Advanced Synthesis of Axial Chiral Fluoreneamine-Phenol Derivatives for Commercial Scale-up
The chemical industry is constantly evolving, driven by the need for novel molecular scaffolds that offer superior performance in pharmaceutical and material science applications. Patent CN113336670B introduces a groundbreaking approach to synthesizing axial chiral fluoreneamine-phenol derivatives, addressing a critical gap in the current landscape of chiral compound availability. Traditionally, the construction of axial chiral cores has been dominated by biaryl or heteroaryl skeletons, limiting the structural diversity available to medicinal chemists and material scientists. This patent discloses a unique methodology that incorporates a fluorene core into the axial chiral framework, thereby expanding the chemical space for potential drug candidates and functional materials. The significance of this innovation lies not only in the novelty of the molecular architecture but also in the robustness of the synthetic route, which utilizes accessible starting materials and mild reaction conditions to achieve high structural complexity. 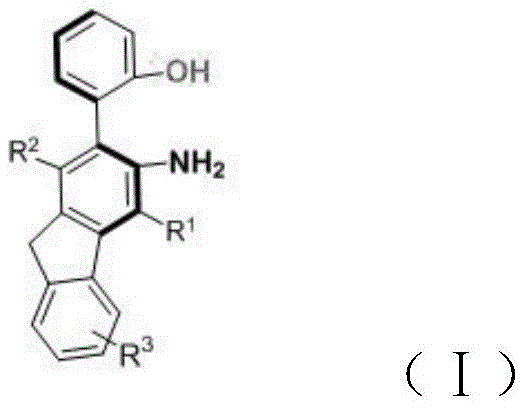 For R&D directors seeking to diversify their pipeline with novel chiral intermediates, this technology represents a reliable pharmaceutical intermediates supplier opportunity that bridges the gap between academic innovation and industrial feasibility. The ability to access these specific derivatives efficiently opens new avenues for asymmetric catalysis and ligand design, positioning this chemistry at the forefront of modern organic synthesis.
For R&D directors seeking to diversify their pipeline with novel chiral intermediates, this technology represents a reliable pharmaceutical intermediates supplier opportunity that bridges the gap between academic innovation and industrial feasibility. The ability to access these specific derivatives efficiently opens new avenues for asymmetric catalysis and ligand design, positioning this chemistry at the forefront of modern organic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of axially chiral compounds has relied heavily on transition metal-catalyzed cross-coupling reactions or resolution of racemic mixtures, both of which present significant drawbacks for large-scale manufacturing. Transition metal catalysts, often based on palladium, rhodium, or iridium, are not only prohibitively expensive but also introduce the risk of heavy metal contamination in the final product, necessitating costly and time-consuming purification steps to meet stringent regulatory standards. Furthermore, conventional methods frequently require harsh reaction conditions, such as high temperatures or strong bases, which can compromise the stability of sensitive functional groups and lead to lower overall yields. The reliance on multi-step sequences with intermediate isolation further exacerbates the issue, resulting in increased solvent consumption, higher waste generation, and extended production timelines. These factors collectively hinder the commercial viability of many promising chiral scaffolds, making them inaccessible for cost-sensitive applications in the fine chemical sector. Consequently, there is an urgent demand for alternative synthetic strategies that can overcome these economic and environmental bottlenecks while maintaining high stereocontrol.
The Novel Approach
In stark contrast to traditional methodologies, the process described in CN113336670B employs an organocatalytic strategy using DABCO (1,4-Diazabicyclo[2.2.2]octane) as a metal-free catalyst, fundamentally altering the economic and operational profile of the synthesis. This novel approach leverages a one-pot multi-step reaction sequence where 3-(benzylidene)benzofuran-2(3H)-one and 1-indenylidenemalononitrile derivatives undergo consecutive transformations in a single reaction vessel. By eliminating the need for intermediate isolation, the process drastically reduces solvent usage and labor costs, directly contributing to cost reduction in pharmaceutical intermediates manufacturing. The reaction proceeds under mild thermal conditions, typically ranging from 20°C to 80°C, which minimizes energy consumption and enhances safety profiles for industrial operators. Moreover, the use of acetonitrile as a common solvent and sodium hydroxide as a base ensures that all reagents are readily available and inexpensive, facilitating a streamlined supply chain. This methodological shift not only improves the atom economy of the reaction but also aligns with green chemistry principles, making it an attractive option for companies aiming to reduce their environmental footprint while scaling up production of complex chiral molecules.
Mechanistic Insights into DABCO-Catalyzed Cyclization
The mechanistic pathway of this transformation is a testament to the power of organocatalysis in constructing complex molecular architectures with high precision. The reaction initiates with the nucleophilic activation of the 1-indenylidenemalononitrile by the DABCO catalyst, which enhances its electrophilicity and facilitates the subsequent attack on the 3-(benzylidene)benzofuran-2(3H)-one substrate. This initial step is crucial for establishing the carbon-carbon bonds that form the backbone of the fluorene skeleton. Following the initial coupling, the addition of sodium hydroxide triggers a cascade of intramolecular cyclization and ring-opening events that ultimately yield the axially chiral fluoreneamine-phenol structure. The stereoselectivity of the reaction is governed by the steric interactions between the substituents on the starting materials and the chiral environment created during the transition state, ensuring the formation of the desired axial chirality. 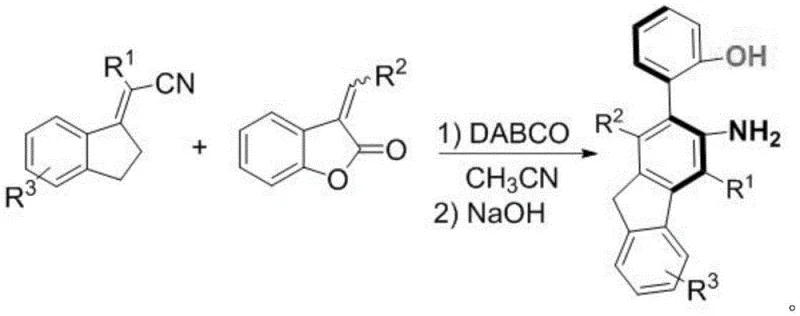 Understanding these mechanistic details is vital for process chemists who need to optimize reaction parameters such as temperature and stoichiometry to maximize yield and purity. The ability to fine-tune these variables allows for the accommodation of various substituents, as indicated by the broad scope of R groups defined in the patent, thereby enabling the synthesis of a diverse library of derivatives tailored for specific biological or material properties.
Understanding these mechanistic details is vital for process chemists who need to optimize reaction parameters such as temperature and stoichiometry to maximize yield and purity. The ability to fine-tune these variables allows for the accommodation of various substituents, as indicated by the broad scope of R groups defined in the patent, thereby enabling the synthesis of a diverse library of derivatives tailored for specific biological or material properties.
Impurity control is another critical aspect of this mechanism that holds significant value for quality assurance teams. The specificity of the DABCO-catalyzed pathway minimizes the formation of side products that are commonly associated with transition metal catalysis, such as homocoupling byproducts or dehalogenated species. The use of a mild base like sodium hydroxide in the second step ensures that sensitive functional groups remain intact, preventing degradation pathways that could complicate downstream purification. Furthermore, the reaction conditions are designed to favor the thermodynamic product, which enhances the diastereoselectivity and reduces the burden on chromatographic separation. For procurement managers, this high level of selectivity translates to reduced waste and lower costs associated with refining crude products. The robust nature of the reaction mechanism also implies a high degree of reproducibility, which is essential for maintaining consistent quality across different production batches. This reliability is a key factor for supply chain heads who need to guarantee the continuity of supply for critical intermediates without the risk of batch-to-batch variability affecting final product performance.
How to Synthesize Axial Chiral Fluoreneamine-Phenol Derivatives Efficiently
Implementing this synthesis route in a practical setting requires a clear understanding of the operational parameters defined in the patent to ensure optimal outcomes. The process begins with the precise weighing of 3-(benzylidene)benzofuran-2(3H)-one and 1-indenylidenemalononitrile derivatives, maintaining a molar ratio that favors the formation of the desired product while minimizing excess reagent waste. These reactants are dissolved in acetonitrile, and the DABCO catalyst is added to initiate the first stage of the reaction, which is maintained at a controlled temperature between 20°C and 80°C for a duration of 5 to 72 hours depending on the specific substrate reactivity. Upon completion of this initial phase, sodium hydroxide is introduced to the reaction mixture to drive the second stage of the transformation, which continues for an additional 10 to 48 hours under similar thermal conditions. The detailed standardized synthesis steps see the guide below for specific stoichiometric ratios and workup procedures. This structured approach ensures that the reaction proceeds smoothly through both stages, maximizing the conversion of starting materials into the target axial chiral derivative while maintaining the integrity of the chiral axis.
- React 3-(benzylidene)benzofuran-2(3H)-one and 1-indenylidenemalononitrile with DABCO catalyst in acetonitrile at 20-80°C for 5-72 hours.
- Add sodium hydroxide to the reaction system and continue reacting at 20-80°C for 10-48 hours to complete the transformation.
- Quench with saturated ammonium chloride, extract with dichloromethane, dry, and purify via column chromatography to obtain the final derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits that extend beyond mere technical feasibility, directly impacting the bottom line and operational resilience of chemical manufacturing enterprises. The elimination of transition metal catalysts removes a major cost driver associated with precious metal procurement and the specialized equipment required for their handling and recovery. Additionally, the simplified workup procedure, which involves standard extraction and drying techniques followed by column chromatography, reduces the need for complex purification infrastructure. This streamlining of the process flow enhances the overall throughput of the manufacturing facility, allowing for faster turnaround times and improved responsiveness to market demands. For supply chain leaders, the reliance on commodity chemicals like acetonitrile and sodium hydroxide mitigates the risk of supply disruptions that are often associated with specialized reagents. The robustness of the reaction conditions also means that the process can be easily transferred between different production sites without significant re-validation, providing flexibility in sourcing and production planning. These factors collectively contribute to a more agile and cost-efficient supply chain capable of supporting the growing demand for high-value chiral intermediates.
- Cost Reduction in Manufacturing: The primary economic advantage of this technology lies in its ability to significantly lower the cost of goods sold through the substitution of expensive catalytic systems with affordable organocatalysts. By utilizing DABCO instead of precious metals, manufacturers can avoid the volatility of metal prices and the high capital expenditure associated with metal scavenging technologies. Furthermore, the one-pot nature of the reaction reduces solvent consumption and energy usage, as there is no need for heating and cooling cycles between multiple reaction steps. This cumulative effect results in substantial cost savings that can be passed on to customers or reinvested into further R&D initiatives. The qualitative improvement in process efficiency also means that labor costs are optimized, as fewer manual interventions are required to monitor and manage the reaction progress. Overall, the economic model supported by this patent is designed to maximize profitability while maintaining competitive pricing in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: Supply chain stability is paramount for pharmaceutical and agrochemical companies that depend on consistent access to high-quality intermediates. This synthesis method enhances reliability by utilizing raw materials that are widely available from multiple global suppliers, reducing dependency on single-source vendors. The mild reaction conditions also minimize the risk of equipment failure or safety incidents that could lead to unplanned downtime. Moreover, the scalability of the process ensures that production volumes can be ramped up quickly to meet surges in demand without compromising product quality. For procurement managers, this means a lower risk of stockouts and a more predictable lead time for order fulfillment. The ability to source key inputs locally in many regions further strengthens the supply chain against geopolitical disruptions or logistics bottlenecks. This resilience is a critical asset in today's volatile market environment, where continuity of supply is often as valuable as the price of the product itself.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental sustainability continue to intensify, the ability to scale processes that adhere to green chemistry principles is a significant competitive advantage. This method supports scalability by avoiding the generation of hazardous waste streams associated with heavy metal catalysts, simplifying compliance with environmental regulations. The use of acetonitrile, a solvent that can be readily recovered and recycled, further reduces the environmental footprint of the manufacturing process. The high atom economy of the reaction ensures that a maximum proportion of the starting materials is incorporated into the final product, minimizing waste disposal costs. For companies aiming to achieve carbon neutrality or reduce their ecological impact, adopting this technology demonstrates a commitment to sustainable practices. The ease of scale-up from laboratory to pilot and then to commercial production ensures that the environmental benefits observed at small scale are maintained at larger volumes, making it a future-proof solution for responsible chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical and commercial aspects of this synthesis technology, providing clarity for stakeholders evaluating its potential integration into their operations. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation, ensuring accuracy and relevance. Understanding these details is essential for making informed decisions about process adoption and resource allocation. The information covers key areas such as catalyst selection, reaction conditions, and product purification, which are critical for successful implementation. By addressing these points proactively, we aim to facilitate a smoother transition from evaluation to execution for our partners. We encourage interested parties to review these insights closely as they consider the strategic value of this innovative synthetic route for their specific application needs.
Q: What are the primary advantages of this DABCO-catalyzed method over traditional transition metal catalysis?
A: This method eliminates the need for expensive and toxic transition metal catalysts, significantly reducing raw material costs and simplifying the purification process by removing heavy metal residue concerns.
Q: How does the one-pot multi-step reaction impact production efficiency?
A: By conducting consecutive reactions in a single vessel without isolating intermediates, the process saves substantial time and solvent usage, thereby enhancing overall operational efficiency and throughput.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the use of mild reaction temperatures (20-80°C) and common solvents like acetonitrile makes the process highly adaptable for scale-up from laboratory to industrial production levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Fluoreneamine-Phenol Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methods described in CN113336670B and are fully equipped to bring this technology to commercial fruition for our global partners. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from the laboratory bench to full-scale manufacturing. Our facilities are designed to handle complex organic syntheses with the highest standards of safety and quality, supported by rigorous QC labs that guarantee stringent purity specifications for every batch produced. We understand that the successful commercialization of novel chiral intermediates requires not just technical capability but also a deep commitment to partnership and reliability. Our team of experts is ready to collaborate with you to optimize the process parameters for your specific requirements, ensuring that the final product meets all necessary regulatory and performance criteria. By choosing us as your partner, you gain access to a wealth of knowledge and infrastructure that can accelerate your time to market and enhance your competitive position.
We invite you to engage with our technical procurement team to discuss how we can support your specific needs for high-purity axial chiral fluoreneamine-phenol derivatives. Whether you require a Customized Cost-Saving Analysis to evaluate the economic benefits of switching to this new route or need specific COA data to verify product quality, we are here to provide the necessary support. We also offer route feasibility assessments to help you determine the best path forward for your development projects. Our goal is to be more than just a vendor; we strive to be a strategic ally in your success, providing the chemical solutions and expertise needed to drive innovation in your field. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our capabilities can add value to your supply chain.
