Advanced Asymmetric Hydrogenation for Commercial Scale-up of Complex Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access high-value chiral building blocks with uncompromising optical purity. Patent CN112047856B introduces a groundbreaking advancement in this domain by detailing a highly efficient preparation method for chiral alpha-amidoaldehydes. This technology leverages asymmetric catalytic hydrogenation under the influence of specialized bisphosphine-rhodium complexes to transform alpha-dehydroamidoaldehydes into their corresponding chiral counterparts. The significance of this innovation cannot be overstated, as chiral alpha-amidoaldehyde structures serve as critical precursors for a vast array of physiologically active molecules and drug candidates. By addressing the longstanding challenges of racemization and low efficiency associated with conventional synthetic routes, this patent provides a viable pathway for the reliable pharmaceutical intermediate supplier to deliver superior quality materials. The process is characterized by its remarkable versatility, accommodating a broad spectrum of substituents on the aromatic ring, including halogens, alkyl groups, and electron-withdrawing moieties, thereby expanding the chemical space accessible to medicinal chemists.
Furthermore, the operational simplicity of this method aligns perfectly with the demands of modern green chemistry initiatives. The reaction proceeds under relatively mild conditions, utilizing hydrogen gas as the reducing agent, which generates no toxic byproducts, thus enhancing the environmental profile of the synthesis. For R&D directors focused on impurity profiles, the high stereoselectivity reported in the patent ensures that the resulting products possess minimal levels of unwanted enantiomers, simplifying downstream purification processes. This level of control is essential when synthesizing intermediates for potent APIs where even trace impurities can have significant biological consequences. The ability to tune the stereochemistry by simply selecting the appropriate enantiomer of the chiral ligand offers a strategic advantage in the rapid development of drug candidates, allowing for the swift generation of both R and S configured intermediates as needed for biological screening.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of chiral alpha-amidoaldehydes has been fraught with significant technical hurdles that hindered their widespread adoption in industrial settings. Traditional approaches, such as the asymmetric hydroformylation of alkenyl amines or the direct alpha-amination of aldehydes, often suffer from poor atom economy and require complex multi-step sequences. These legacy methods frequently necessitate the use of harsh reaction conditions, including strong acids or bases, which pose a severe threat to the stability of the sensitive aldehyde functionality. Under such aggressive environments, the chiral center is prone to racemization, leading to a erosion of optical purity that is difficult and costly to rectify in later stages. Moreover, the catalysts employed in older methodologies often lack the necessary turnover numbers, requiring high loadings of expensive metals which drastically inflates the production cost. The combination of low yields, difficult purification requirements, and safety concerns associated with hazardous reagents has made the commercial scale-up of complex pharmaceutical intermediates via these routes economically unfeasible for many manufacturers.
The Novel Approach
In stark contrast, the novel approach detailed in patent CN112047856B revolutionizes the synthesis landscape by employing a direct asymmetric hydrogenation strategy. This method bypasses the need for unstable intermediates and harsh reagents, instead utilizing a sophisticated bisphosphine-rhodium catalyst system that operates with exceptional precision. The reaction conditions are remarkably mild, typically conducted at temperatures ranging from 0 to 50 degrees Celsius and hydrogen pressures between 1 and 100 bar, which preserves the integrity of the sensitive functional groups present in the molecule. This gentle approach effectively eliminates the risk of racemization, ensuring that the high enantiomeric excess achieved during the catalytic cycle is retained in the final isolated product. The versatility of this system is further demonstrated by its compatibility with a wide array of organic solvents, including dichloromethane, ethyl acetate, and alcohols, providing process engineers with the flexibility to optimize solubility and workup procedures. By streamlining the synthesis into a single catalytic step with high conversion rates, this new methodology offers a compelling solution for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Bisphosphine-Rhodium Catalyzed Asymmetric Hydrogenation
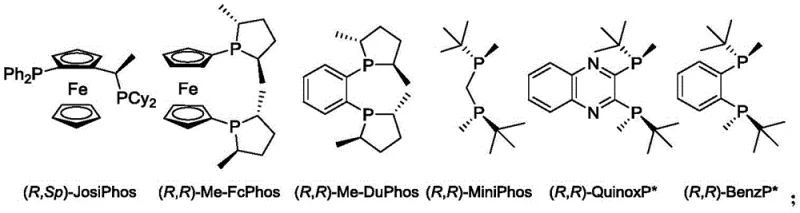
The core of this technological breakthrough lies in the intricate design of the catalytic system, specifically the synergy between the rhodium metal center and the chiral bisphosphine ligands. The patent highlights a series of advanced ligands, including (R,R)-BenzP*, (R,R)-QuinoxP*, and various Josiphos derivatives, which create a highly defined chiral environment around the active metal site. When the alpha-dehydroamidoaldehyde substrate coordinates to the rhodium complex, the bulky substituents on the ligand framework exert significant steric influence, directing the approach of the hydrogen molecule to one specific face of the double bond. This facial selectivity is the fundamental driver of the observed high enantioselectivity, allowing for the preferential formation of either the R or S configuration depending on the chirality of the ligand employed. The electronic properties of the ligands also play a crucial role in modulating the reactivity of the rhodium center, facilitating the oxidative addition of hydrogen and the subsequent migratory insertion steps that constitute the catalytic cycle. Understanding these mechanistic nuances is vital for R&D teams aiming to further optimize the process for specific substrate classes or to develop next-generation catalysts with even higher turnover frequencies.
Beyond the primary catalytic cycle, the robustness of the system against impurity formation is a key attribute for industrial application. The mild conditions prevent side reactions such as aldol condensation or over-reduction of the aldehyde group to the corresponding alcohol, which are common pitfalls in aldehyde chemistry. The stability of the catalyst itself, even at low loadings down to a molar ratio of 1/20000, suggests a long catalyst lifetime and resistance to deactivation by potential poisons in the reaction mixture. This durability translates directly into consistent product quality batch after batch, a critical factor for maintaining stringent purity specifications required by regulatory bodies. Furthermore, the ability to recover and potentially recycle the rhodium catalyst, given its high value, adds another layer of economic and environmental benefit to the process. The precise control over the stereochemical outcome ensures that the impurity profile is dominated by the desired enantiomer, simplifying the analytical burden and reducing the need for extensive chiral chromatography during purification.
How to Synthesize Chiral Alpha-Amidoaldehyde Efficiently
The practical implementation of this synthesis route is designed to be straightforward and adaptable to existing hydrogenation infrastructure found in most pilot and production plants. The process begins with the careful selection of the appropriate bisphosphine-rhodium catalyst based on the specific substrate structure to maximize enantioselectivity. Once the catalyst and substrate are charged into the reactor, the system is purged and pressurized with hydrogen, initiating the reduction. The reaction progress can be monitored using standard analytical techniques such as HPLC, with completion typically achieved within 1 to 48 hours depending on the scale and specific conditions. Following the reaction, the workup involves simple solvent removal and, if necessary, column chromatography or crystallization to isolate the pure chiral product. The detailed standardized synthesis steps see the guide below for specific parameters regarding solvent choice, temperature control, and catalyst loading optimization.
- Prepare the reaction vessel by charging the alpha-dehydroamidoaldehyde substrate and the bisphosphine-rhodium catalyst complex, such as [Rh((R,R)-BenzP*)(cod)]SbF6, ensuring a molar ratio between 1/100 and 1/20000.
- Introduce an organic solvent like dichloromethane or ethyl acetate under an inert atmosphere, followed by pressurizing the system with hydrogen gas to a range of 1 to 100 bar.
- Maintain the reaction mixture at a temperature between 0 and 50 degrees Celsius with vigorous stirring for 1 to 48 hours, then isolate the chiral product via concentration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers transformative benefits that extend far beyond the laboratory bench. The shift towards this catalytic hydrogenation method addresses several critical pain points associated with the sourcing of high-purity chiral intermediates. By eliminating the need for stoichiometric chiral auxiliaries or resolution steps, the process inherently reduces the number of unit operations required, leading to a drastically simplified manufacturing workflow. This simplification not only accelerates the production timeline but also minimizes the consumption of raw materials and solvents, contributing to substantial cost savings in the overall production budget. The reliance on hydrogen gas as the reductant is particularly advantageous from a supply chain perspective, as it is a commodity chemical with a stable and secure global supply, unlike specialized chiral reagents that may be subject to market volatility or geopolitical disruptions.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the exceptional efficiency of the catalyst system. With the ability to operate at catalyst loadings as low as 1/20000 molar ratio, the consumption of expensive rhodium and chiral ligands is minimized to negligible levels per kilogram of product. This drastic reduction in catalyst cost, combined with the high yields often exceeding 90%, ensures a highly favorable cost of goods sold (COGS). Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, further lowering the operational expenditure. The elimination of complex protection and deprotection steps often required in alternative syntheses also contributes to a leaner, more cost-effective process flow that maximizes resource utilization.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions enhances the reliability of the supply chain by reducing the risk of batch failures. Traditional methods involving harsh chemicals are prone to variability due to slight deviations in temperature or reagent quality, whereas this hydrogenation process is forgiving and consistent. The use of common, commercially available solvents like dichloromethane and ethyl acetate ensures that raw material availability is never a bottleneck. Furthermore, the high stability of the product under the reaction conditions means that storage and transportation risks are mitigated, ensuring that the delivered material meets the specified quality standards upon arrival. This reliability is crucial for maintaining continuous manufacturing schedules and meeting the tight deadlines of drug development projects.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this method aligns perfectly with modern sustainability goals. The atom economy of the hydrogenation reaction is near perfect, as the only byproduct is the saturated bond, generating minimal waste streams that require treatment. The absence of heavy metal waste from stoichiometric reagents simplifies wastewater treatment and reduces the environmental footprint of the facility. Scalability is inherent in the design, as hydrogenation reactors are standard equipment in the fine chemical industry, allowing for seamless transition from gram-scale R&D to ton-scale commercial production. This ease of scale-up ensures that supply can be rapidly ramped up to meet market demand without the need for significant capital investment in new specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of the synthesis. Understanding these details helps stakeholders make informed decisions about integrating this method into their existing production pipelines or R&D programs. The answers reflect the high standards of performance and reliability that characterize this innovative approach to chiral synthesis.
Q: What represents the primary advantage of this rhodium-catalyzed method over traditional hydroformylation?
A: The primary advantage lies in the exceptional atom economy and mild reaction conditions. Unlike traditional methods that often require harsh acidic or basic environments leading to racemization, this asymmetric hydrogenation operates at ambient temperatures (25-30°C) and achieves enantiomeric excess values up to 99.9%, ensuring superior optical purity without degradation.
Q: Can this synthesis method be scaled for industrial production of drug intermediates?
A: Yes, the process is highly scalable due to the use of stable catalysts and common organic solvents. The patent demonstrates successful conversion across a wide range of substrates with yields reaching 99%, indicating robust performance suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: How does the catalyst loading impact the overall production cost?
A: The method allows for extremely low catalyst loading, with molar ratios as low as 1/20000 (catalyst to substrate). This drastic reduction in precious metal usage significantly lowers the raw material cost per kilogram of product, making the process economically viable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Alpha-Amidoaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality chiral intermediates to drive your drug discovery and development programs forward. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging advanced technologies such as the rhodium-catalyzed asymmetric hydrogenation described in CN112047856B, we can offer you a competitive edge through superior product quality and optimized manufacturing processes. Our dedication to technical excellence ensures that every batch of chiral alpha-amidoaldehyde we supply is ready for immediate use in your most demanding synthetic applications.
We invite you to collaborate with us to explore how this cutting-edge synthesis method can benefit your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume and purity requirements, demonstrating the tangible economic advantages of switching to this efficient route. Please contact us today to request specific COA data and route feasibility assessments for your target molecules. Let us be your partner in transforming complex chemical challenges into commercial successes, ensuring a secure and efficient supply of the critical building blocks your business depends on.
