Scalable Synthesis of High-Purity Alpha-Amino Aryl Alkyl Ketones for Pharmaceutical Applications
Introduction to Advanced Ketone Synthesis Technology
The pharmaceutical and agrochemical industries continuously demand high-purity intermediates that can be manufactured with exceptional efficiency and minimal environmental impact. A significant breakthrough in this domain is detailed in patent CN101633647A, which discloses a robust method for synthesizing alpha-amino aryl alkyl ketone compounds with high selectivity and yield. This technology addresses critical bottlenecks in traditional synthetic routes, offering a streamlined four-step process that transforms simple aromatic hydrocarbons into complex, functionalized ketones suitable for diverse applications including UV curing agents and active pharmaceutical ingredients. The core innovation lies in the strategic modification of reaction conditions, particularly during the bromination and ring-opening stages, to maximize atom economy and product quality.
The general structure of these valuable compounds, as depicted in the patent, allows for significant structural diversity through variations in the aromatic ring substituents and the amine moiety. This flexibility makes the process highly attractive for a reliable pharmaceutical intermediate supplier seeking to expand their portfolio with versatile building blocks. By leveraging this methodology, manufacturers can access a wide range of derivatives where R1 can be hydrogen, alkyl, alkoxy, or halogens, and the amine component can form various heterocyclic rings. The ability to tune these parameters while maintaining high process efficiency is a key driver for adoption in commercial scale-up of complex fine chemicals.
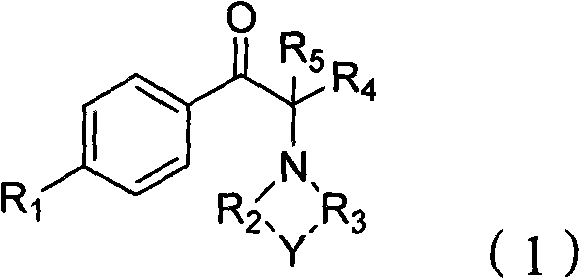
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for synthesizing similar aryl alkyl ketones, such as those disclosed in U.S. Patents 4582862A and 4308400A, suffer from inherent inefficiencies that hinder large-scale production. Traditional routes often rely on direct bromination strategies that exhibit poor atom economy, with bromine utilization rates typically capped at around 50%. This inefficiency not only drives up raw material costs but also generates substantial amounts of hazardous waste, complicating disposal and increasing the environmental footprint of the manufacturing process. Furthermore, conventional ring-opening reactions of epoxy intermediates often lack selectivity, leading to the formation of unwanted by-products that require energy-intensive purification steps to remove. These limitations result in lower overall yields and inconsistent product quality, posing significant risks for supply chain stability.
The Novel Approach
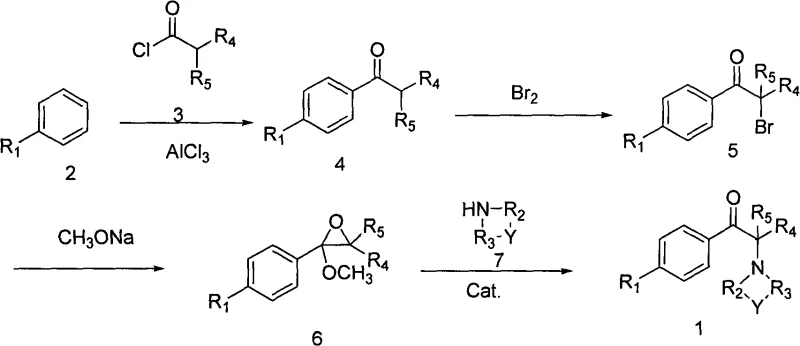
The patented methodology introduces a transformative four-step sequence comprising Friedel-Crafts acylation, assisted bromination, epoxidation, and catalytic amine substitution. A pivotal improvement is the introduction of an auxiliary composition during the bromination step, consisting of an oxidant like hydrogen peroxide or peracetic acid combined with a mineral acid. This addition dramatically enhances the utilization of bromine, pushing efficiency well beyond traditional limits. Additionally, the process employs a specialized solid acid catalyst in the final ring-opening step, which significantly boosts reaction selectivity. This novel approach ensures that the transformation from starting material to the final alpha-amino ketone is not only chemically efficient but also economically viable for industrial applications.
Mechanistic Insights into Assisted Bromination and Catalytic Ring-Opening
The chemical elegance of this synthesis lies in its precise control over reaction kinetics and thermodynamics. The process begins with a classic Friedel-Crafts acylation, where an aromatic hydrocarbon reacts with a fatty acid acyl chloride in the presence of anhydrous aluminum chloride to form the ketone precursor. This is followed by the critical bromination step, where the auxiliary composition acts to regenerate active brominating species in situ, thereby minimizing the loss of elemental bromine as waste. The subsequent epoxidation utilizes sodium methoxide in methanol to convert the alpha-bromo ketone into an epoxy intermediate. Finally, the ring-opening reaction is facilitated by a unique solid acid catalyst prepared from treated clays like bentonite or kaolin. This heterogeneous catalyst provides acidic sites that activate the epoxide ring towards nucleophilic attack by the amine, ensuring high regioselectivity and preventing polymerization or degradation side reactions.
Impurity control is rigorously managed throughout this cascade. The use of the auxiliary composition in bromination reduces the formation of poly-brominated by-products, which are common contaminants in standard halogenation reactions. In the final step, the specific activation of the clay catalyst at temperatures between 100°C and 800°C creates a surface chemistry that favors the desired substitution over elimination pathways. This results in a crude product with high purity, often exceeding 95% before recrystallization, and final purified material reaching greater than 99.5% purity. Such tight control over the impurity profile is essential for meeting the stringent specifications required by regulatory bodies for pharmaceutical and agrochemical intermediates.
How to Synthesize Alpha-Amino Aryl Alkyl Ketones Efficiently
Implementing this synthesis requires careful attention to reaction parameters and reagent quality to replicate the high yields reported in the patent literature. The process is designed to be telescoped where possible, minimizing solvent swaps and isolation steps to enhance throughput. Operators must strictly control the temperature during the exothermic bromination phase and ensure the catalyst is properly activated prior to use in the ring-opening stage. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for process chemists aiming to adopt this technology for pilot or commercial production.
- Perform Friedel-Crafts acylation of substituted aromatic hydrocarbons with fatty acid acyl chlorides using anhydrous aluminum chloride.
- Conduct alpha-bromination using an auxiliary composition of hydrogen peroxide or peracetic acid combined with mineral acids to enhance bromine utilization.
- Execute epoxidation with sodium methoxide in methanol, followed by ring-opening with organic amines using a solid acid clay catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible benefits that extend beyond mere chemical yield. The optimization of raw material usage, particularly bromine, directly translates to reduced input costs and a more predictable cost of goods sold. By eliminating the need for excessive reagent equivalents and reducing the generation of hazardous waste, the process aligns with modern sustainability goals while lowering disposal expenses. Furthermore, the robustness of the reaction conditions allows for flexible manufacturing schedules, reducing the risk of batch failures that can disrupt supply continuity. This reliability is crucial for maintaining just-in-time inventory levels in a volatile global market.
- Cost Reduction in Manufacturing: The significant improvement in bromine utilization from less than 50% to over 80% represents a major reduction in raw material consumption. Since bromine is a costly and regulated reagent, this efficiency gain drastically lowers the variable cost per kilogram of the final product. Additionally, the use of inexpensive, earth-abundant clay catalysts instead of precious metals or complex homogeneous catalysts further reduces the catalyst cost burden. The simplified workup procedures, which often involve simple phase separations and crystallizations, minimize solvent usage and energy consumption during distillation and drying phases.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as aromatic hydrocarbons, acyl chlorides, and standard mineral acids ensures that the supply chain is not vulnerable to shortages of exotic reagents. The high selectivity of the process means that fewer batches are rejected due to out-of-specification impurities, leading to a more consistent output volume. This predictability allows supply chain planners to optimize inventory levels and reduce safety stock requirements, freeing up working capital. The ability to source raw materials from multiple vendors without compromising process performance adds another layer of resilience to the supply network.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in multi-hundred-gram to kilogram scales in the patent examples with consistent results. The reduction in three-waste emissions, particularly bromine-containing waste streams, simplifies compliance with increasingly strict environmental regulations. The solid acid catalyst can potentially be regenerated or disposed of with less environmental impact than liquid acid wastes. This clean production profile facilitates easier permitting for new manufacturing lines and reduces the long-term liability associated with waste management, making it an attractive option for sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: How does the new bromination method improve atom economy?
A: By adding an auxiliary composition comprising hydrogen peroxide or peracetic acid along with mineral acids, the bromine utilization rate increases from less than 50% in conventional methods to over 80%, significantly reducing raw material costs and waste.
Q: What is the advantage of the solid acid catalyst in the ring-opening step?
A: The use of acid-treated clay catalysts (such as bentonite or kaolin) in the amine substitution step enhances reaction selectivity to over 98%, minimizing by-product formation and simplifying downstream purification compared to uncatalyzed thermal methods.
Q: What is the overall yield and purity achievable with this process?
A: The patented four-step sequence achieves a total molar yield of 70% to 88% based on the starting aromatic hydrocarbon, with final product purity exceeding 99.5% as determined by HPLC analysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Amino Aryl Alkyl Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation therapeutics and agrochemicals. Our technical team has extensively analyzed the pathway described in CN101633647A and possesses the expertise to execute this complex four-step synthesis with precision. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from laboratory bench to full-scale manufacturing. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including HPLC analysis and residual solvent testing, to guarantee that every batch meets your exacting standards.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific application needs. Our team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this high-yield route for your specific target molecule. Please contact our technical procurement team today to request specific COA data from our pilot runs and to discuss detailed route feasibility assessments tailored to your project timeline and volume requirements.
