Advanced Copper-Catalyzed Synthesis of Polysubstituted Pyrrolidine Compounds for CNS Drug Development
Advanced Copper-Catalyzed Synthesis of Polysubstituted Pyrrolidine Compounds for CNS Drug Development
The pharmaceutical industry is constantly seeking robust and scalable methodologies for constructing complex heterocyclic scaffolds, particularly those with proven biological activity. Patent CN113620856B introduces a groundbreaking approach to synthesizing polysubstituted pyrrolidine compounds, a structural motif prevalent in numerous bioactive molecules and central nervous system (CNS) stimulants. This technology leverages a novel copper-catalyzed radical coupling strategy that significantly streamlines the construction of the pyrrolidine core compared to traditional multi-step sequences. By utilizing nitrogen-fluoro sulfonamides and aryl acetylenes as key building blocks, this method achieves high yields under remarkably mild conditions, addressing critical pain points in process chemistry such as harsh reaction environments and poor atom economy. For R&D teams focused on CNS drug discovery, this patent offers a versatile platform for generating diverse libraries of pyrrolidine derivatives with high structural fidelity.
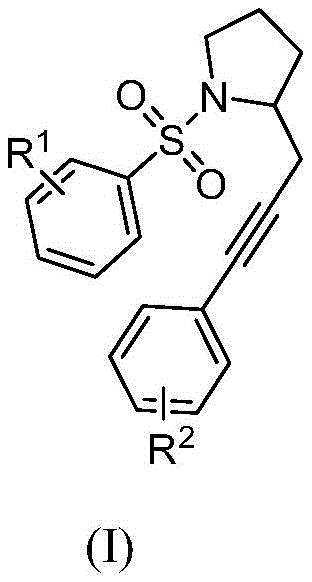
The core innovation lies in the specific structural architecture of the resulting polysubstituted pyrrolidine compound, designated as Formula (I). In this structure, the substituents R1 and R2 can be independently selected from a wide range of groups including hydrogen, C1-C6 alkyl, C1-C6 alkoxy, or halogens, providing immense flexibility for medicinal chemists to tune the physicochemical properties of the final drug candidate. The ability to introduce diverse functional groups at these positions without compromising the integrity of the pyrrolidine ring is a significant advantage for structure-activity relationship (SAR) studies. Furthermore, the patent highlights that these compounds hold important research value specifically as central nervous system stimulants, aligning with current market demands for novel therapeutics in neurology. This structural versatility ensures that the methodology is not limited to a single molecule but serves as a generalizable route for a broad class of valuable intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted pyrrolidines has been fraught with challenges that hinder efficient large-scale production. Conventional routes often rely on cycloaddition reactions or cyclization of amino alcohols, which frequently require elevated temperatures, strong acids or bases, and expensive transition metal catalysts that are difficult to remove from the final product. These harsh conditions can lead to the degradation of sensitive functional groups, limiting the scope of substrates that can be utilized and resulting in complex impurity profiles that are costly to purify. Additionally, many traditional methods suffer from poor regioselectivity and stereoselectivity, necessitating additional resolution steps that drastically reduce overall yield and increase manufacturing costs. The reliance on stoichiometric amounts of reagents in older protocols also generates significant chemical waste, posing environmental compliance issues for modern green chemistry initiatives.
The Novel Approach
In stark contrast, the method disclosed in CN113620856B utilizes a catalytic radical coupling mechanism that operates under exceptionally mild conditions, typically at 30°C, thereby preserving sensitive functional groups and minimizing byproduct formation. This novel approach employs a copper catalyst system, specifically Cu(CH3CN)4PF6, in conjunction with a base like potassium tert-butoxide to activate the nitrogen-fluoro sulfonamide precursor. The reaction proceeds through a nitrogen-centered radical intermediate which undergoes intramolecular addition to an olefin followed by intermolecular coupling with an aryl acetylene. This cascade process constructs the pyrrolidine ring and installs the alkyne functionality in a single operational step, dramatically reducing the number of unit operations required. The use of inert gas protection and common organic solvents like DCM further enhances the practicality of this method for industrial application.
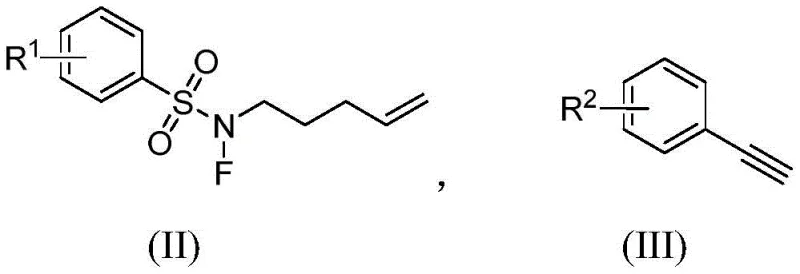
Mechanistic Insights into Copper-Catalyzed Radical Coupling
The mechanistic pathway of this transformation is a sophisticated example of modern radical chemistry applied to heterocycle synthesis. Upon activation by the copper catalyst and base, the nitrogen-fluoro sulfonamide (II) undergoes single-electron transfer to generate a reactive nitrogen-centered radical species. This radical is highly electophilic and rapidly adds to the tethered olefin moiety within the same molecule, initiating a 5-exo-trig cyclization that forms the five-membered pyrrolidine ring skeleton. The resulting carbon-centered radical is then trapped by the aryl acetylene (III) through a radical coupling event, establishing the new carbon-carbon bond and completing the polysubstituted framework. This sequence avoids the formation of high-energy ionic intermediates that often lead to rearrangement side products in classical ionic cyclizations, ensuring a cleaner reaction profile.
Impurity control in this process is inherently superior due to the specificity of the radical generation and the mild thermal conditions employed. Since the reaction temperature is maintained at a low 30°C, thermal decomposition pathways and non-selective radical polymerization of the alkyne are effectively suppressed. The choice of ligand and counter-ion in the copper catalyst plays a crucial role in modulating the redox potential, ensuring that the radical is generated at a controlled rate that matches the cyclization kinetics. This precise control minimizes the accumulation of free radicals that could otherwise lead to dimerization or oligomerization byproducts. Consequently, the crude reaction mixture typically contains fewer impurities, simplifying downstream purification and enhancing the overall purity of the isolated polysubstituted pyrrolidine product.
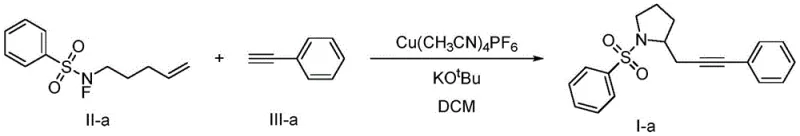
How to Synthesize Polysubstituted Pyrrolidine Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of handling in a standard laboratory or pilot plant setting. The protocol begins with the preparation of the catalytic system under an inert atmosphere to prevent oxidation of the active copper species. Substrates are added sequentially to manage the exotherm and ensure proper mixing, followed by a stirring period that allows the radical cascade to reach completion. The workup involves standard aqueous quenching and extraction techniques, followed by purification via silica gel chromatography using common eluent systems. Detailed standardized synthetic steps for this procedure are provided in the guide below to ensure consistent results across different batches.
- Prepare the reaction vessel by adding the copper catalyst Cu(CH3CN)4PF6 and potassium tert-butoxide base under an inert argon atmosphere.
- Introduce the anhydrous DCM solvent, followed by the sequential addition of nitrogen-fluoro sulfonamide and phenylacetylene substrates.
- Maintain the reaction mixture at 30°C for 12 hours, monitoring progress via TLC, then purify the crude product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond mere technical feasibility. The shift from multi-step traditional syntheses to this concise one-pot radical coupling significantly reduces the consumption of raw materials and solvents, directly impacting the cost of goods sold (COGS). By eliminating the need for intermediate isolation and purification steps, the process throughput is increased, allowing for faster turnaround times from raw material intake to finished intermediate. This efficiency gain is critical for maintaining lean inventory levels and responding swiftly to fluctuating market demands for CNS active pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of commercially available and relatively inexpensive starting materials such as aryl acetylenes and sulfonamides. The catalytic nature of the copper system means that only small loadings (4-6%) are required to drive the reaction to completion, avoiding the high costs associated with stoichiometric noble metal reagents. Furthermore, the mild reaction conditions eliminate the need for specialized high-pressure or high-temperature equipment, reducing capital expenditure and energy consumption. The simplified purification process also lowers the cost of consumables like silica gel and solvents, contributing to substantial overall cost savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions and the availability of key reagents. The reagents used, including the copper catalyst and potassium tert-butoxide, are commodity chemicals with stable global supply chains, mitigating the risk of shortages that often plague specialized reagents. The tolerance of the reaction to various substituents allows for flexibility in sourcing; if a specific substituted aryl acetylene is unavailable, alternative analogs can often be substituted without re-optimizing the entire process. This flexibility ensures continuous production capability even when specific raw material streams face logistical disruptions.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram scale is facilitated by the absence of hazardous reagents and the control of exothermic events through moderate temperature settings. The use of dichloromethane (DCM) or other common solvents allows for easy recovery and recycling, aligning with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions. The high atom economy of the coupling reaction minimizes waste generation, reducing the burden on waste treatment facilities and lowering disposal costs. This environmental compatibility makes the process attractive for manufacturers aiming to meet green chemistry standards and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this polysubstituted pyrrolidine synthesis. These answers are derived directly from the experimental data and specifications outlined in the patent documentation to provide clarity on operational parameters. Understanding these details is essential for process engineers and quality assurance teams evaluating the technology for potential licensing or contract manufacturing opportunities.
Q: What is the optimal catalyst loading for this pyrrolidine synthesis?
A: The patent specifies a metal catalyst loading of 4-6% relative to the nitrogen-fluoro sulfonamide substrate, with copper catalysts like Cu(CH3CN)4PF6 showing excellent efficiency.
Q: Can this method tolerate various substituents on the aromatic rings?
A: Yes, the method demonstrates broad substrate scope, successfully accommodating hydrogen, alkyl, alkoxy, and halogen groups at both para and other positions on the aromatic rings.
Q: What are the typical reaction conditions required?
A: The reaction proceeds efficiently under mild conditions, typically at 30°C in solvents like DCM, using potassium tert-butoxide as the base under an inert gas atmosphere.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Pyrrolidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed technology in accelerating the development of next-generation CNS therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of radical chemistry, including rigorous inert atmosphere controls and stringent purity specifications to deliver intermediates that meet the highest quality standards. Our rigorous QC labs utilize advanced analytical techniques to verify the structural integrity and purity of every batch, guaranteeing consistency for your clinical and commercial needs.
We invite you to leverage our technical expertise to optimize this synthesis for your specific pipeline requirements. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency and reduce your time to market.
