Advanced Synthetic Route for High-Purity Pyrrole Derivatives and Commercial Scalability
Advanced Synthetic Route for High-Purity Pyrrole Derivatives and Commercial Scalability
The pharmaceutical and agrochemical industries constantly demand more efficient pathways to access complex heterocyclic scaffolds, particularly pyrrole derivatives which serve as critical building blocks for bioactive molecules. Patent CN102199154A discloses a novel and highly efficient method for the preparation of pyrrole-1,2-dicarboxylic acid and its derivatives, addressing long-standing challenges in regioselective substitution on the electron-rich pyrrole ring. This technology represents a significant leap forward for any reliable pharmaceutical intermediates supplier aiming to streamline the production of NK1 receptor antagonist candidates and related therapeutic agents. By utilizing inexpensive and easily available 2-pyrrole carboxylic acid ester compounds as raw materials, the process bypasses the need for complex transition metal catalysis often required for direct functionalization. The strategic sequence of bromination, decarboxylation, and subsequent N-alkylation offers a robust platform for generating diverse 2,3-disubstituted pyrroles with high fidelity.
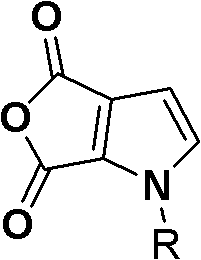
Furthermore, the versatility of this synthetic approach allows for the introduction of various R groups, including methyl, ethyl, phenyl, and halogenated variants, thereby expanding the chemical space available for medicinal chemistry campaigns. The ability to synthesize these structures through simple unit operations not only enhances process safety but also significantly lowers the barrier to entry for large-scale manufacturing. For procurement teams evaluating cost reduction in pharmaceutical intermediates manufacturing, this route offers a compelling alternative to traditional methods that often suffer from low yields or require hazardous reagents. The patent explicitly highlights the feasibility of preparing 2,3-pyrrole acid anhydrides, which are pivotal intermediates for further derivatization into high-value active pharmaceutical ingredients (APIs).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of adjacent disubstituted pyrrole compounds, especially 2,3-disubstituted derivatives, has been fraught with difficulties due to the inherent electronic properties of the pyrrole ring. The electron-rich nature of the pyrrole system makes it highly susceptible to electrophilic attack, yet controlling the regioselectivity to achieve specific 2,3-substitution patterns without forming complex mixtures of isomers remains a formidable challenge. Conventional methods often rely on harsh conditions or multi-step sequences that result in poor overall yields and generate substantial amounts of waste, complicating downstream purification and increasing the environmental footprint. Additionally, many existing routes depend on specialized starting materials that are not commercially available in bulk, leading to supply chain bottlenecks and inflated costs for the final intermediates. The lack of reported methods for efficiently accessing these specific substitution patterns has limited the exploration of pyrrole-based scaffolds in drug discovery programs.
The Novel Approach
In stark contrast to these limitations, the novel approach detailed in the patent utilizes a rational synthesis type that transforms simple esters into complex anhydrides through a logical progression of functional group manipulations. The process begins with the dibromination of the starting raw material S1, followed by saponification and decarboxylation to form the key intermediate S4, effectively setting the stage for subsequent functionalization. This strategy elegantly circumvents the regioselectivity issues by leveraging the directing effects of the existing substituents and the reactivity of the brominated positions. The subsequent steps involve N-alkylation followed by metal-mediated carboxylation, allowing for the precise installation of the second carboxylic acid moiety required for anhydride formation. By breaking down the synthesis into manageable unit reactions that are easy and simple to operate, the method ensures reproducibility and scalability.

Moreover, the flexibility of this route allows for the substitution of dibromination with di-iodination if specific downstream coupling reactions such as Suzuki or Stille couplings are desired, demonstrating the adaptability of the chemistry to different synthetic needs. The final dehydration step to form the anhydride is straightforward, utilizing acetic anhydride under reflux conditions to drive the equilibrium towards the desired cyclic product. This comprehensive approach not only solves the technical problem of synthesizing Formula (I) compounds but also provides a practical solution for industrial application where reliability and cost-efficiency are paramount.
Mechanistic Insights into Bromination and Decarboxylation Strategy
The core of this synthetic innovation lies in the meticulous control of electrophilic aromatic substitution and subsequent decarboxylation events. In the initial step, the starting material S1 is dissolved in an organic solvent such as tetrachloroethane in the presence of a catalytic amount of iodine, which activates the bromine source for selective electrophilic attack. The slow addition of liquid bromine at room temperature ensures that the reaction proceeds smoothly without excessive exotherms, leading to the formation of the dibrominated intermediate S2 with high selectivity. Following this, the ester group is hydrolyzed using sodium hydroxide solution at elevated temperatures (60-90°C), and the resulting carboxylic acid undergoes thermal decarboxylation. This decarboxylation is driven by the electron-withdrawing effect of the bromine atoms and the stability of the resulting aromatic system, yielding the dibromo-pyrrole intermediate S4 in excellent yields.
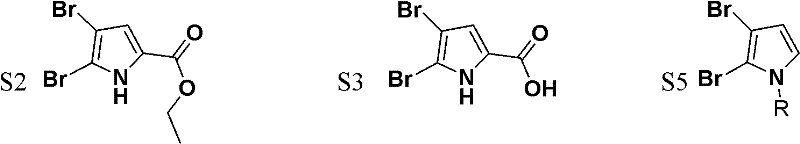
Subsequent mechanistic steps involve the deprotonation of the pyrrole nitrogen using a strong base like sodium hydride in DMF at 0°C, generating a nucleophilic pyrrolyl anion. This anion then attacks an alkyl halide (BrR) to install the N-substituent, forming intermediate S5. The introduction of the second carboxylic acid group is achieved through a directed ortho-metalation strategy using n-butyllithium at cryogenic temperatures (-78°C), followed by quenching with carbon dioxide gas. This lithiation-carboxylation sequence is highly specific, targeting the position adjacent to the nitrogen atom to establish the 1,2-dicarboxylic acid motif found in S6. Finally, the dehydration of S6 using acetic anhydride closes the ring to form the stable anhydride S7, completing the construction of the pyrrole-1,2-dicarboxylic acid core.
How to Synthesize Pyrrole-1,2-dicarboxylic Acid Anhydride Efficiently
The synthesis of these valuable intermediates requires precise control over reaction conditions, particularly temperature and stoichiometry, to maximize yield and minimize impurity formation. The patent outlines a clear protocol starting from ethyl pyrrole-2-carboxylate, moving through bromination, hydrolysis, alkylation, and finally cyclization. Each step has been optimized to ensure that the intermediate products can be isolated or carried forward with minimal loss of material. For research and development teams looking to implement this chemistry, understanding the nuances of the workup procedures, such as pH adjustment during acidification and solvent selection for extraction, is critical for success. The detailed standardized synthesis steps provided in the patent serve as a robust foundation for process development and optimization.
- Perform dibromination of ethyl pyrrole-2-carboxylate followed by saponification and decarboxylation to obtain the dibromo-pyrrole intermediate.
- Conduct N-alkylation on the pyrrole nitrogen using sodium hydride and an alkyl halide, followed by lithiation and carboxylation with CO2.
- Dehydrate the resulting pyrrole-1,2-dicarboxylic acid using acetic anhydride reflux to form the final pyrrole-1,2-dicarboxylic acid anhydride.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers distinct advantages that directly address the pain points of procurement managers and supply chain directors in the fine chemical sector. The reliance on commodity chemicals such as ethyl pyrrole-2-carboxylate, liquid bromine, and sodium hydride ensures that raw material sourcing is stable and not subject to the volatility associated with specialized reagents. Furthermore, the elimination of expensive transition metal catalysts, which often require rigorous removal steps to meet pharmaceutical purity standards, simplifies the downstream processing and reduces the overall cost of goods sold. The high yields reported in the patent embodiments, consistently exceeding 74% per step, translate to better material throughput and reduced waste disposal costs, contributing to a more sustainable and economically viable manufacturing process.
- Cost Reduction in Manufacturing: The process utilizes inexpensive starting materials and avoids the use of precious metal catalysts, which significantly lowers the direct material costs associated with production. By employing standard unit operations like reflux, extraction, and distillation, the method minimizes the need for specialized equipment, thereby reducing capital expenditure and operational overhead. The high efficiency of each reaction step ensures that less raw material is wasted, leading to substantial cost savings over the lifecycle of the product. Additionally, the simplified purification protocols reduce the consumption of solvents and stationary phases, further driving down the variable costs of manufacturing.
- Enhanced Supply Chain Reliability: Since the raw materials are widely available in the global chemical market, the risk of supply disruption is markedly reduced compared to routes relying on bespoke intermediates. The robustness of the reaction conditions means that the process can be transferred between different manufacturing sites with minimal re-validation, ensuring continuity of supply even in the face of regional disruptions. The ability to produce key intermediates like S7 in-house or through trusted partners strengthens the supply chain resilience against external shocks. This reliability is crucial for maintaining production schedules for downstream API manufacturing and meeting strict delivery timelines.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, as demonstrated by the use of standard reactors and common solvents that are compatible with large-scale production facilities. The avoidance of heavy metals and the generation of manageable waste streams facilitate compliance with increasingly stringent environmental regulations. The process design allows for the implementation of green chemistry principles, such as solvent recovery and recycling, which aligns with corporate sustainability goals. Scaling this process from kilogram to tonnage levels presents fewer technical hurdles than alternative methods, enabling rapid response to market demand fluctuations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel pyrrole synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the key advantages of this new pyrrole synthesis method?
A: The method utilizes inexpensive and readily available starting materials like ethyl pyrrole-2-carboxylate. It employs standard unit operations such as bromination and decarboxylation which are easy to operate and scale, achieving yields over 74% in each step.
Q: Can this route be scaled for commercial production of NK1 receptor antagonists?
A: Yes, the synthetic route is designed for efficiency and uses common reagents like liquid bromine, sodium hydride, and n-butyllithium. The process avoids exotic catalysts, making it highly suitable for commercial scale-up in pharmaceutical intermediate manufacturing.
Q: What is the purity profile of the final pyrrole anhydride product?
A: The patent describes purification via silica gel column chromatography yielding white solid compounds. The method allows for the production of high-purity intermediates essential for downstream coupling reactions in drug synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new pharmaceuticals and agrochemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of pyrrole derivatives meets the highest industry standards. Our commitment to technical excellence allows us to tackle complex synthetic challenges, such as the regioselective functionalization of heterocycles, with confidence and precision.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can accelerate your development timeline and optimize your supply chain efficiency.
