Advanced Palladium-Catalyzed Synthesis of 4-Aryl Isoxazole Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, which serve as the backbone for countless bioactive molecules. Patent CN112645896A introduces a groundbreaking approach for the synthesis of 4-aryl isoxazole derivatives, addressing critical bottlenecks in current manufacturing processes. This innovation utilizes a palladium-catalyzed cascade reaction that couples O-benzyl alkynone oxime ethers with diaryl ether derivatives, offering a streamlined pathway to highly functionalized isoxazole scaffolds. Unlike traditional methods that rely on scarce or expensive reagents, this technique leverages the abundance of diaryl ethers, positioning it as a transformative solution for cost reduction in pharmaceutical intermediate manufacturing. The method demonstrates exceptional functional group tolerance, accommodating halogens, heterocycles, and alkenyl groups without degradation, which is paramount for the synthesis of complex drug candidates. By integrating this technology, manufacturers can achieve higher purity profiles and improved process safety, aligning with the rigorous standards required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 4-aryl isoxazole derivatives has relied heavily on palladium-catalyzed direct C-H bond functionalization using aryl halides, particularly aryl iodides. Pioneering work by researchers such as Nakamura demonstrated the feasibility of Pd/C catalyzed arylation, yet these protocols suffered from significant limitations that hindered their industrial adoption. The reported yields for these conventional reactions were notoriously low, often ranging between 30% and 48%, which translates to substantial material waste and increased production costs. Furthermore, the reliance on aryl iodides introduces economic volatility, as these reagents are generally more expensive and less stable than their ether counterparts. The narrow substrate scope of earlier methods also restricted the diversity of accessible chemical space, making it difficult to synthesize derivatives with sensitive functional groups required for modern medicinal chemistry applications. Additionally, many traditional protocols required harsh conditions or stoichiometric amounts of oxidants, raising concerns regarding environmental impact and operational safety in large-scale facilities.
The Novel Approach
In stark contrast to these legacy methods, the novel synthetic route disclosed in the patent utilizes diaryl ether derivatives as the aryl source, unlocking a vast reservoir of renewable and inexpensive starting materials. This innovative strategy employs a one-step palladium-catalyzed cyclization and arylation cascade, effectively merging two distinct bond-forming events into a single operational unit. The reaction proceeds under relatively mild thermal conditions, typically between 100°C and 150°C, and utilizes basic compounds such as pyridine or carbonates to facilitate the transformation. A key feature of this approach is its compatibility with ionic liquids as solvents, which not only enhances reaction efficiency but also aligns with green chemistry principles by reducing volatile organic compound emissions. The versatility of this method is exemplified by its ability to tolerate a wide array of substituents on both the oxime ether and the diaryl ether components, enabling the rapid generation of diverse chemical libraries. 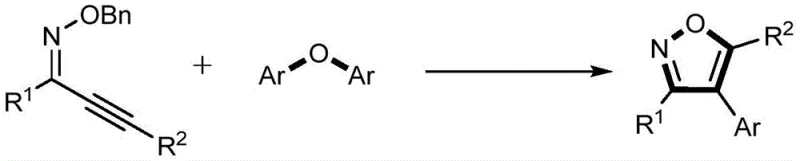
Mechanistic Insights into Pd-Catalyzed Cyclization/Arylation Cascade
The core of this synthetic breakthrough lies in the sophisticated mechanistic pathway driven by the palladium catalyst, specifically N-heterocyclic carbene (NHC) palladium complexes. The reaction initiates with the oxidative addition of the palladium species into the C-O bond of the diaryl ether, a challenging transformation that is facilitated by the electron-rich nature of the NHC ligand. Following this activation, the alkyne moiety of the O-benzyl alkynone oxime ether coordinates to the metal center, setting the stage for the subsequent cyclization event. 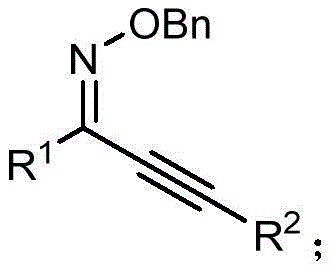
This intramolecular nucleophilic attack by the oxime nitrogen onto the activated alkyne generates the isoxazole ring system while simultaneously forming a new carbon-carbon bond with the aryl group derived from the ether. The catalytic cycle is completed through a reductive elimination step that releases the final 4-aryl isoxazole product and regenerates the active palladium species for further turnover. Crucially, the use of ionic liquids as the reaction medium appears to stabilize the cationic palladium intermediates, thereby suppressing side reactions and improving overall turnover numbers. This mechanistic understanding allows for precise control over impurity profiles, ensuring that the final product meets the stringent purity specifications demanded by high-purity pharmaceutical intermediates markets. The robustness of the catalyst system ensures that even sterically hindered substrates can be converted efficiently, broadening the applicability of this technology across various therapeutic areas.
How to Synthesize 4-Aryl Isoxazole Derivatives Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize yield and minimize impurities, leveraging the optimized conditions detailed in the patent examples. The process begins with the preparation of the reaction mixture under an inert atmosphere to prevent catalyst deactivation by oxygen, utilizing precise molar ratios of the oxime ether and diaryl ether substrates. Operators should select the appropriate base and solvent system, with pyridine and imidazolium-based ionic liquids showing superior performance in benchmarking studies. Detailed standardized synthesis steps see the guide below.
- Under a protective nitrogen atmosphere, combine O-benzyl alkynone oxime ether, diaryl ether derivative, alkaline compound (e.g., pyridine or sodium carbonate), and a palladium catalyst (preferably N-heterocyclic carbene palladium chloride) in a suitable solvent such as an ionic liquid or DMSO.
- Heat the reaction mixture to a temperature between 100°C and 150°C, preferably around 140°C, and maintain stirring for a duration of 10 to 16 hours to ensure complete conversion via the cyclization/arylation cascade.
- Upon completion, cool the reaction to room temperature, concentrate the mixture under reduced pressure to remove solvents, and purify the crude residue using column chromatography with a petroleum ether and ethyl acetate gradient to isolate the high-purity 4-aryl isoxazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling strategic advantages that extend beyond mere technical feasibility. By shifting the feedstock from expensive aryl halides to abundant diaryl ethers, companies can significantly decouple their production costs from the volatility of the halogenated reagent market. The simplified one-pot nature of the reaction reduces the number of unit operations required, leading to lower energy consumption and reduced labor overheads in the manufacturing plant. Furthermore, the high functional group tolerance minimizes the need for complex protection and deprotection sequences, which are often the most time-consuming and waste-generating steps in multi-step synthesis. This efficiency gain directly translates to shorter lead times and enhanced responsiveness to market demands for reliable pharmaceutical intermediate suppliers.
- Cost Reduction in Manufacturing: The substitution of costly aryl iodides with readily available diaryl ethers represents a fundamental shift in raw material economics, driving down the bill of materials for every kilogram produced. The high catalytic efficiency of the NHC-palladium system allows for lower catalyst loadings while maintaining high conversion rates, further reducing the cost burden associated with precious metal usage. Additionally, the ability to use recyclable ionic liquids as solvents contributes to long-term operational savings by minimizing solvent purchase and disposal costs. These cumulative factors create a leaner cost structure that enhances competitiveness in the global supply chain for complex organic molecules.
- Enhanced Supply Chain Reliability: Diaryl ethers are commodity chemicals with stable and diversified supply chains, reducing the risk of production stoppages due to raw material shortages. The robustness of the reaction conditions means that the process is less sensitive to minor variations in input quality, ensuring consistent output even when sourcing from different vendors. This reliability is critical for maintaining continuous manufacturing schedules and meeting the just-in-time delivery requirements of downstream API producers. By securing a synthesis route based on abundant feedstocks, organizations can build a more resilient supply network capable of withstanding geopolitical or logistical disruptions.
- Scalability and Environmental Compliance: The reaction operates at moderate temperatures and does not require high-pressure equipment, making it inherently safer and easier to scale from pilot plant to commercial tonnage production. The use of ionic liquids and the avoidance of stoichiometric oxidants align with increasingly strict environmental regulations regarding waste discharge and VOC emissions. This green chemistry profile simplifies the permitting process for new manufacturing lines and reduces the liability associated with hazardous waste management. Consequently, this technology supports the commercial scale-up of complex pharmaceutical intermediates while adhering to corporate sustainability goals and regulatory compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and technical disclosures within the patent documentation to ensure accuracy and relevance. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this route into their existing pipelines.
Q: What are the primary advantages of using diaryl ethers over traditional aryl halides in this synthesis?
A: Diaryl ethers serve as abundant, renewable, and cost-effective aryl sources compared to expensive and often unstable aryl iodides or boronic acids. This method leverages the cleavage of the C-O bond in diaryl ethers, significantly reducing raw material costs and expanding the scope of accessible substituents for pharmaceutical intermediate manufacturing.
Q: Which catalyst system provides the optimal yield for this transformation?
A: Experimental data indicates that N-heterocyclic carbene (NHC) palladium chloride complexes provide superior catalytic activity compared to standard palladium chloride or phosphine-based catalysts. When combined with ionic liquid solvents, this system achieves yields up to 80%, demonstrating robust efficiency for complex molecule construction.
Q: Is this process scalable for industrial production of API intermediates?
A: Yes, the process utilizes mild reaction conditions (100-150°C) and readily available solvents, making it highly amenable to scale-up. The strong functional group tolerance allows for the synthesis of diverse derivatives without requiring extensive protection-deprotection strategies, streamlining the supply chain for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Aryl Isoxazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed methodology for the production of high-value isoxazole scaffolds used in next-generation therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 4-aryl isoxazole derivatives meets the exacting standards of the global pharmaceutical industry. We are committed to delivering not just a product, but a comprehensive supply solution that mitigates risk and accelerates your drug development timeline.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can quantify the economic benefits of switching to this diaryl ether-based route for your specific target molecules. We encourage potential partners to contact us immediately to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your supply chain.
