Advanced Metal-Free Synthesis of Nitrogen-Containing Fused Rings for Commercial API Production
Advanced Metal-Free Synthesis of Nitrogen-Containing Fused Rings for Commercial API Production
The landscape of organic synthesis for complex heterocyclic scaffolds is undergoing a significant paradigm shift, driven by the urgent need for greener, more sustainable, and cost-effective manufacturing processes. Patent CN115260192B introduces a groundbreaking methodology for the preparation of nitrogen-containing fused ring compounds, specifically utilizing an N-heterocyclic carbene (NHC) catalytic system. This innovation addresses critical bottlenecks in the production of high-value pharmaceutical intermediates and agrochemical building blocks by replacing traditional transition metal catalysis with an efficient organocatalytic approach. The disclosed technology enables the construction of intricate polycyclic architectures, such as pyrido[4,3,2-gh]phenanthridinones, which are prevalent in bioactive natural products and therapeutic agents, through a radical tandem-cyclization strategy that operates under exceptionally mild conditions.
For research and development directors overseeing process chemistry, the implications of this patent are profound. The ability to synthesize these complex nitrogenous frameworks without the involvement of expensive and toxic heavy metals like iridium, ruthenium, or silver represents a major advancement in process safety and regulatory compliance. Traditional methods often struggle with regioselectivity issues and the generation of hazardous waste streams due to the requirement for stoichiometric oxidants. In contrast, the NHC-catalyzed route described in CN115260192B offers high chemical selectivity and atom economy, facilitating the production of high-purity intermediates essential for downstream drug development. This technological leap not only streamlines the synthetic pathway but also aligns perfectly with the global industry's push towards green chemistry principles and sustainable manufacturing practices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing fused heterocycles has relied heavily on transition metal-catalyzed radical tandem-cyclization reactions or photocatalytic processes. While effective in academic settings, these conventional methodologies present severe limitations when scaled for industrial application. The primary concern is the reliance on precious metals such as palladium, iridium, or silver, which are not only costly but also pose significant toxicity risks. In the context of active pharmaceutical ingredient (API) manufacturing, the presence of residual heavy metals is strictly regulated, necessitating additional, expensive purification steps such as metal scavenging or recrystallization to meet stringent quality specifications. Furthermore, many oxidative coupling reactions in these traditional routes require the use of stoichiometric amounts of strong oxidants, leading to the generation of substantial quantities of chemical waste and creating environmental disposal challenges that increase the overall carbon footprint of the manufacturing process.
Beyond the economic and environmental drawbacks, conventional transition metal methods often suffer from poor regioselectivity, resulting in complex mixtures of isomers that are difficult to separate. This lack of selectivity can drastically reduce the overall yield of the desired product and complicate the purification workflow, thereby extending production timelines and increasing operational costs. Additionally, the harsh reaction conditions frequently associated with metal catalysis, such as high temperatures or strong acidic/basic environments, can be incompatible with sensitive functional groups present in advanced intermediates. These factors collectively hinder the widespread application of traditional methods in the large-scale production of complex drug candidates, creating a pressing need for alternative synthetic strategies that are both robust and environmentally benign.
The Novel Approach
The novel approach detailed in patent CN115260192B circumvents these challenges by employing an organic small molecule catalyst, specifically an N-heterocyclic carbene (NHC), to drive the cyclization of alpha-bromo N-aryl acrylamides. This metal-free strategy operates under mild conditions, typically at room temperature, utilizing cesium carbonate as a base and 1,4-dioxane as the solvent. The elimination of transition metals fundamentally alters the impurity profile of the reaction, removing the risk of heavy metal contamination and simplifying the downstream purification process significantly. By avoiding the need for expensive metal catalysts and stoichiometric oxidants, this method offers a inherently greener and more cost-effective pathway for constructing the nitrogen-containing fused ring core. The reaction proceeds via a radical mechanism that exhibits high chemoselectivity and regioselectivity, ensuring that the desired polycyclic product is formed predominantly without the formation of troublesome byproducts.
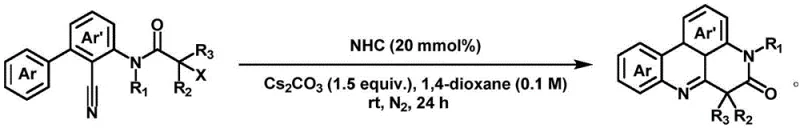
Moreover, the versatility of this NHC-catalyzed system is demonstrated by its broad substrate scope, accommodating a wide range of electronic substituents on the aromatic rings. Whether the substrate contains electron-donating groups like methoxy or tert-butyl, or electron-withdrawing groups like cyano, the reaction proceeds efficiently to deliver the target fused ring compounds in high yields. This robustness makes the method highly attractive for the synthesis of diverse chemical libraries required for structure-activity relationship (SAR) studies in drug discovery. The operational simplicity of the protocol, which involves mixing reagents in a Schlenk tube under a nitrogen atmosphere followed by standard workup procedures, further enhances its appeal for commercial scale-up. For procurement and supply chain managers, this translates to a more reliable and predictable manufacturing process that is less susceptible to the volatility of precious metal markets and regulatory changes regarding heavy metal residues.
Mechanistic Insights into NHC-Catalyzed Radical Cyclization
The mechanistic underpinning of this transformation involves the generation of radical species facilitated by the N-heterocyclic carbene catalyst, which initiates a cascade cyclization sequence. Unlike transition metal catalysis that often relies on oxidative addition and reductive elimination cycles involving metal centers, the NHC-mediated pathway leverages the unique electronic properties of the carbene to activate the alpha-bromo amide substrate. The catalyst likely interacts with the substrate to promote homolytic cleavage of the carbon-bromine bond, generating a carbon-centered radical that subsequently undergoes intramolecular addition to the adjacent unsaturated system. This radical tandem-cyclization process is highly efficient, constructing multiple bonds and rings in a single operation, which is a hallmark of ideal synthetic design. The mild basic conditions provided by cesium carbonate assist in deprotonation steps or stabilizing intermediates without promoting unwanted side reactions that are common under harsher acidic or strongly basic conditions.
From an impurity control perspective, the metal-free nature of this catalytic cycle is a decisive advantage. In traditional metal-catalyzed reactions, trace amounts of metal can coordinate with product molecules or intermediates, leading to stable complexes that are difficult to remove and can catalyze decomposition during storage. The NHC system avoids these coordination issues entirely, resulting in a cleaner reaction profile. Furthermore, the high regioselectivity observed in this process suggests that the radical intermediate is well-controlled, likely due to steric and electronic guidance provided by the substrate structure and the reaction environment. This precision minimizes the formation of regioisomers, which are often the most challenging impurities to separate chromatographically. For R&D teams, understanding this mechanism allows for the rational design of substrates with specific substituents to further tune the reactivity and selectivity, optimizing the process for specific target molecules within the nitrogen-containing fused ring class.
How to Synthesize Nitrogen-Containing Fused Ring Compounds Efficiently
The practical implementation of this synthesis involves a straightforward protocol that is amenable to both laboratory scale and potential commercial production. The process begins with the precise weighing of the alpha-bromo N-aryl acrylamide starting material, the NHC catalyst, and the cesium carbonate base, which are then charged into a reaction vessel such as a Schlenk tube. The system is subjected to vacuum-nitrogen cycles to ensure an oxygen-free environment, which is crucial for the stability of the radical intermediates involved in the cyclization. Upon the addition of the 1,4-dioxane solvent, the mixture is stirred at ambient temperature, allowing the reaction to proceed to completion as monitored by thin-layer chromatography (TLC). This operational simplicity reduces the need for specialized high-pressure or high-temperature equipment, lowering the capital expenditure required for setting up the production line.
- Charge a Schlenk tube with alpha-bromo N-aryl acrylamide substrate, cesium carbonate base, and NHC catalyst under inert atmosphere.
- Add 1,4-dioxane solvent and stir the mixture at room temperature or mild heating until TLC indicates complete consumption of starting material.
- Remove solvent under reduced pressure and purify the crude residue via column chromatography to isolate the high-purity fused ring product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this NHC-catalyzed technology offers tangible strategic advantages that directly impact the bottom line and operational resilience. The most significant benefit is the drastic reduction in raw material costs associated with the elimination of precious metal catalysts. By substituting expensive iridium or palladium complexes with an organic small molecule catalyst, the direct material cost of the synthesis is significantly lowered. Furthermore, the removal of heavy metals from the process flow eliminates the need for costly metal scavenger resins and the associated validation testing required to prove low metal levels in the final API. This simplification of the purification train not only saves money but also shortens the overall production cycle time, allowing for faster turnaround from synthesis to final product release.
- Cost Reduction in Manufacturing: The economic benefits of this metal-free approach extend beyond just the cost of the catalyst itself. By operating at room temperature, the process consumes significantly less energy compared to traditional methods that require prolonged heating or cryogenic cooling. The high selectivity of the reaction means that less starting material is wasted on byproducts, improving the overall mass balance and yield of the process. Additionally, the simplified workup procedure, which typically involves solvent removal and column chromatography without complex aqueous extractions or metal filtration steps, reduces the consumption of solvents and consumables. These cumulative efficiencies result in a substantially lower cost of goods sold (COGS), making the final nitrogen-containing fused ring intermediates more competitive in the global market.
- Enhanced Supply Chain Reliability: Relying on transition metals for critical synthetic steps introduces supply chain vulnerabilities, as the availability and price of metals like palladium and iridium can be highly volatile and subject to geopolitical instability. By shifting to an organocatalytic system based on readily available organic precursors and common inorganic bases like cesium carbonate, the supply chain becomes much more robust and predictable. The raw materials for the NHC catalyst and the substrates are generally easier to source from multiple vendors, reducing the risk of single-source dependency. This diversification of the supply base ensures continuity of supply even during market disruptions, providing a strategic buffer for long-term production planning and inventory management.
- Scalability and Environmental Compliance: The green chemistry attributes of this process facilitate easier regulatory approval and environmental compliance. The absence of heavy metals simplifies the environmental impact assessment and waste disposal protocols, as the effluent streams do not require specialized treatment for metal removal. This aligns with increasingly stringent environmental regulations globally, reducing the risk of compliance-related shutdowns or fines. Moreover, the mild reaction conditions and simple equipment requirements make the process highly scalable from gram to kilogram and eventually ton scales without significant re-engineering. The ability to scale up smoothly ensures that the technology can meet growing market demand for these valuable intermediates without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this NHC-catalyzed synthesis technology. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on the practical aspects of adopting this method for industrial applications. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: Why is NHC catalysis preferred over transition metals for pharmaceutical intermediates?
A: Transition metal catalysts often leave toxic residues that require expensive removal steps to meet strict pharmacopeial standards. NHC organocatalysis eliminates heavy metals entirely, simplifying purification and ensuring higher safety profiles for API manufacturing.
Q: What are the typical reaction conditions for this cyclization process?
A: The process operates under remarkably mild conditions, typically at room temperature in 1,4-dioxane with cesium carbonate as the base. This reduces energy consumption and minimizes thermal degradation of sensitive functional groups.
Q: Does this method support diverse substrate scopes for drug discovery?
A: Yes, the methodology demonstrates excellent tolerance for various electronic groups including methoxy, thioether, cyano, and tert-butyl substituents, allowing for the rapid generation of diverse chemical libraries for SAR studies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitrogen-Containing Fused Ring Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free NHC-catalyzed synthesis described in CN115260192B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of nitrogen-containing fused ring compounds delivered meets the highest quality standards required by global regulatory bodies. We are committed to leveraging our technical expertise to optimize this green synthesis route for our clients, delivering cost-effective and sustainable solutions.
We invite pharmaceutical and agrochemical companies to collaborate with us to explore the full potential of this advanced synthetic methodology. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how this metal-free approach can optimize your budget. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your target molecules. Let us help you secure a reliable supply of high-purity intermediates while driving down costs and enhancing the sustainability of your supply chain.
