Revolutionizing Chiral Synthesis: Catalytic Mitsunobu Technology for Commercial Scale-up
The chemical industry is currently witnessing a paradigm shift in stereoselective synthesis, driven by the urgent need for greener and more economically viable manufacturing processes. Patent CN114308121A introduces a groundbreaking series of novel phosphine oxide catalysts that fundamentally transform the classic Mitsunobu reaction from a stoichiometric burden into a catalytic opportunity. Traditionally, the Mitsunobu reaction has been a cornerstone for the inversion of chiral secondary alcohols, yet its reliance on molar equivalents of triphenylphosphine and azodicarboxylates has long plagued process chemists with substantial waste generation. This new technology leverages specifically designed phosphine oxide structures to facilitate the reaction cycle regeneratively, producing water as the sole byproduct. By integrating this innovation, manufacturers can achieve high-purity pharmaceutical intermediates while adhering to stringent environmental regulations. The implications for the supply chain are profound, offering a reliable phosphine oxide catalyst supplier pathway that aligns with modern sustainability goals without compromising on yield or stereochemical integrity.
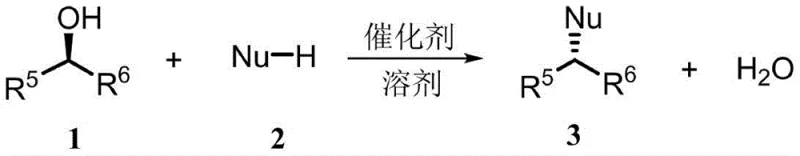
The limitations of conventional Mitsunobu protocols are well-documented in industrial organic synthesis, primarily centering on the atom inefficiency and the downstream challenges associated with byproduct removal. In the traditional stoichiometric approach, the formation of triphenylphosphine oxide (TPPO) and hydrazine derivatives creates a significant purification bottleneck, often requiring extensive chromatography or recrystallization steps that erode overall process yield. Furthermore, the handling of azodicarboxylates such as DEAD or DIAD presents safety hazards due to their potential explosivity and toxicity, necessitating specialized containment and disposal procedures. These factors collectively inflate the cost of goods sold (COGS) and extend lead times for critical active pharmaceutical ingredients (APIs). The accumulation of phosphorus-containing waste also poses severe environmental compliance risks, making the traditional method increasingly untenable for large-scale operations aiming for green chemistry certification.
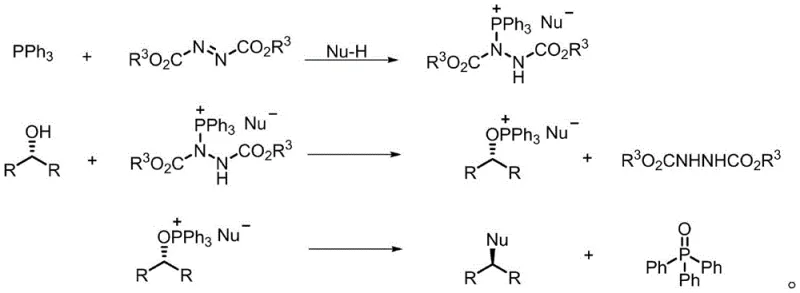
In stark contrast, the novel approach detailed in the patent utilizes a catalytic cycle that circumvents the generation of stoichiometric phosphine waste. By employing the new phosphine oxide catalysts, the reaction proceeds through a mechanism where the catalyst is regenerated in situ, allowing for loading levels as low as 0.05 equivalents while maintaining high conversion rates. This shift from stoichiometric to catalytic reagents drastically simplifies the workup procedure, as the removal of water via azeotropic distillation is far more straightforward than separating solid TPPO from complex organic mixtures. The broad substrate applicability ensures that even sterically hindered or electronically diverse chiral alcohols can be inverted with high fidelity. This technological leap not only enhances the economic feasibility of synthesizing complex molecules but also significantly reduces the environmental footprint, positioning it as a superior choice for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Phosphine Oxide-Catalyzed Stereochemical Inversion
The core of this technological advancement lies in the unique structural features of the phosphine oxide catalysts, defined generally by Formula I and Formula II in the patent documentation. These molecules are engineered to possess specific electronic and steric properties that facilitate the activation of the hydroxyl group while stabilizing the transition state for nucleophilic attack. The presence of the ortho-hydroxyl group in Formula I, for instance, likely plays a crucial role in hydrogen bonding interactions that orient the substrate for optimal backside attack, ensuring the characteristic SN2 inversion of configuration. The versatility of the R groups allows for fine-tuning the catalyst's solubility and reactivity profile, making it adaptable to a wide range of reaction conditions and solvent systems. Understanding these mechanistic nuances is essential for R&D directors aiming to implement this chemistry for high-purity OLED material or agrochemical intermediate synthesis where stereochemical purity is non-negotiable.
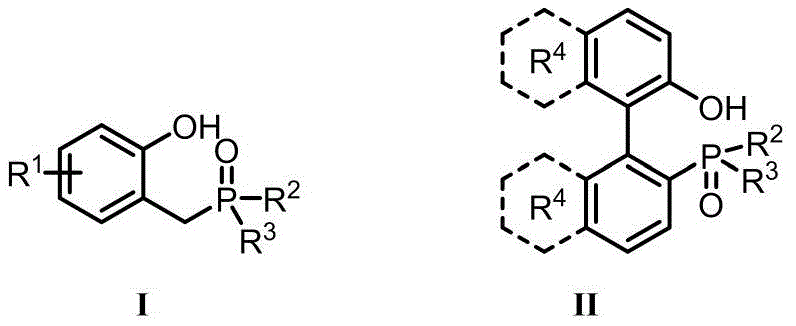
Impurity control is another critical aspect where this catalytic system excels, particularly in the context of regulatory compliance for drug substances. In traditional methods, residual phosphine species and hydrazine byproducts are notorious for persisting through purification stages, potentially leading to genotoxic impurity alerts. The catalytic nature of the new process minimizes the introduction of these extraneous species from the outset. Moreover, the high selectivity of the catalyst reduces the formation of side products such as elimination byproducts or over-oxidized species, which are common pitfalls in non-optimized Mitsunobu reactions. The ability to achieve high enantiomeric excess (ee) values, often exceeding 90% as demonstrated in the patent examples, ensures that the final product meets the rigorous specifications required for clinical applications. This level of control over the impurity profile significantly de-risks the development timeline and facilitates smoother regulatory filings.
How to Synthesize Phosphine Oxide Catalysts Efficiently
The preparation of these advanced catalysts involves robust and scalable synthetic routes that are amenable to industrial production. For Formula I type catalysts, the process typically begins with the condensation of o-hydroxybenzaldehyde derivatives with chlorophosphine compounds under inert atmosphere, followed by a controlled oxidation step using hydrogen peroxide to establish the phosphine oxide moiety. Formula II catalysts, often featuring biphenyl backbones, may require demethylation strategies using boron tribromide to reveal the active phenolic groups. These synthetic pathways are designed to maximize yield and minimize hazardous waste during the catalyst manufacturing itself, reflecting the green chemistry principles inherent in the technology. Detailed standardized synthesis steps see the guide below for precise operational parameters.
- Prepare o-hydroxybenzaldehyde derivatives and chlorophosphine derivatives under argon atmosphere, reacting at 80°C followed by oxidation with H2O2.
- For biphenyl-based catalysts, perform demethylation of methoxy-biphenyl phosphine oxides using boron tribromide at low temperatures.
- Apply the purified catalyst (0.05-0.2 equivalents) in a Dean-Stark apparatus with alcohol substrates and nucleophiles in xylene under reflux.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this catalytic technology offers substantial strategic advantages by fundamentally altering the raw material cost structure. The elimination of stoichiometric amounts of expensive and hazardous azodicarboxylates represents a direct saving in material costs, while the reduction in waste disposal fees contributes to significant overhead reductions. Supply chain reliability is enhanced because the process is less dependent on the volatile market availability of specialized stoichiometric reagents, which can often face supply disruptions. Instead, the reliance shifts to more commodity-grade starting materials and a reusable catalyst system, ensuring greater continuity of supply for critical pharmaceutical intermediates. This stability is crucial for maintaining production schedules and meeting delivery commitments to downstream partners in the global healthcare market.
- Cost Reduction in Manufacturing: The transition to a catalytic regime removes the need for purchasing triphenylphosphine and azo reagents in molar quantities, which traditionally constitute a major portion of the raw material budget. Additionally, the simplification of purification processes reduces solvent consumption and energy usage associated with extensive chromatography or recrystallization. These operational efficiencies translate into a lower cost per kilogram of the final API intermediate, improving overall margin potential without sacrificing quality standards.
- Enhanced Supply Chain Reliability: By minimizing the generation of hazardous phosphorus waste, the process alleviates bottlenecks associated with waste treatment capacity, which can often limit production throughput in regulated jurisdictions. The robustness of the catalyst system allows for consistent batch-to-batch performance, reducing the risk of campaign failures that could disrupt supply. This reliability is essential for securing long-term contracts with multinational corporations that prioritize vendor stability and risk mitigation in their sourcing strategies.
- Scalability and Environmental Compliance: The green chemistry attributes of this method, specifically the production of water as the only byproduct, align perfectly with increasingly stringent environmental regulations globally. Scaling this process from laboratory to commercial tonnage does not incur the exponential increase in waste management costs seen with traditional methods. This scalability ensures that the technology remains economically viable at high volumes, supporting the commercial scale-up of complex polymer additives or fine chemicals while maintaining a sustainable corporate image.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this phosphine oxide catalyst technology. These insights are derived directly from the patent data and practical considerations for industrial application, providing clarity on performance metrics and integration requirements. Understanding these details helps stakeholders make informed decisions about adopting this innovative synthetic route for their specific product portfolios.
Q: How does this catalytic method improve upon traditional Mitsunobu conditions?
A: Traditional methods require stoichiometric triphenylphosphine and azodicarboxylates, generating significant phosphine oxide waste. This patented catalytic approach uses water as the only byproduct, drastically reducing waste disposal costs and purification complexity.
Q: What is the stereoselectivity performance of these phosphine oxide catalysts?
A: The catalysts demonstrate high stereoselectivity, achieving complete configuration inversion of chiral secondary alcohols with ee values frequently exceeding 90% across diverse substrate scopes including complex pharmaceutical intermediates.
Q: Is this technology suitable for large-scale industrial production?
A: Yes, the catalysts are designed for high atom economy and recyclability. The elimination of stoichiometric hazardous reagents and the simplification of downstream processing make it highly viable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphine Oxide Catalyst Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating cutting-edge academic research into commercially viable chemical solutions. As experts in CDMO services, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to full-scale manufacturing is seamless. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which guarantee that every batch of catalyst or intermediate meets the highest international standards. We understand the critical nature of chiral synthesis in drug development and are equipped to support your projects with the technical depth required to navigate complex regulatory landscapes.
We invite you to collaborate with us to leverage this transformative technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific synthesis requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our phosphine oxide catalysts can optimize your manufacturing process. Let us partner with you to drive efficiency, sustainability, and success in your chemical supply chain.
