Scalable One-Pot Synthesis of Anthracene-Triazole Derivatives for Advanced Optoelectronic Applications
Scalable One-Pot Synthesis of Anthracene-Triazole Derivatives for Advanced Optoelectronic Applications
The rapid evolution of the organic electronics sector demands intermediates that combine structural rigidity with tunable electronic properties, a challenge addressed directly by the innovative methodology disclosed in patent CN103772303A. This intellectual property introduces a robust synthetic route for 1-[9-(1H-1,2,4-triazol-1-yl)anthracen-10-yl]-1H-1,2,4-triazole, a novel anthracene nucleus-triazole compound with significant potential in the realm of photoelectric materials. Unlike conventional approaches that often suffer from low atom economy or require hazardous reagents, this patent outlines a streamlined 'one-pot' strategy leveraging copper oxide catalysis to achieve high yields with minimal environmental impact. For R&D directors and procurement specialists alike, this represents a pivotal shift towards more sustainable and economically viable manufacturing of complex heterocyclic systems. The ability to produce such sophisticated molecular architectures through a simplified process not only enhances supply chain reliability but also opens new avenues for the development of next-generation luminescent agents and dye sensitizers.
![Chemical structure of 1-[9-(1H-1,2,4-triazol-1-yl)anthracen-10-yl]-1H-1,2,4-triazole showing the anthracene core flanked by two triazole rings](/insights/img/anthracene-triazole-synthesis-supplier-optoelectronic-20260309013851-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the functionalization of polycyclic aromatic hydrocarbons like anthracene with nitrogen-containing heterocycles has been fraught with synthetic challenges that hinder large-scale commercialization. Classical nucleophilic substitution reactions often require extremely harsh conditions, such as high temperatures and strong bases, which can lead to the degradation of the sensitive anthracene backbone or the formation of unwanted regioisomers. Furthermore, many standard cross-coupling protocols rely on expensive homogeneous palladium catalysts that necessitate rigorous downstream purification to remove trace metal contaminants, a critical requirement for electronic grade materials. These multi-step procedures typically involve the isolation of intermediates, resulting in significant material loss, increased solvent consumption, and prolonged production cycles. For supply chain managers, these inefficiencies translate into higher costs of goods sold (COGS) and unpredictable lead times, making it difficult to secure a consistent supply of high-purity intermediates needed for mass production of optoelectronic devices.
The Novel Approach
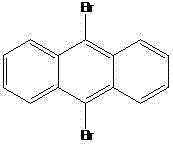
In stark contrast to these legacy methods, the technology described in CN103772303A utilizes a direct, copper-catalyzed C-N bond formation strategy that dramatically simplifies the synthetic landscape. By employing 9,10-dibromoanthracene as the starting scaffold and reacting it directly with 1,2,4-triazole in the presence of cupric oxide and potassium carbonate, the inventors have achieved a highly efficient double arylation in a single reaction vessel. This 'one-pot' methodology eliminates the need for intermediate isolation and reduces the overall number of unit operations, thereby minimizing waste generation and energy consumption. The use of inexpensive and readily available copper oxide as a heterogeneous catalyst not only lowers the raw material costs but also facilitates easier catalyst removal compared to soluble metal complexes. This approach ensures that the final product retains the structural integrity of the anthracene core while successfully installing the triazole moieties, resulting in a material with superior purity profiles suitable for demanding applications in display technologies.
Mechanistic Insights into Copper-Catalyzed N-Arylation
The core of this synthetic breakthrough lies in the mechanistic pathway of the copper-catalyzed Ullmann-type coupling, which facilitates the formation of carbon-nitrogen bonds between the electron-deficient anthracene ring and the triazole nucleophile. In this system, cupric oxide acts as a precatalyst that is likely reduced in situ to active copper(I) species, which then coordinate with the nitrogen atoms of the triazole ring to form a reactive copper-triazole complex. This complex subsequently undergoes oxidative addition with the carbon-bromine bonds of the 9,10-dibromoanthracene, followed by reductive elimination to forge the stable C-N linkage. The presence of potassium carbonate serves a dual purpose: it acts as a base to deprotonate the triazole, enhancing its nucleophilicity, and it helps to neutralize the hydrogen bromide byproduct generated during the coupling, driving the equilibrium towards product formation. Understanding this catalytic cycle is crucial for process chemists aiming to optimize reaction kinetics and minimize side reactions such as homocoupling or debromination.
Furthermore, the control of impurities in this specific transformation is paramount for ensuring the performance of the final optoelectronic material. The patent highlights the importance of maintaining specific molar ratios, particularly an excess of triazole and base, to ensure complete conversion of the dibromo-anthracene starting material and prevent the accumulation of mono-substituted intermediates. The crystalline nature of the final product, as evidenced by the detailed X-ray diffraction data provided in the patent, suggests a high degree of molecular order and purity, which is essential for achieving consistent fluorescence properties. The rigid planar structure of the anthracene core combined with the electron-withdrawing nature of the triazole rings creates a conjugated system that is highly favorable for charge transport and light emission. By meticulously controlling reaction parameters such as temperature (optimized between 100°C and 150°C) and time, manufacturers can effectively suppress the formation of oligomeric byproducts, ensuring a clean impurity profile that meets the stringent specifications required by the semiconductor and display industries.
How to Synthesize 1-[9-(1H-1,2,4-triazol-1-yl)anthracen-10-yl]-1H-1,2,4-triazole Efficiently
To implement this synthesis effectively in a pilot or production setting, precise adherence to the stoichiometric ratios and thermal profiles outlined in the patent is essential for reproducibility. The process begins with the careful charging of 9,10-dibromoanthracene, 1,2,4-triazole, potassium carbonate, and cupric oxide into a reactor equipped with efficient agitation and temperature control systems. Dimethylformamide (DMF) is utilized as the polar aprotic solvent to solubilize the reactants and facilitate the catalytic cycle, though alternative high-boiling polar solvents could be evaluated for green chemistry improvements. The reaction mixture is then heated to the target temperature range, where it is maintained under vigorous stirring to ensure homogeneous heat transfer and mass transfer throughout the heterogeneous catalyst bed. Following the completion of the reaction, indicated by the consumption of the starting material, the workup involves a simple filtration to remove the solid copper catalyst and inorganic salts, followed by precipitation of the product upon addition of water.
- Charge a reaction vessel with 9,10-dibromoanthracene, 1,2,4-triazole, potassium carbonate, and cupric oxide in a specific molar ratio.
- Add polar solvent DMF and heat the mixture to a temperature range between 80°C and 200°C under stirring.
- Maintain reaction conditions for 12 to 120 hours, then cool, filter, and precipitate the product using water to isolate the high-purity crystal.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this one-pot synthesis route offers profound advantages that extend far beyond the laboratory bench, directly impacting the bottom line and operational resilience of chemical manufacturing enterprises. The elimination of multiple isolation and purification steps inherent in traditional multi-step syntheses results in a drastically simplified process flow, which translates to reduced capital expenditure on equipment and lower operational overheads. For procurement managers, the reliance on commodity chemicals such as 9,10-dibromoanthracene, triazole, and copper oxide ensures a stable and cost-effective supply of raw materials, mitigating the risks associated with sourcing exotic or proprietary reagents. The robustness of the reaction conditions allows for flexibility in scaling, enabling manufacturers to ramp up production volumes rapidly in response to market demand without the need for extensive process re-engineering. This agility is a critical competitive advantage in the fast-paced electronics industry, where time-to-market for new materials can determine commercial success.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the high atom efficiency and the use of non-precious metal catalysts. By replacing expensive palladium systems with abundant copper oxide, the direct material costs are significantly lowered, while the heterogeneous nature of the catalyst allows for potential recovery and reuse, further enhancing cost savings. Additionally, the simplified workup procedure reduces the volume of solvents and consumables required for purification, leading to substantial reductions in waste disposal costs and environmental compliance burdens. These cumulative efficiencies allow for a more competitive pricing structure for the final anthracene-triazole derivative, making it an attractive option for cost-sensitive applications in consumer electronics and lighting.
- Enhanced Supply Chain Reliability: The simplicity of the synthetic route inherently reduces the number of potential failure points in the manufacturing process, thereby increasing the overall reliability of supply. Since the reaction does not require moisture-sensitive reagents or inert atmosphere conditions as strictly as some organometallic couplings, it can be performed in standard glass-lined or stainless steel reactors found in most fine chemical facilities. This compatibility with existing infrastructure minimizes the need for specialized equipment investments and shortens the timeline for technology transfer from lab to plant. Consequently, suppliers can offer more consistent lead times and guarantee continuity of supply, which is vital for downstream customers who rely on just-in-time inventory models for their own production lines.
- Scalability and Environmental Compliance: The 'one-pot' design is intrinsically scalable, as the reaction kinetics and heat management issues are easier to control in a single vessel compared to a sequence of batch operations. The reduction in solvent usage and waste generation aligns perfectly with modern green chemistry principles and increasingly stringent environmental regulations. By minimizing the discharge of halogenated byproducts and heavy metal residues, manufacturers can operate with a smaller environmental footprint and reduced regulatory risk. This sustainability profile is becoming a key differentiator in B2B negotiations, as global corporations seek to partner with suppliers who demonstrate a commitment to responsible manufacturing practices and circular economy principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this anthracene-triazole derivative, based on the specific data and embodiments provided in the patent literature. Understanding these details is essential for technical teams evaluating the feasibility of integrating this material into their existing product portfolios or R&D pipelines. The answers provided reflect the optimized conditions and observed outcomes reported in the intellectual property, serving as a reliable foundation for further process development and scale-up activities.
Q: What are the critical reaction parameters for maximizing yield in this anthracene-triazole synthesis?
A: The patent specifies a molar ratio of 9,10-dibromoanthracene to triazole to potassium carbonate to cupric oxide of approximately 2:10:30:1, with temperatures maintained between 100°C and 150°C for optimal conversion.
Q: Why is the one-pot method preferred over traditional multi-step arylation processes?
A: The one-pot approach significantly reduces operational complexity and solvent usage by combining the coupling and crystallization steps, eliminating the need for intermediate isolation and purification stages.
Q: What are the primary industrial applications for this specific anthracene derivative?
A: This compound exhibits strong fluorescence properties with an excitation wavelength of 365 nm and emission at 558 nm, making it highly suitable for use as a luminescent material in OLEDs and other photoelectric devices.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-[9-(1H-1,2,4-triazol-1-yl)anthracen-10-yl]-1H-1,2,4-triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced anthracene derivatives in the next generation of optoelectronic devices and are fully committed to supporting our partners in realizing this vision. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 1-[9-(1H-1,2,4-triazol-1-yl)anthracen-10-yl]-1H-1,2,4-triazole meets the exacting standards required for high-performance display and lighting applications. Our dedication to quality assurance means that you can rely on us not just as a vendor, but as a strategic partner dedicated to the success of your final products.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how optimizing this supply chain can reduce your overall manufacturing expenses. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and innovation in your organization. Let us collaborate to bring high-quality, cost-effective photoelectric materials to the global market.
