Advanced MOF-Catalyzed Synthesis of Voranolan Fumarate Impurity for Commercial Scale-Up
Advanced MOF-Catalyzed Synthesis of Voranolan Fumarate Impurity for Commercial Scale-Up
The pharmaceutical industry's relentless pursuit of high-purity reference standards has led to significant innovations in the synthesis of complex drug impurities, particularly for potent acid blockers like Vonoprazan. Patent CN113214223A introduces a groundbreaking preparation method for Voranolan fumarate impurity, utilizing advanced Metal-Organic Framework (MOF) coordination compounds as catalysts. This technology represents a paradigm shift from traditional heterogeneous catalysis, offering superior selectivity and milder reaction conditions that are critical for maintaining the structural integrity of sensitive pyrrole-sulfonyl scaffolds. For R&D directors and procurement specialists, this patent outlines a pathway to obtaining high-purity impurities essential for regulatory compliance and method validation. The integration of iron, aluminum, or chromium-based MOFs not only enhances reaction efficiency but also simplifies downstream processing, making it a highly attractive solution for industrial-scale manufacturing of pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the methylation of complex heterocyclic amines like Compound II has relied heavily on molecular sieves or phase transfer catalysts, which present substantial operational challenges in a commercial setting. These conventional catalysts typically require aggressive reaction temperatures ranging from 120°C to 130°C to drive the conversion, which poses a significant risk of thermal degradation to the sensitive fluorophenyl and pyridine moieties within the molecule. Furthermore, the selectivity of these traditional methods is often compromised, leading to the formation of numerous unidentified by-products that complicate the purification landscape and drastically reduce the overall yield of the target intermediate. The resulting crude mixtures frequently exhibit purities below 80%, necessitating extensive and costly chromatographic purification steps that are difficult to scale and generate significant solvent waste. From a supply chain perspective, the reliance on harsh conditions and inefficient catalysts translates to longer batch cycles and inconsistent quality, creating bottlenecks in the production of critical reference materials.
The Novel Approach
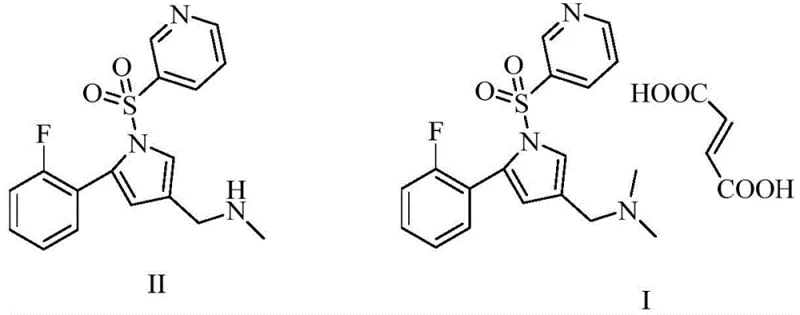
In stark contrast, the novel approach detailed in the patent leverages the unique porous structure and tunable acidity of Metal-Organic Frameworks to facilitate methylation under remarkably mild conditions. By employing MOFs formed from metals such as aluminum, iron, or chromium coordinated with 1,3,5-tris(4-carboxyphenyl)benzene, the reaction temperature can be effectively lowered to a range of 60°C to 110°C, with optimal results observed between 80°C and 90°C. This reduction in thermal stress preserves the chemical stability of the substrate while simultaneously enhancing the selectivity of the methylation at the desired nitrogen atom. The process utilizes dimethyl carbonate as a green methylating agent, which, in conjunction with the MOF catalyst and a base like potassium carbonate, drives the reaction to completion with exceptional efficiency. As illustrated in the reaction scheme below, this methodology transforms Compound II into the intermediate Compound III with yields exceeding 97% and purities above 95%, setting a new benchmark for process robustness.
Mechanistic Insights into MOF-Catalyzed Methylation
The efficacy of this synthesis lies in the precise interaction between the substrate and the active sites within the Metal-Organic Framework catalyst. Unlike amorphous molecular sieves, MOFs possess a highly ordered crystalline structure with uniform pore sizes that can be engineered to specifically accommodate the transition state of the methylation reaction. The metal nodes (Al, Fe, or Cr) within the framework act as Lewis acid sites, activating the dimethyl carbonate and facilitating the nucleophilic attack by the secondary amine of Compound II. This cooperative catalysis ensures that the methyl group is transferred selectively to the target nitrogen without affecting other sensitive functional groups, such as the sulfonyl linkage or the fluorine substituent. The aluminum-based MOF, in particular, demonstrates superior performance, likely due to the optimal balance of Lewis acidity and steric environment provided by the trimesic acid-derived linkers. This mechanistic precision is what allows the reaction to proceed rapidly at lower temperatures, minimizing energy consumption and reducing the formation of thermal decomposition products.
Furthermore, the mechanism inherently supports superior impurity control, which is a paramount concern for the production of reference standards. The high selectivity of the MOF catalyst means that side reactions, such as over-methylation or hydrolysis of the ester groups, are significantly suppressed compared to traditional methods. The patent data indicates that the intermediate Compound III is obtained with a purity of up to 96.72%, which directly correlates to the high purity of the final fumarate salt. The subsequent salt formation with fumaric acid is straightforward, occurring at mild temperatures of 40°C to 50°C, and the final product can be isolated with a purity exceeding 99% through simple crystallization. This level of purity is achieved without the need for complex column chromatography, relying instead on the inherent selectivity of the catalytic step and the differential solubility of the product in solvents like ethyl acetate and methanol/water mixtures.
How to Synthesize Voranolan Fumarate Impurity Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible roadmap for laboratories aiming to produce this critical impurity standard. The process begins with the in-situ or ex-situ preparation of the MOF catalyst, followed by the methylation reaction in a polar aprotic solvent such as N,N-dimethylacetamide. The reaction is monitored via TLC to ensure complete conversion before proceeding to the workup, which involves a simple filtration to remove the solid catalyst and excess base. The filtrate is then treated with fumaric acid to induce salt formation, followed by crystallization to isolate the high-purity product. This streamlined workflow eliminates many of the unit operations associated with traditional synthesis, such as extensive washing or distillation, thereby reducing the overall processing time and solvent usage. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized synthesis guide below.
- Prepare the MOF catalyst by reacting metal chloride (Al, Fe, or Cr) with 1,3,5-tris(4-carboxyphenyl)benzene in DMF at 140-180°C.
- Perform methylation of Compound II with dimethyl carbonate using the MOF catalyst and potassium carbonate in DMAc at 80-90°C.
- Filter the reaction mixture, react the filtrate with fumaric acid at 40-50°C, and crystallize the final impurity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this MOF-catalyzed process offers tangible strategic advantages that extend beyond mere technical performance. The ability to operate at significantly lower temperatures reduces the energy load on manufacturing facilities and lowers the risk of safety incidents associated with high-pressure or high-temperature reactors. Moreover, the high selectivity of the catalyst minimizes the generation of hazardous waste streams, aligning with increasingly stringent environmental regulations and reducing the costs associated with waste disposal and treatment. The simplified downstream processing, characterized by simple filtration and crystallization rather than complex chromatography, drastically shortens the production cycle time, allowing for faster turnaround on custom synthesis orders. This efficiency gain translates directly into improved supply chain reliability, ensuring that critical impurity standards are available when needed for regulatory filings and quality control testing without lengthy lead times.
- Cost Reduction in Manufacturing: The elimination of expensive and inefficient molecular sieves in favor of highly active MOF catalysts leads to a more economical use of raw materials and reagents. By achieving higher yields and purities in the initial reaction step, the need for costly reprocessing or extensive purification is virtually eliminated, resulting in substantial overall cost savings. The use of dimethyl carbonate as a methylating agent is also economically favorable due to its low cost and low toxicity compared to traditional alkylating agents like methyl iodide. Additionally, the reduced reaction temperature lowers utility costs, contributing to a more sustainable and cost-effective manufacturing profile that enhances the competitiveness of the final product in the global market.
- Enhanced Supply Chain Reliability: The robustness of the MOF-catalyzed process ensures consistent batch-to-batch quality, which is critical for maintaining the integrity of the supply chain for pharmaceutical intermediates. The availability of the key catalyst components, such as aluminum chloride and trimesic acid derivatives, ensures that production is not dependent on scarce or geopolitically sensitive materials. The simplified workflow reduces the number of potential failure points in the manufacturing process, thereby minimizing the risk of production delays or batch failures. This reliability allows suppliers to offer more predictable delivery schedules and maintain adequate inventory levels of high-purity impurities, supporting the continuous operation of their clients' quality assurance and R&D departments.
- Scalability and Environmental Compliance: The process is inherently scalable, as the heterogeneous nature of the MOF catalyst facilitates easy separation and potential reuse, which is a key factor in transitioning from laboratory to commercial scale. The reduction in solvent usage and the avoidance of toxic reagents align with green chemistry principles, making the process easier to permit and operate in regulated environments. The high purity of the final product reduces the burden on wastewater treatment systems, as fewer organic impurities need to be degraded. This environmental compatibility not only reduces regulatory compliance costs but also enhances the corporate social responsibility profile of the manufacturing entity, appealing to environmentally conscious partners in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Voranolan fumarate impurity using the patented MOF technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a reliable basis for decision-making. Understanding the nuances of catalyst selection, reaction conditions, and purification strategies is essential for optimizing the production of this high-value intermediate. The answers below reflect the current state of the art as defined by the intellectual property surrounding this specific chemical transformation.
Q: What are the advantages of using MOF catalysts over molecular sieves for this synthesis?
A: MOF catalysts allow for significantly lower reaction temperatures (80-90°C vs 120-130°C) and achieve higher purity (>99%) compared to traditional molecular sieves which often result in lower purity intermediates.
Q: Is the catalyst reusable in this process?
A: The process involves a simple filtration step to remove the solid catalyst after the reaction, suggesting potential for recovery, although the patent focuses on the high selectivity and ease of separation rather than explicit recycling loops.
Q: What is the expected purity of the final Voranolan fumarate impurity?
A: The patented method consistently achieves a final product purity of over 99%, with intermediate yields exceeding 97%, ensuring high-quality reference standards for pharmaceutical analysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Voranolan Fumarate Impurity Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity impurities play in the development and quality control of modern pharmaceuticals like Vonoprazan. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your demands whether for milligram-scale analytical standards or kilogram-scale process validation batches. We adhere to stringent purity specifications and utilize rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Voranolan fumarate impurity meets the highest industry standards. Our commitment to technical excellence allows us to replicate and optimize complex synthetic routes, such as the MOF-catalyzed methylation described in recent patents, delivering products that support your regulatory submissions and research goals with unwavering consistency.
We invite you to collaborate with us to leverage these advanced synthetic technologies for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality specifications. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in fine chemical synthesis can enhance your supply chain efficiency and product quality. Together, we can navigate the complexities of pharmaceutical intermediate manufacturing with confidence and precision.
