Advanced Gold-Catalyzed Synthesis of Dihydropyrrolidone Derivatives for Scalable Pharmaceutical Manufacturing
Advanced Gold-Catalyzed Synthesis of Dihydropyrrolidone Derivatives for Scalable Pharmaceutical Manufacturing
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of modern pharmaceutical manufacturing, where efficiency, selectivity, and environmental sustainability are paramount. A significant breakthrough in this domain is detailed in patent CN113087649B, which discloses a novel preparation method for dihydropyrrolidone derivatives. This technology leverages a sophisticated gold-catalyzed multicomponent reaction strategy to construct the pyrrolidone core, a structural motif ubiquitous in bioactive molecules ranging from HIV integrase inhibitors to anticancer agents. By utilizing butynedioic acid ester compounds, hexahydro-1,3,5-triazine compounds, and aryl or fatty primary amines as starting materials, this process achieves high regioselectivity and yield under remarkably mild conditions. For R&D directors and procurement specialists alike, this represents a pivotal shift towards more sustainable and cost-effective manufacturing pathways for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dihydropyrrol-2-one derivatives has been fraught with significant technical and operational challenges that hinder efficient commercial scale-up. Traditional methodologies often rely on multi-step sequences that require harsh reaction conditions, including elevated temperatures and the use of aggressive reagents, which can compromise the integrity of sensitive functional groups. Furthermore, many established protocols necessitate the use of chlorinated solvents, posing severe environmental hazards and complicating waste disposal compliance. Post-reaction processing in these conventional routes is frequently cumbersome, involving complex extraction and purification steps that erode overall process efficiency and increase the cost of goods sold. These limitations create bottlenecks in the supply chain, making it difficult to secure reliable sources of high-purity intermediates without incurring substantial lead times and expenses.
The Novel Approach
In stark contrast, the methodology outlined in patent CN113087649B introduces a streamlined, one-pot synthetic route that effectively circumvents these historical obstacles. By employing a dual catalyst system comprising LAuCl and AgOTf, the reaction proceeds smoothly at temperatures ranging from 10°C to 50°C, drastically reducing energy consumption and thermal stress on the reactants. This approach not only enhances the safety profile of the operation but also significantly improves the regioselectivity of the cyclization, minimizing the formation of unwanted by-products. The versatility of this method is further evidenced by its compatibility with a wide array of solvents, including greener options like ethanol and ethyl acetate, aligning perfectly with modern green chemistry principles. 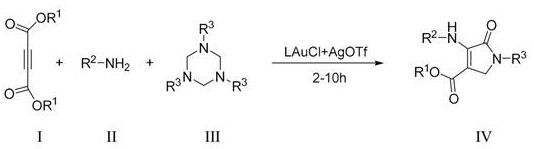
Mechanistic Insights into Gold-Catalyzed Cyclization
The core of this technological advancement lies in the unique activation mode provided by the gold catalyst, which facilitates the construction of the dihydropyrrolidone ring through a highly controlled mechanistic pathway. The gold species, generated in situ from the LAuCl and AgOTf precursor combination, acts as a potent pi-acid to activate the alkyne moiety of the butynedioic acid ester. This activation renders the triple bond susceptible to nucleophilic attack by the amine component, initiating a cascade of bond-forming events that ultimately lead to the cyclic product. The presence of the hexahydro-1,3,5-triazine serves as a crucial nitrogen source or formaldehyde equivalent, participating in the multicomponent assembly to forge the necessary carbon-nitrogen bonds with precision. This catalytic cycle ensures that the reaction proceeds with exceptional atom economy, maximizing the incorporation of starting materials into the final desired structure while minimizing waste generation.
From an impurity control perspective, the mild nature of the gold-catalyzed mechanism offers distinct advantages over thermal or acid-catalyzed alternatives. The lower operating temperatures prevent the degradation of thermally labile substituents on the amine or ester components, thereby preserving the chemical fidelity of the product. Additionally, the high regioselectivity inherent to the gold-mediated pathway ensures that the cyclization occurs exclusively at the desired position, effectively suppressing the formation of regioisomers that are notoriously difficult to separate. This intrinsic selectivity translates directly into a cleaner crude reaction profile, which simplifies downstream purification and reduces the burden on analytical quality control teams. For manufacturers targeting stringent purity specifications required for pharmaceutical applications, this level of mechanistic control is indispensable for ensuring batch-to-batch consistency and regulatory compliance.
How to Synthesize Dihydropyrrolidone Derivatives Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the stoichiometry and addition sequence of the reagents to maximize yield and purity. The process begins with the activation of the gold catalyst in a suitable organic solvent, followed by the sequential introduction of the butynedioic acid ester, the triazine component, and the primary amine. Maintaining the reaction temperature within the optimal window of 20°C to 30°C is critical for balancing reaction rate and selectivity, although the protocol tolerates a broader range from 10°C to 50°C depending on the specific substrate reactivity. Upon completion, typically monitored by TLC within 2 to 10 hours, the workup involves a straightforward concentration step followed by recrystallization, avoiding the need for column chromatography.
- Prepare the catalyst system by mixing LAuCl and AgOTf in an organic solvent such as toluene or ethanol at room temperature.
- Add butynedioic acid ester, hexahydro-1,3,5-triazine, and the primary amine substrate sequentially to the reaction mixture.
- Stir the reaction at mild temperatures between 10°C and 50°C for 2 to 10 hours, then purify the crude product via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this gold-catalyzed synthesis method presents a compelling value proposition centered around cost optimization and supply reliability. The primary raw materials, including dialkyl butynedioates and various primary amines, are commodity chemicals that are widely available from multiple global suppliers, mitigating the risk of single-source dependency. The elimination of harsh reaction conditions and chlorinated solvents not only reduces the environmental footprint but also lowers the operational costs associated with specialized equipment maintenance and hazardous waste disposal. Furthermore, the simplicity of the purification process, which relies on recrystallization rather than complex chromatographic techniques, significantly shortens the production cycle time and reduces solvent consumption. These factors collectively contribute to a more resilient and cost-efficient supply chain capable of meeting fluctuating market demands without compromising on quality or delivery schedules.
- Cost Reduction in Manufacturing: The transition to this mild catalytic system eliminates the need for expensive high-temperature reactors and extensive energy inputs, leading to substantial operational savings. By avoiding the use of chlorinated solvents and simplifying the purification workflow to a single recrystallization step, the process drastically reduces solvent purchase and disposal costs. The high yields reported across various substrate scopes ensure that raw material utilization is maximized, further driving down the cost per kilogram of the final active pharmaceutical ingredient intermediate. These cumulative efficiencies allow for a more competitive pricing structure while maintaining healthy profit margins for manufacturers.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method against variations in substrate structure ensures consistent production output even when sourcing different grades of raw materials. Since the reaction tolerates a wide range of aryl and aliphatic amines, procurement teams have the flexibility to switch suppliers based on availability and price without needing to revalidate the entire process. The shortened reaction times and simplified workup procedures enable faster turnaround from order to shipment, effectively reducing lead times for high-purity pharmaceutical intermediates. This agility is crucial for maintaining continuous production lines and preventing stockouts in downstream drug manufacturing operations.
- Scalability and Environmental Compliance: Scaling this process from gram to ton quantities is facilitated by the homogeneous nature of the catalytic system and the absence of exothermic hazards associated with high-temperature reactions. The use of greener solvents like ethanol and ethyl acetate aligns with increasingly strict environmental regulations, ensuring long-term compliance and reducing the risk of regulatory shutdowns. The minimal generation of hazardous by-products simplifies waste treatment protocols, making the facility more sustainable and socially responsible. This environmental stewardship not only protects the company's reputation but also future-proofs the manufacturing asset against tightening global emission standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dihydropyrrolidone synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of this gold-catalyzed method over traditional pyrrolidone synthesis?
A: This method operates under significantly milder conditions (10°C-50°C) compared to traditional high-temperature processes, utilizes readily available raw materials, and simplifies post-treatment through straightforward recrystallization, thereby reducing operational complexity and environmental impact.
Q: Can this synthesis protocol accommodate diverse amine substrates?
A: Yes, the protocol demonstrates excellent substrate adaptability, successfully reacting with various aryl and aliphatic primary amines, including those with electron-withdrawing or electron-donating groups, ensuring broad applicability for generating diverse chemical libraries.
Q: Is the purification process suitable for large-scale commercial production?
A: Absolutely. The method avoids complex chromatographic separations, relying instead on simple recrystallization from common solvents like ethanol or acetone, which is highly scalable, cost-effective, and ideal for industrial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dihydropyrrolidone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to stay competitive in the global pharmaceutical market. Our team of expert chemists has extensively evaluated the gold-catalyzed pathway described in patent CN113087649B and confirmed its potential for delivering high-quality intermediates with exceptional purity profiles. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of materials regardless of volume requirements. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch meets the exacting standards required for clinical and commercial drug development.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume needs and timeline constraints. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency. Let us help you transform complex chemical challenges into streamlined commercial successes with our reliable dihydropyrrolidone derivatives supply solutions.
