver consistent quality at a competitive cost structure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of similar bis-phenol compounds has relied on methods described in older patents such as US 3,739,035, which involve reacting 2,6-diphenylphenol with excessive amounts of gaseous perfluoroacetone or utilizing methylsulfonic acid under harsh conditions. These conventional approaches suffer from several inherent drawbacks, including the handling of hazardous gaseous reagents which poses significant safety risks and requires specialized containment infrastructure. Additionally, the use of large excesses of reagents not only drives up raw material costs but also complicates the downstream purification process, leading to lower overall yields and increased waste generation. The reliance on gas-phase dehydrogenation in some prior art methods further exacerbates these issues by requiring extremely high temperatures and offering limited control over selectivity, often resulting in the formation of undesirable by-products that are difficult to separate.
The Novel Approach
In contrast, the method disclosed in Patent CN101903320B introduces a streamlined liquid-phase process that mitigates many of these challenges by utilizing cyclohexanone as a primary feedstock. This novel approach involves the base-catalyzed self-condensation of cyclohexanone to form tricyclic condensation products, which are then subjected to a controlled dehydrogenation step in the condensed phase. 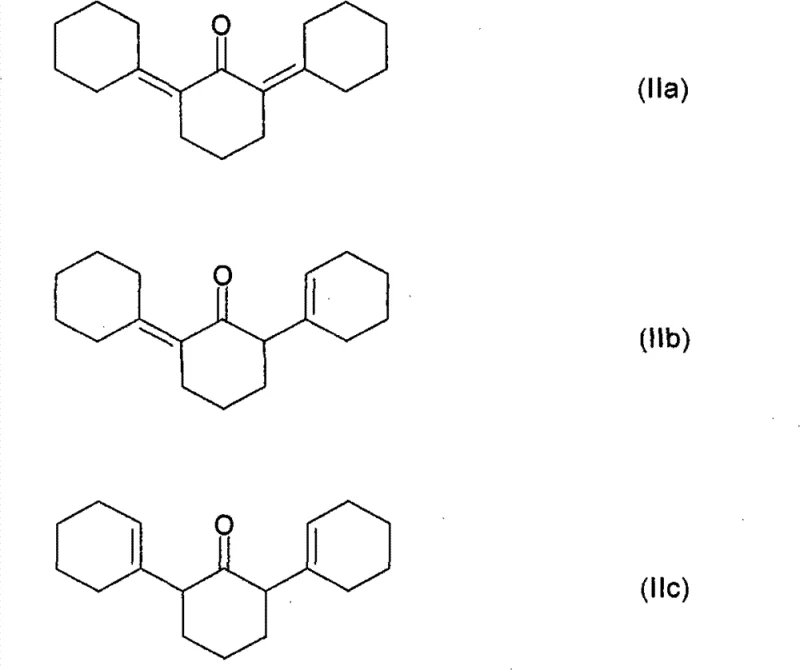 This shift to a condensed-phase reaction environment allows for much finer control over temperature and pressure, significantly reducing the risk of thermal runaway and improving the selectivity towards the desired aromatic products. Furthermore, the process employs carrier-bound transition metal catalysts that can be easily separated and recycled, addressing both economic and environmental concerns associated with homogeneous catalysis. The final coupling step utilizes strong organic acids in a controlled manner to link the phenolic units with the trifluoromethyl group, ensuring high conversion rates without the need for extreme conditions.
This shift to a condensed-phase reaction environment allows for much finer control over temperature and pressure, significantly reducing the risk of thermal runaway and improving the selectivity towards the desired aromatic products. Furthermore, the process employs carrier-bound transition metal catalysts that can be easily separated and recycled, addressing both economic and environmental concerns associated with homogeneous catalysis. The final coupling step utilizes strong organic acids in a controlled manner to link the phenolic units with the trifluoromethyl group, ensuring high conversion rates without the need for extreme conditions.
Mechanistic Insights into Cyclohexanone Self-Condensation and Dehydrogenation
The heart of this innovative synthesis lies in the intricate mechanistic pathway that transforms simple cyclohexanone into the complex 2,6-diphenylphenol intermediate. The process initiates with an aldol self-condensation reaction where three molecules of cyclohexanone react in the presence of a strong basic catalyst, such as sodium hydroxide or potassium carbonate, to form a mixture of tricyclic ketones designated as formulas (IIa), (IIb), and (IIc). This step is critical as it establishes the carbon skeleton required for the subsequent aromatization, and the use of azeotropic distillation to remove water drives the equilibrium towards the formation of these tricyclic products. Following the isolation of these intermediates, typically via fractional distillation, the mixture undergoes a heterogeneous dehydrogenation reaction.
This dehydrogenation step is catalyzed by transition metals like palladium or platinum supported on carriers such as alumina or activated carbon, operating at elevated temperatures in the liquid phase. 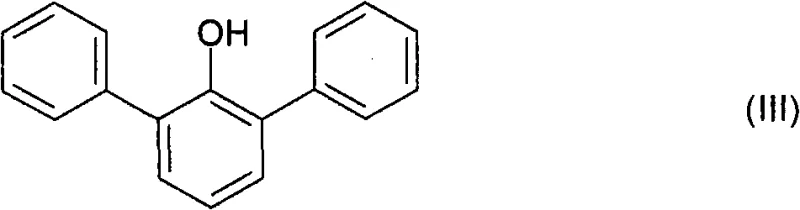 The mechanism involves the abstraction of hydrogen atoms from the saturated rings of the tricyclic ketones, facilitated by the metal surface, leading to the formation of the aromatic 2,6-diphenylphenol structure. The presence of basic promoters like potassium carbonate during this step has been found to enhance catalyst activity and longevity, allowing for multiple recycling cycles without significant degradation. Finally, the purified 2,6-diphenylphenol reacts with a trifluoromethyl ketone, such as 1,1,1-trifluoroacetone, in the presence of a strong organic acid like methanesulfonic acid.
The mechanism involves the abstraction of hydrogen atoms from the saturated rings of the tricyclic ketones, facilitated by the metal surface, leading to the formation of the aromatic 2,6-diphenylphenol structure. The presence of basic promoters like potassium carbonate during this step has been found to enhance catalyst activity and longevity, allowing for multiple recycling cycles without significant degradation. Finally, the purified 2,6-diphenylphenol reacts with a trifluoromethyl ketone, such as 1,1,1-trifluoroacetone, in the presence of a strong organic acid like methanesulfonic acid.  This electrophilic aromatic substitution links the two phenolic units via the trifluoromethyl alkylidene bridge, completing the synthesis of the target bis-phenol compound with high regioselectivity and purity.
This electrophilic aromatic substitution links the two phenolic units via the trifluoromethyl alkylidene bridge, completing the synthesis of the target bis-phenol compound with high regioselectivity and purity.
How to Synthesize 4,4'-[1-(Trifluoromethyl)ethylidene]-bis-(2,6-diphenylphenol) Efficiently
To achieve optimal results in the synthesis of 4,4'-[1-(trifluoromethyl)ethylidene]-bis-(2,6-diphenylphenol), precise control over reaction conditions and purification steps is paramount. The process begins with the careful selection of the basic catalyst and solvent system for the initial condensation, followed by rigorous distillation to isolate the tricyclic intermediates. The subsequent dehydrogenation requires a robust heterogeneous catalyst system capable of withstanding high temperatures while maintaining activity over repeated cycles.
- Perform self-condensation of cyclohexanone in the presence of a basic catalyst to form tricyclic condensation products (IIa, IIb, IIc).
- Separate the tricyclic condensation products and subject them to dehydrogenation using a carrier-bound transition metal catalyst to yield 2,6-diphenylphenol.
- React the purified 2,6-diphenylphenol with a trifluoromethyl ketone in the presence of a strong organic acid to form the final bis-phenol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers substantial strategic benefits that extend beyond mere technical feasibility. The shift from gas-phase to condensed-phase processing significantly reduces the capital expenditure required for specialized high-pressure equipment and safety systems, thereby lowering the barrier to entry for production and potentially reducing the unit cost of the final intermediate. Moreover, the ability to recycle the carrier-bound transition metal catalysts multiple times without significant loss of performance translates directly into reduced consumption of precious metals, which is a major cost driver in fine chemical manufacturing. This efficiency gain allows suppliers to offer more competitive pricing structures while maintaining healthy margins, providing a distinct advantage in price-sensitive markets.
- Cost Reduction in Manufacturing: The elimination of hazardous gaseous reagents and the use of recyclable heterogeneous catalysts drastically simplify the production workflow, leading to significant operational cost savings. By avoiding the need for complex gas handling infrastructure and minimizing waste disposal costs associated with excess reagents, manufacturers can achieve a leaner and more cost-effective production model. The use of readily available cyclohexanone as a starting material further stabilizes raw material costs, shielding the supply chain from volatility associated with exotic or scarce precursors.
- Enhanced Supply Chain Reliability: The robustness of the liquid-phase process ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted production schedules for downstream polymer manufacturers. The ability to purify intermediates effectively through crystallization and distillation minimizes the risk of impurity carryover, reducing the likelihood of batch failures and delivery delays. This reliability makes the supplier a more dependable partner for long-term contracts, ensuring a steady flow of high-quality intermediates necessary for just-in-time manufacturing environments.
- Scalability and Environmental Compliance: The condensed-phase nature of the reaction facilitates easier scale-up from pilot plant to commercial production volumes without the engineering challenges associated with gas-phase reactors. Additionally, the reduced generation of hazardous waste and the potential for solvent recovery align with increasingly stringent environmental regulations, reducing the compliance burden and associated costs. This sustainability aspect not only future-proofs the supply chain against regulatory changes but also enhances the corporate social responsibility profile of the end-product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these bis-phenol intermediates, based on the detailed specifications provided in the patent literature. Understanding these aspects helps stakeholders make informed decisions about integrating this technology into their existing supply chains.
Q: What are the advantages of the condensed-phase dehydrogenation method over conventional gas-phase processes?
A: The condensed-phase method allows for better temperature control and utilizes recyclable carrier-bound transition metal catalysts, significantly reducing operational hazards and catalyst consumption compared to traditional high-temperature gas-phase dehydrogenation.
Q: How is the purity of the intermediate 2,6-diphenylphenol ensured?
A: High purity is achieved through a specific crystallization process using solvents like heptane or heptane/isopropanol mixtures, which effectively separates the desired phenol from partially dehydrogenated by-products and tricyclic ketones.
Q: Can the transition metal catalyst be reused in this synthesis?
A: Yes, the carrier-bound transition metal catalysts, particularly palladium on alumina or carbon, can be separated by filtration and recycled multiple times without significant loss of activity or selectivity, enhancing process economics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-[1-(Trifluoromethyl)ethylidene]-bis-(2,6-diphenylphenol) Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of next-generation polymers and pharmaceuticals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the complex multi-step synthesis described in Patent CN101903320B can be executed with precision and efficiency. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 4,4'-[1-(trifluoromethyl)ethylidene]-bis-(2,6-diphenylphenol) meets the exacting standards required by global R&D teams. Our commitment to quality assurance means that you can rely on us for consistent supply without compromising on the chemical integrity of your final products.
We invite you to engage with our technical procurement team to discuss how our advanced manufacturing capabilities can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our process efficiencies translate into tangible value for your organization. We encourage you to reach out for specific COA data and route feasibility assessments to verify our capability to meet your unique project requirements. Let us be your partner in driving innovation through reliable and cost-effective chemical solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
