Advanced Neutral Hydrogenation Strategy for High-Purity 2-(S)-Amino-4-Arylbutyric Acid Production
Introduction to Advanced Synthetic Methodologies
The pharmaceutical industry continuously seeks robust and scalable pathways for producing critical chiral intermediates, particularly those serving as building blocks for Angiotensin Converting Enzyme Inhibitors (ACEIs) and multiple sclerosis treatments. Patent CN115784916B introduces a transformative approach for preparing 2-(S)-amino-4-arylbutyric acid compounds, addressing long-standing challenges associated with equipment corrosion and harsh reaction conditions. This technology leverages a strategic indirect reduction pathway that bypasses the necessity for strong acidic media during the critical hydrogenation step. By converting the ketone precursor into a lactone intermediate prior to hydrogenolysis, the process significantly lowers the activation energy required for C-O bond cleavage. This innovation not only enhances the safety profile of the manufacturing process but also aligns perfectly with the rigorous demands of modern green chemistry initiatives. For R&D directors and procurement specialists, understanding this shift from direct acidic reduction to neutral lactone hydrogenolysis is crucial for optimizing supply chains and reducing overall production costs in the competitive landscape of pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-(S)-amino-4-arylbutyric acid derivatives has relied heavily on direct catalytic hydrogenation of the carbonyl group to a methylene group, a transformation that presents significant engineering hurdles. As illustrated in prior art such as US 005438150 A, these conventional routes typically mandate the use of mixed strong acid solvents, including sulfuric acid and glacial acetic acid, to facilitate the reaction mechanism. The reliance on such aggressive chemical environments creates a severe bottleneck for industrial scale-up, as standard stainless steel hydrogenation kettles are incompatible with these corrosive media. Consequently, manufacturers are forced to invest in specialized Hastelloy reactors, which drastically inflate capital expenditure and complicate maintenance schedules. Furthermore, the handling of large volumes of strong acids introduces substantial safety risks and generates hazardous waste streams that require costly treatment protocols. These factors collectively render traditional acidic hydrogenation processes economically inefficient and operationally rigid for high-volume API intermediate production.
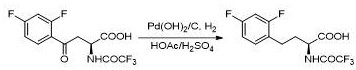
The Novel Approach
In stark contrast to these legacy methods, the novel methodology disclosed in the patent employs a clever three-step sequence that circumvents the need for corrosive acids during the final reduction stage. The process begins with the selective reduction of the ketone to a hydroxy intermediate, followed by a ring-closure esterification to form a stable arylbutyrolactone. This lactone structure is key; it possesses a lower bond dissociation energy for the C-O bond compared to the direct reduction of a benzylic alcohol, allowing hydrogenolysis to proceed smoothly under neutral conditions. By utilizing a palladium catalyst in a neutral alcoholic solvent, the reaction avoids the formation of oxonium ions that typically necessitate strong acid catalysis. This shift enables the use of standard industrial equipment, eliminates acid waste, and maintains excellent stereochemical control. The strategic insertion of the lactonization step effectively decouples the reduction chemistry from the corrosive constraints of the past, offering a streamlined pathway for cost reduction in pharmaceutical manufacturing.
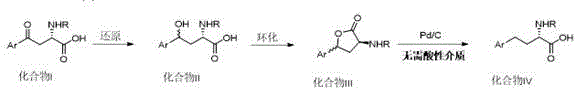
Mechanistic Insights into Neutral Lactone Hydrogenolysis
The core mechanistic advantage of this technology lies in the thermodynamic and kinetic facilitation provided by the lactone ring structure. In traditional direct reduction pathways, the conversion of a benzylic alcohol to a methylene group requires protonation of the hydroxyl group to form a good leaving group (water), a step that demands a high concentration of protons (strong acid). However, in the patented process, the intramolecular esterification creates a cyclic ester where the carbonyl carbon is more electrophilic and the C-O bond is activated for cleavage by the metal catalyst surface without external protonation. The palladium catalyst facilitates the adsorption of hydrogen and the subsequent oxidative addition into the C-O bond of the lactone ring. This surface-mediated reaction proceeds efficiently at mild temperatures ranging from 0°C to 45°C, preventing thermal degradation or racemization of the sensitive chiral center at the alpha-position. The absence of strong Bronsted acids ensures that the amine protecting groups (such as trifluoroacetyl or Fmoc) remain stable, thereby preserving the integrity of the molecule throughout the synthesis.
From an impurity control perspective, the neutral conditions offer a distinct advantage by suppressing acid-catalyzed side reactions that often plague traditional syntheses. In acidic media, there is a heightened risk of epimerization at the chiral alpha-carbon, which would compromise the optical purity of the final product—a critical quality attribute for pharmaceutical applications. Additionally, strong acids can promote polymerization or decomposition of the aryl ring substituents, leading to complex impurity profiles that are difficult to purge. By maintaining a neutral pH environment during the hydrogenation step, the process minimizes these degradation pathways, resulting in a cleaner crude product with higher assay values. This inherent purity reduces the burden on downstream purification units, such as crystallization or chromatography, further enhancing the overall process efficiency and yield consistency required for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 2-(S)-Amino-4-Arylbutyric Acid Efficiently
The implementation of this synthetic route involves precise control over stoichiometry and reaction parameters to maximize yield and purity. The process initiates with the reduction of the keto-acid starting material using sodium borohydride, where temperature control between -25°C and 30°C is vital to prevent over-reduction or side reactions. Following the isolation of the hydroxy intermediate, the cyclization step utilizes trifluoroacetic acid, which acts as both a solvent and a catalyst for lactone formation, typically completing within 1 to 24 hours depending on the specific substrate substituents. The final hydrogenation step is remarkably robust, tolerating various palladium catalysts such as Pd/C or Pd(OH)2/C, and can be conducted in common solvents like methanol or ethanol. Detailed operational parameters, including specific molar ratios and workup procedures, are critical for reproducing the high yields reported in the patent examples. For a comprehensive breakdown of the standardized operating procedures and safety guidelines, please refer to the technical guide below.
- Reduce the starting 2-(S)-amino-4-oxo-arylbutyric acid using sodium borohydride in solvents like THF or methanol at temperatures between -25°C and 30°C to form the hydroxy intermediate.
- Perform ring-closure esterification on the hydroxy intermediate using trifluoroacetic acid at 0-40°C to generate the corresponding arylbutyrolactone derivative.
- Conduct hydrogenation of the lactone under neutral conditions using a palladium catalyst (Pd/C or Pd(OH)2/C) in alcoholic solvents to yield the final 2-(S)-amino-4-arylbutyric acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this neutral hydrogenation technology represents a significant opportunity to optimize operational expenditures and mitigate supply risks. The elimination of strong corrosive acids from the critical hydrogenation step removes the dependency on exotic metallurgy for reactor construction, allowing facilities to utilize standard stainless steel equipment that is widely available and easier to maintain. This compatibility with existing infrastructure drastically reduces the barrier to entry for contract manufacturing organizations and internal production sites, ensuring a more resilient and flexible supply base. Furthermore, the simplified waste profile, devoid of large volumes of spent strong acid, lowers environmental compliance costs and streamlines the disposal process, contributing to a more sustainable manufacturing footprint. These operational efficiencies translate directly into improved margin structures and more competitive pricing for downstream customers seeking reliable sources of high-value intermediates.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the substantial reduction in capital and operational costs associated with equipment and waste management. By avoiding the need for Hastelloy-lined reactors, manufacturers can leverage existing stainless steel assets, deferring heavy capital investments. Additionally, the neutral conditions eliminate the costs related to neutralizing and treating large quantities of acidic effluent, which is a significant expense in traditional processes. The milder reaction conditions also tend to extend the lifespan of the heterogeneous palladium catalyst, allowing for potential recycling or reduced loading rates, which further drives down the cost of goods sold. These cumulative savings create a robust economic model that supports cost reduction in API manufacturing without compromising on quality.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of handling hazardous materials and the limited availability of specialized corrosion-resistant equipment. This novel process mitigates these risks by utilizing common reagents and standard reaction vessels, making it easier for multiple suppliers to qualify and produce the material. The robustness of the chemistry under mild conditions reduces the likelihood of batch failures due to equipment corrosion or thermal runaway, ensuring consistent delivery schedules. Moreover, the flexibility in solvent choice (methanol, ethanol, THF) allows manufacturers to adapt to raw material availability fluctuations, thereby strengthening the overall resilience of the supply chain against market volatility and logistical disruptions.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this methodology is inherently designed for safe expansion. The absence of strong acids reduces the risk of catastrophic containment failures, simplifying safety audits and regulatory approvals for larger production scales. From an environmental standpoint, the process generates less hazardous waste, aligning with increasingly stringent global regulations on chemical discharge and sustainability. This compliance advantage facilitates smoother operations across different geographic regions, enabling manufacturers to establish multi-site production networks that ensure reducing lead time for high-purity pharmaceutical intermediates while meeting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for their specific product portfolios. The answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a factual basis for decision-making. Whether you are concerned about equipment compatibility, stereochemical integrity, or waste management, these insights clarify how the neutral hydrogenation strategy outperforms legacy acidic methods in real-world production scenarios.
Q: Why is the neutral hydrogenation method superior to traditional acidic reduction for this compound?
A: Traditional methods require strong Bronsted acids like sulfuric or hydrochloric acid to facilitate the hydrogenolysis of benzyl alcohol intermediates, which necessitates expensive Hastelloy equipment due to corrosion risks. The novel method utilizes a lactone intermediate that undergoes hydrogenolysis under neutral conditions, allowing the use of standard stainless steel reactors and significantly lowering capital expenditure.
Q: What are the typical purity levels achievable with this lactonization-hydrogenation route?
A: According to the patent data, the process consistently achieves high purity levels, often exceeding 95% to 98% without the need for complex purification steps. The mild reaction conditions minimize side reactions and racemization, ensuring the stereochemical integrity of the 2-(S) configuration is maintained throughout the synthesis.
Q: Can this process be scaled for industrial production of ACEI inhibitors?
A: Yes, the process is specifically designed for industrial scalability. By eliminating the need for specialized acid-corrosion-resistant materials and operating under mild temperatures (0-45°C), the method simplifies safety protocols and equipment requirements, making it highly suitable for large-scale commercial manufacturing of intermediates like those for Enalapril.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(S)-Amino-4-Arylbutyric Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the fast-paced pharmaceutical industry. Our team of expert chemists has extensively analyzed the technological advantages presented in patent CN115784916B and is fully prepared to implement this advanced neutral hydrogenation strategy for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with versatile hydrogenation capabilities and rigorous QC labs dedicated to maintaining stringent purity specifications for every batch of 2-(S)-amino-4-arylbutyric acid produced. We understand that consistency and quality are non-negotiable for your downstream synthesis of ACE inhibitors and other vital therapeutics.
We invite you to collaborate with us to leverage these process improvements for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this corrosion-free manufacturing method for your projects. We encourage you to contact our technical procurement team today to request specific COA data from our pilot runs and discuss route feasibility assessments tailored to your unique volume requirements. Let us help you secure a sustainable, cost-effective, and high-quality supply of this essential chiral building block.
