Scalable Electrochemical Oxidation of Diarylacetylenes for High-Purity Pharmaceutical Intermediates
Introduction to Green Electrochemical Oxidation Technology
The pharmaceutical and fine chemical industries are currently undergoing a paradigm shift towards sustainable manufacturing processes, driven by stringent environmental regulations and the need for cost-efficient production. Patent CN109518211B introduces a groundbreaking electrochemical synthesis method for aromatic acyl compounds, specifically targeting the oxidation of diarylacetylenes. This technology represents a significant departure from traditional stoichiometric oxidation methods, utilizing electrons as clean reagents to drive the transformation. By employing constant current electrolysis (C.C.E.) in a divided cell system, this process achieves high atom economy and operational simplicity. The core innovation lies in the ability to oxidize the carbon-carbon triple bond directly without the necessity for hazardous chemical oxidants or precious metal catalysts. This approach not only aligns with green chemistry principles but also offers a robust pathway for the reliable pharmaceutical intermediate supplier seeking to modernize their production capabilities. The versatility of this method is demonstrated through its applicability to a wide range of substituted substrates, making it a valuable asset for complex molecule synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aromatic acyl compounds, such as benzils, has relied heavily on transition metal-catalyzed oxidation or the use of strong stoichiometric oxidants. Traditional routes often involve catalysts based on palladium, ruthenium, or copper, which pose significant challenges regarding residual metal contamination in the final active pharmaceutical ingredients (APIs). Furthermore, these conventional methods frequently require harsh reaction conditions, including elevated temperatures and the use of toxic organic solvents, which complicate downstream processing and waste management. The reliance on external oxidants like potassium permanganate or chromium-based reagents generates substantial amounts of hazardous waste, increasing the environmental footprint and disposal costs for manufacturers. Additionally, the sensitivity of these catalytic systems to air and moisture often necessitates inert atmosphere operations, adding complexity and equipment costs to the manufacturing process. These limitations collectively restrict the scalability and economic viability of traditional synthetic routes for large-scale industrial applications.
The Novel Approach
In stark contrast, the electrochemical method disclosed in the patent offers a streamlined and environmentally benign alternative that addresses the critical pain points of conventional synthesis. By utilizing electricity as the driving force for oxidation, this novel approach completely eliminates the need for external chemical oxidants and transition metal catalysts. The reaction proceeds under mild conditions, typically at room temperature (25°C) and atmospheric pressure, thereby enhancing operational safety and reducing energy consumption. The use of a divided cell configuration ensures that the anodic oxidation of the alkyne is not compromised by cathodic reduction processes, leading to improved selectivity and yield. This method demonstrates exceptional compatibility with various functional groups, allowing for the synthesis of diverse aromatic acyl derivatives without the need for protecting group strategies. Consequently, this technology provides a scalable and cost-effective solution for the commercial scale-up of complex pharmaceutical intermediates, offering a distinct competitive advantage in terms of purity and process efficiency.
Mechanistic Insights into Electrochemical Alkyne Oxidation
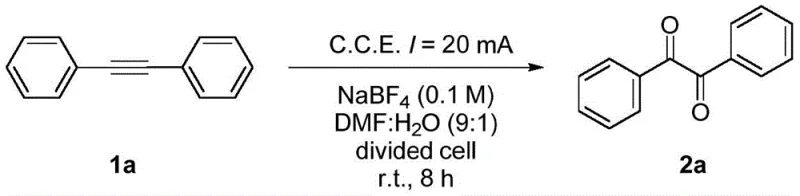
The mechanistic foundation of this synthesis relies on the anodic oxidation of the carbon-carbon triple bond in diarylacetylenes. In the presence of a supporting electrolyte such as sodium tetrafluoroborate (NaBF4), the application of a constant current facilitates the removal of electrons from the alkyne substrate at the anode surface. This electron transfer generates a reactive cationic intermediate, which subsequently undergoes nucleophilic attack by water molecules present in the solvent system. The specific solvent mixture of DMF and water (9:1) plays a crucial role in stabilizing the intermediates and providing the necessary oxygen source for the formation of the carbonyl groups. Unlike chemical oxidation where the oxidant strength is fixed, electrochemical potential can be finely tuned by adjusting the current density, allowing for precise control over the reaction pathway. This tunability minimizes side reactions such as over-oxidation or polymerization, which are common pitfalls in traditional chemical oxidation methods. The result is a clean conversion to the corresponding 1,2-diketone structure with high fidelity.
Furthermore, the divided cell setup is instrumental in maintaining the integrity of the reaction environment. By physically separating the anode and cathode compartments with a membrane, the method prevents the reduced species generated at the cathode from interacting with the oxidized product at the anode. This separation is critical for achieving the high yields reported in the patent examples, such as the 87% yield observed for the unsubstituted diphenylacetylene substrate. The choice of electrode materials, specifically a graphite or glassy carbon anode paired with a platinum cathode, optimizes the electron transfer kinetics while resisting corrosion under the electrolytic conditions. This robust electrode system ensures long-term stability and reproducibility, which are essential parameters for industrial adoption. The mechanism effectively transforms electrical energy into chemical potential, driving the synthesis with minimal waste generation and maximum atom utilization.
How to Synthesize Aromatic Acyl Compounds Efficiently
The practical implementation of this electrochemical synthesis is designed to be straightforward and adaptable to existing laboratory and pilot-scale infrastructure. The process begins with the preparation of the electrolytic solution, where the diarylacetylene substrate is dissolved in a mixture of N,N-dimethylformamide (DMF) and water, supplemented with a supporting electrolyte like NaBF4. This solution is placed in the anode compartment of an H-type divided cell, while the cathode compartment contains a compatible electrolyte solution to complete the circuit. The reaction is initiated by applying a constant current, typically optimized around 20 mA for small-scale batches, and maintained at ambient temperature for a duration of approximately 8 hours. Detailed standardized synthesis steps see the guide below.
- Dissolve the diarylacetylene substrate and supporting electrolyte (e.g., NaBF4) in a DMF: Water solvent mixture within the anode chamber of a divided H-type cell.
- Insert a graphite or glassy carbon anode and a platinum sheet cathode, then apply a constant current of 20 mA at room temperature.
- After 8 hours of electrolysis, quench the reaction with water, extract with ethyl acetate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical technology presents compelling opportunities for cost reduction in pharmaceutical intermediate manufacturing. The most significant economic benefit stems from the elimination of expensive transition metal catalysts, such as palladium or ruthenium complexes, which are subject to volatile market pricing and supply chain constraints. By removing these costly reagents from the bill of materials, manufacturers can achieve substantial cost savings while simultaneously simplifying the purification process. The absence of heavy metals also reduces the regulatory burden associated with residual metal testing and clearance, accelerating the time-to-market for new drug candidates. Furthermore, the mild reaction conditions reduce the energy requirements for heating and cooling, contributing to lower operational expenditures (OPEX) over the lifecycle of the production process.
- Cost Reduction in Manufacturing: The electrochemical process fundamentally alters the cost structure by replacing stoichiometric chemical oxidants with electricity, which is generally cheaper and more consistent in price. This shift eliminates the procurement risks associated with hazardous oxidizing agents and reduces the costs related to their safe handling, storage, and disposal. Additionally, the simplified workup procedure, which involves basic extraction and chromatography, lowers the consumption of auxiliary chemicals and solvents compared to multi-step catalytic cycles. These factors combine to create a leaner manufacturing process that enhances overall profit margins without compromising product quality.
- Enhanced Supply Chain Reliability: Relying on electricity as a primary reagent decouples the production process from the supply chain vulnerabilities of specialized chemical reagents. Since the key inputs are readily available commodity chemicals (substrates and electrolytes) and power, the risk of production stoppages due to reagent shortages is significantly mitigated. This reliability is crucial for maintaining continuous supply to downstream API manufacturers, especially in a global market prone to logistical disruptions. The robustness of the method also allows for flexible production scheduling, enabling manufacturers to respond quickly to fluctuating demand patterns.
- Scalability and Environmental Compliance: Electrochemical synthesis is inherently scalable, particularly with the advent of flow electrochemistry technologies that allow for easy capacity expansion. The green nature of this process, characterized by low waste generation and the absence of toxic heavy metals, ensures compliance with increasingly strict environmental regulations worldwide. This compliance reduces the risk of fines and shutdowns, securing the long-term viability of the manufacturing site. Moreover, the reduced environmental footprint enhances the corporate sustainability profile, which is becoming a key differentiator in B2B procurement decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of aromatic acyl compounds. These answers are derived directly from the experimental data and specifications outlined in the patent documentation to ensure accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production workflows.
Q: What are the primary advantages of this electrochemical method over traditional oxidation?
A: This method eliminates the need for expensive transition metal catalysts and stoichiometric chemical oxidants, significantly reducing heavy metal impurities and waste generation while operating under mild room temperature conditions.
Q: Is this process suitable for substrates with sensitive functional groups?
A: Yes, the patent demonstrates excellent functional group tolerance, successfully converting substrates containing halogens, nitro groups, esters, and trifluoromethyl groups without degradation.
Q: What type of electrolytic cell configuration is required?
A: The process utilizes a divided H-type cell separated by a membrane to prevent cathodic reduction of the product, ensuring high selectivity and yield during the constant current electrolysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Acyl Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in the production of high-value pharmaceutical intermediates. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this can be successfully translated into industrial reality. We are committed to delivering high-purity aromatic acyl compounds that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement metal-free synthetic routes positions us as a strategic partner for clients seeking to minimize impurity profiles and streamline their regulatory filings.
We invite you to collaborate with us to leverage this advanced electrochemical technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality standards. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can drive value for your supply chain.
