Scalable Electrochemical Route for High-Purity Aza-Anthraquinone Derivatives Manufacturing
Scalable Electrochemical Route for High-Purity Aza-Anthraquinone Derivatives Manufacturing
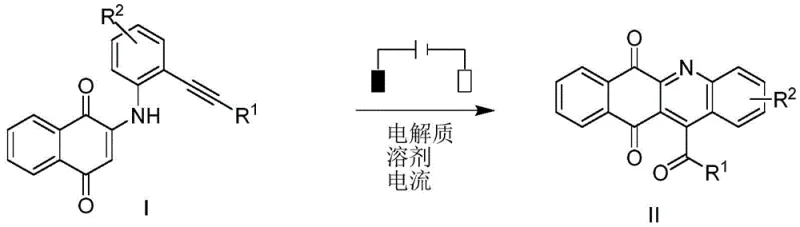
The pharmaceutical and fine chemical industries are currently witnessing a paradigm shift towards sustainable manufacturing technologies, driven by the urgent need to reduce environmental footprints and eliminate toxic heavy metal residues from active pharmaceutical ingredients (APIs). A groundbreaking development in this sector is detailed in patent CN114196973A, which discloses a novel method for the electrochemical synthesis of aza-anthraquinone derivatives. This technology represents a significant departure from conventional transition-metal catalyzed processes, utilizing clean electric energy to drive the oxidative cyclization of 1,6-eneyne precursors. By replacing stoichiometric chemical oxidants and expensive metal catalysts with electrons, this method not only aligns with the principles of green chemistry but also offers a streamlined pathway for producing complex nitrogen-containing heterocycles that are ubiquitous in antitumor, antifungal, and antiviral drug candidates.
For R&D directors and process chemists, the implications of this patent are profound, as it addresses the critical challenge of constructing polycyclic aromatic systems without generating stoichiometric amounts of hazardous waste. The core innovation lies in the electrochemical anodic oxidation of 2-((2-(phenylethynyl)phenyl)amino)naphthalene-1,4-dione compounds, which triggers a cascade of radical reactions leading to the formation of the quinoline-fused anthraquinone scaffold. This approach effectively bypasses the multi-step sequences traditionally required for such transformations, offering a direct, atom-economical route that simplifies downstream purification and significantly enhances the overall process mass intensity (PMI) of the synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of aza-anthraquinone frameworks has relied heavily on transition-metal catalysis, often involving palladium, copper, or rhodium complexes to facilitate intramolecular pi-extension reactions. While effective on a small laboratory scale, these traditional methods suffer from severe drawbacks when considered for commercial manufacturing, primarily due to the high cost of precious metal catalysts and the necessity for stoichiometric exogenous oxidants such as silver salts or hypervalent iodine reagents. Furthermore, the removal of trace metal residues from the final product is a rigorous and costly process, requiring specialized scavengers and extensive analytical testing to meet stringent regulatory limits for pharmaceutical intermediates. The generation of substantial chemical waste from oxidants and ligands also poses significant environmental compliance challenges, increasing the burden on waste treatment facilities and driving up the overall cost of goods sold (COGS).
The Novel Approach
In stark contrast, the electrochemical method disclosed in patent CN114196973A utilizes electricity as a traceless reagent, effectively decoupling the oxidation potential from chemical additives. By employing an undivided electrolytic cell with a carbon cloth anode and a platinum cathode, the system generates nitrogen-centered radicals directly from the substrate under mild constant current conditions. This eliminates the need for any external oxidants or transition metal catalysts, resulting in a reaction mixture that is inherently cleaner and easier to process. The use of hexafluoroisopropanol (HFIP) as a solvent further enhances the reaction efficiency by stabilizing radical intermediates through hydrogen bonding, allowing the cyclization to proceed smoothly at room temperature under an air atmosphere. This novel approach not only reduces the complexity of the reaction setup but also drastically simplifies the post-reaction workup, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Electrochemical Anodic Oxidation and Radical Cyclization
From a mechanistic perspective, the success of this transformation relies on the precise control of electrode potentials to selectively oxidize the amino-naphthoquinone substrate without degrading sensitive functional groups. Upon application of a constant current (typically 5-15 mA), the nitrogen atom of the aniline moiety undergoes single-electron oxidation at the anode surface to form a nitrogen-centered radical cation. This highly reactive species subsequently initiates an intramolecular attack on the pendant alkyne group, triggering a 6-endo-dig cyclization that forms the new carbon-nitrogen bond essential for the quinoline ring closure. The resulting vinyl radical is then further oxidized and deprotonated to restore aromaticity, completing the formation of the fused aza-anthraquinone core. This cascade sequence is remarkably efficient, avoiding the high-energy barriers associated with thermal cyclization methods and proceeding with high regioselectivity.
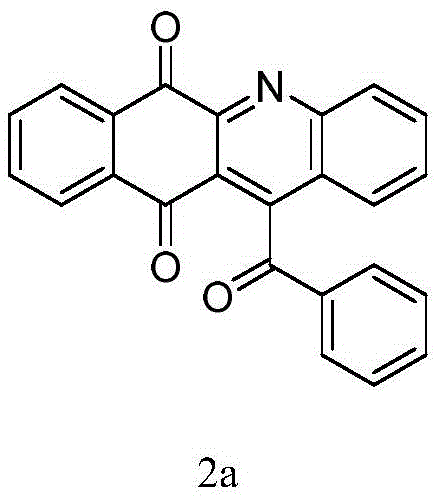
Crucially for quality control, this metal-free mechanism ensures that the final impurity profile is free from heavy metal contaminants, a common pain point in API synthesis. The tolerance of the electrochemical conditions towards various substituents is exceptional, as evidenced by the successful synthesis of derivatives bearing electron-withdrawing groups like chlorides and esters, as well as electron-donating groups like methoxy and alkyl chains. For instance, the synthesis of compound 2a serves as a prime example of the core scaffold's stability and the method's robustness, yielding the target molecule in high purity without the need for protective group manipulation on the quinone system. This broad substrate scope allows medicinal chemists to rapidly access diverse libraries of analogs for structure-activity relationship (SAR) studies without being constrained by catalyst compatibility issues.
How to Synthesize Aza-Anthraquinone Derivatives Efficiently
To implement this cutting-edge technology in a laboratory or pilot plant setting, operators must adhere to specific electrochemical parameters to ensure reproducibility and safety. The process begins with the dissolution of the 1,6-eneyne precursor and a supporting electrolyte, preferably tetrabutylammonium acetate, in a fluorinated alcohol solvent such as hexafluoroisopropanol. The reaction is conducted in an open system under ambient air, which serves as a benign terminal oxidant if needed, although the primary oxidation is driven by the anode. Detailed standard operating procedures regarding electrode spacing, current density, and monitoring techniques are critical for maintaining consistent yields across different batch sizes. The following guide outlines the standardized synthesis steps derived from the patent examples to assist technical teams in replicating this high-efficiency route.
- Dissolve the 2-((2-(phenylethynyl)phenyl)amino)naphthalene-1,4-dione substrate and tetrabutylammonium acetate electrolyte in hexafluoroisopropanol (HFIP) solvent within an undivided electrolytic cell.
- Insert a carbon cloth anode and platinum cathode, then apply a constant current of 10 mA under an air atmosphere at room temperature (20-25°C) while stirring.
- Monitor reaction progress via TLC, then perform aqueous workup followed by silica gel column chromatography to isolate the pure aza-anthraquinone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this electrochemical synthesis route offers tangible strategic benefits that extend beyond mere technical novelty. By fundamentally altering the input materials required for production, this method disrupts the traditional cost structure associated with complex heterocycle manufacturing. The elimination of precious metal catalysts removes a volatile cost component from the bill of materials, shielding the supply chain from fluctuations in the prices of palladium or rhodium. Additionally, the simplified purification process reduces the consumption of silica gel and solvents during chromatography, leading to substantial cost savings in consumables and waste disposal fees. These factors collectively contribute to a more resilient and economically viable supply chain for high-value pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the complete removal of expensive transition metal catalysts and stoichiometric oxidants from the reaction recipe. In traditional synthesis, these reagents can account for a significant portion of the raw material costs, and their removal often requires additional processing steps like filtration through celite or treatment with metal scavengers. By using electricity as the reagent, the process converts a variable chemical cost into a fixed, relatively low utility cost. Furthermore, the high atom economy of the electrochemical cyclization minimizes the generation of byproduct salts, reducing the load on wastewater treatment systems and lowering the environmental compliance costs associated with hazardous waste disposal.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals rather than specialized catalysts significantly de-risks the supply chain. The starting materials, such as substituted naphthoquinones and phenylacetylenes, are widely available from multiple global suppliers, ensuring continuity of supply even during market disruptions. The electrolyte, tetrabutylammonium acetate, is a common industrial chemical with a stable supply base, unlike bespoke ligands that may have long lead times or single-source dependencies. This diversification of the supplier base enhances procurement leverage and reduces the risk of production stoppages due to raw material shortages, ensuring reliable delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: Electrochemical reactions are inherently scalable, particularly when adapted to continuous flow reactors, which offer superior heat and mass transfer compared to batch vessels. This scalability facilitates the commercial scale-up of complex pharmaceutical intermediates from kilogram to tonne quantities without the need for massive reactor vessels or complex safety systems for handling energetic oxidants. Moreover, the "green" nature of the process, characterized by the absence of heavy metals and reduced solvent usage, aligns perfectly with the increasingly strict environmental regulations faced by chemical manufacturers. This compliance advantage simplifies the permitting process for new production lines and enhances the corporate sustainability profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical technology. These answers are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on the practical aspects of the synthesis. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Does this electrochemical method leave heavy metal residues in the final API intermediate?
A: No. Unlike traditional transition-metal catalyzed methods, this electrochemical protocol uses electrons as the primary oxidant, completely eliminating the need for palladium, copper, or other transition metal catalysts, thereby ensuring a metal-free impurity profile.
Q: What is the substrate scope for this electrochemical cyclization?
A: The method demonstrates excellent functional group tolerance, successfully accommodating substrates with halogens (Cl, Br), esters, ethers, Boc-protected amines, and heterocycles like thiophene and pyridine without compromising yield.
Q: Is this process suitable for large-scale commercial production?
A: Yes. The reaction operates under mild conditions (room temperature, atmospheric pressure) using simple undivided cells, making it highly amenable to scale-up and continuous flow electrochemical processing for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aza-Anthraquinone Supplier
As the demand for sustainable and cost-effective synthetic routes grows, partnering with a CDMO that possesses deep expertise in advanced manufacturing technologies is crucial for success. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, combining rigorous scientific inquiry with industrial pragmatism to deliver superior chemical solutions. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the absence of metal residues and confirm structural integrity.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this electrochemical advantage for your next project. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this metal-free route for your specific target molecules. We encourage you to contact us today to discuss your specific COA data requirements and to receive comprehensive route feasibility assessments tailored to your production timelines and quality standards. Let us help you build a more sustainable and profitable supply chain for your critical pharmaceutical intermediates.
