Revolutionizing Omeprazole Intermediate Production via Continuous Flow Technology and Commercial Scale-Up
Revolutionizing Omeprazole Intermediate Production via Continuous Flow Technology and Commercial Scale-Up
The pharmaceutical industry is currently witnessing a paradigm shift in the manufacturing of critical intermediates, driven by the urgent need for safer, more efficient, and scalable synthesis routes. Patent CN111704555A, published in September 2020, introduces a groundbreaking method for synthesizing 4-methoxy-2-nitroaniline, a pivotal precursor in the production of omeprazole, utilizing advanced continuous flow reactor technology. This innovation addresses the longstanding safety hazards and inefficiencies associated with traditional batch processing, particularly during the highly exothermic nitration step. By transitioning from static kettle reactors to a dynamic continuous flow system, the process achieves exceptional control over reaction parameters, resulting in significantly improved selectivity and yield. For global supply chain leaders and R&D directors, this technology represents not just a chemical optimization, but a strategic asset for ensuring consistent supply of high-purity pharmaceutical intermediates while mitigating operational risks inherent in large-scale nitration chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 4-methoxy-2-nitroaniline has relied heavily on intermittent batch processes conducted in static kettle reactors, a methodology fraught with significant technical and safety challenges. The nitration step, which is inherently exothermic, poses a severe risk of thermal runaway; as the reaction temperature rises, the rate of heat generation accelerates, often outpacing the cooling capacity of traditional vessels. This imbalance can lead to catastrophic failures, including fires and explosions, necessitating rigorous and costly safety protocols. Furthermore, the poor heat and mass transfer characteristics of batch reactors result in non-uniform temperature distribution, which promotes undesirable side reactions such as multi-nitration and sulfonation. These side reactions generate impurities that are structurally similar to the target product, making downstream purification arduous and leading to substantial yield losses, with some prior art reporting yields as low as 60% for the nitration step alone.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages a fully integrated continuous flow system comprising three distinct reactor stages to orchestrate acetylation, nitration, and hydrolysis seamlessly. This methodology fundamentally alters the reaction environment by maximizing the surface-area-to-volume ratio, thereby facilitating rapid and efficient heat dissipation that effectively eliminates the risk of thermal runaway. The continuous flow architecture allows for precise regulation of residence time and stoichiometric ratios through metering pumps, ensuring that reactants are mixed instantly and uniformly. This level of control suppresses the formation of isomeric by-products, such as 4-methoxy-3-nitroaniline, and enables the reaction to proceed with high selectivity. Crucially, the system is designed to operate without the need for isolating intermediate products, which streamlines the workflow and reduces the overall processing time from hours to mere minutes or seconds for specific steps.
Mechanistic Insights into Continuous Flow Acetylation-Nitration-Hydrolysis
The core of this technological advancement lies in the precise orchestration of three consecutive chemical transformations within a closed flow system. The process initiates with the acetylation of 4-methoxyaniline using acetic anhydride in a solvent such as glacial acetic acid or dichloromethane, forming 4-methoxyacetanilide. This protected intermediate is then immediately subjected to nitration using a mixed acid reagent (concentrated sulfuric and nitric acid) within the second reactor stage. The continuous flow environment ensures that the highly reactive nitronium ion interacts with the substrate under strictly controlled thermal conditions, typically between 0°C and 100°C, preventing the degradation of the sensitive aniline derivative. Finally, the nitrated acetanilide undergoes hydrolysis in the third reactor stage using a base like sodium hydroxide or an acid, cleaving the acetyl group to reveal the free amine of the final product. 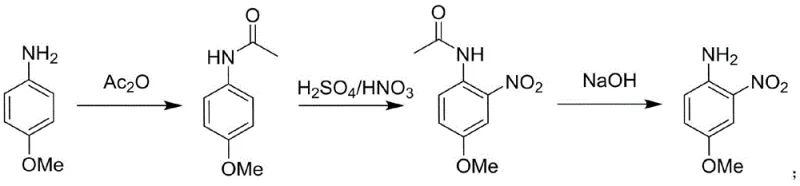
From an impurity control perspective, the continuous flow mechanism offers distinct advantages over batch processing by minimizing the residence time distribution of reactants. In a batch reactor, some molecules may remain in the reaction zone longer than others, leading to over-reaction and the formation of di-nitrated species or oxidized by-products. The plug-flow nature of the reactor system ensures that all fluid elements experience nearly identical reaction conditions and times, drastically narrowing the impurity profile. This uniformity is critical for pharmaceutical applications where strict limits on genotoxic impurities and isomers are enforced. The ability to tune flow rates dynamically allows operators to optimize the balance between conversion and selectivity in real-time, ensuring that the final crude product emerging from the third reactor already possesses a purity profile exceeding 99% before any crystallization occurs.
How to Synthesize 4-Methoxy-2-Nitroaniline Efficiently
Implementing this continuous flow synthesis requires a systematic approach to equipment setup and parameter optimization to replicate the high yields and safety profiles demonstrated in the patent literature. The process relies on the synchronized operation of four metering pumps to deliver the aniline solution, acetic anhydride, nitration reagent, and hydrolysate into their respective reactor zones at precise volumetric ratios. Operators must carefully calibrate the flow rates to maintain the desired residence times, which can range from seconds for rapid acetylation to minutes for hydrolysis, depending on the specific thermal conditions employed. The following guide outlines the standardized procedural framework derived from the patent examples, serving as a foundational reference for process engineers aiming to transition from batch to flow chemistry.
- Acetylation: Pump 4-methoxyaniline solution and acetic anhydride into Reactor I at controlled flow rates (e.g., 1:0.5 ratio) to form 4-methoxyacetanilide.
- Nitration: Mix the acetylation stream with mixed acid (H2SO4/HNO3) in Reactor II, maintaining precise temperature control to prevent runaway reactions.
- Hydrolysis & Workup: React the nitrated stream with hydrolysate (e.g., NaOH) in Reactor III, followed by cooling, crystallization, and filtration to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this continuous flow technology translates into tangible strategic benefits that extend far beyond simple chemical yield improvements. The elimination of intermediate isolation steps significantly reduces the consumption of solvents and energy required for multiple workups and drying processes, leading to a leaner and more cost-effective manufacturing operation. Furthermore, the inherent safety of the flow system lowers the insurance premiums and facility infrastructure costs associated with handling hazardous nitration reactions on a large scale. By mitigating the risk of batch failures and explosions, manufacturers can ensure a more reliable and uninterrupted supply of this critical omeprazole intermediate, safeguarding against market volatility and production delays.
- Cost Reduction in Manufacturing: The continuous flow process eliminates the need for expensive and time-consuming purification of intermediate compounds, which traditionally accounts for a significant portion of production costs. By integrating the three reaction steps into a single streamlined sequence, the method reduces labor hours, solvent usage, and waste disposal fees. The high selectivity of the reaction minimizes the loss of raw materials to by-products, ensuring that a greater proportion of the input 4-methoxyaniline is converted into valuable final product, thereby optimizing the overall material cost structure without compromising quality.
- Enhanced Supply Chain Reliability: Traditional batch processes are susceptible to variability and scale-up effects, where yields often drop when moving from pilot to production scale. This continuous flow method demonstrates no amplification effect, meaning the performance observed in small-scale trials is directly transferable to commercial production volumes. This consistency allows supply chain planners to forecast output with greater accuracy and confidence. Additionally, the reduced reaction times and simplified post-treatment procedures enable faster turnaround times, allowing manufacturers to respond more agilely to fluctuations in demand for omeprazole and related pharmaceutical products.
- Scalability and Environmental Compliance: The modular nature of continuous flow reactors facilitates straightforward scalability; increasing production capacity often involves running the system for longer durations or numbering up reactor units rather than building massive, complex batch vessels. This approach aligns well with modern environmental regulations by reducing the overall footprint of the chemical plant and minimizing the generation of hazardous waste. The precise control over reagent dosing prevents the excess use of corrosive acids and bases, simplifying effluent treatment and ensuring compliance with increasingly stringent global environmental standards for pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the continuous flow synthesis of 4-methoxy-2-nitroaniline, based on the detailed specifications and experimental data provided in the patent documentation. These insights are intended to clarify the feasibility and advantages of implementing this technology in a commercial setting.
Q: How does continuous flow technology improve safety in the nitration of 4-methoxyaniline?
A: Traditional batch nitration is highly exothermic and prone to thermal runaway. Continuous flow reactors offer superior heat exchange efficiency due to their high surface-area-to-volume ratio, allowing for precise temperature control and preventing explosion risks associated with heat accumulation.
Q: What purity levels can be achieved using this continuous flow method?
A: According to patent data, this method consistently achieves product purity exceeding 99% (by HPLC) with a total three-step yield over 85%, significantly reducing the formation of isomeric by-products like 4-methoxy-3-nitroaniline.
Q: Does this process require intermediate purification steps?
A: No. A key advantage of this integrated continuous flow design is that the reaction streams proceed directly from acetylation to nitration to hydrolysis without the need to isolate or purify intermediate compounds, drastically simplifying the operational workflow.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Methoxy-2-Nitroaniline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing technologies like continuous flow chemistry is essential for meeting the rigorous demands of the modern pharmaceutical industry. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-quality intermediates. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 4-methoxy-2-nitroaniline meets the exacting standards required for API synthesis. We are committed to leveraging innovations such as the continuous flow nitration process to enhance our service offerings and deliver superior value to our global partners.
We invite procurement teams and R&D leaders to collaborate with us to explore how this optimized synthesis route can benefit your specific supply chain requirements. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume needs, along with specific COA data and route feasibility assessments. Let us help you secure a stable, cost-effective, and safe supply of this critical pharmaceutical building block, driving efficiency and reliability in your omeprazole production lines.
