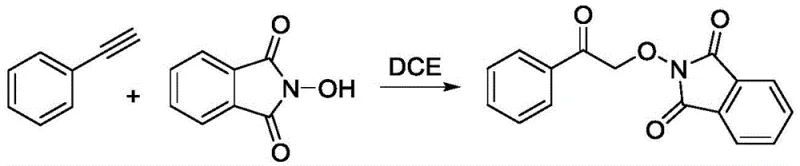
Advanced Catalyst-Free Synthesis of 2-(2-Oxo-2-Phenethoxy)Isobenzyl-1,3-Dione for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high purity with economic efficiency. Patent CN111606876A introduces a groundbreaking methodology for the preparation of 2-(2-oxo-2-phenethoxy)isobenzyl-1,3-dione, a valuable intermediate in organic synthesis. This innovation distinguishes itself by employing a catalyst-free oxidative alkylation strategy, utilizing molecular oxygen as the terminal oxidant to drive the transformation of phenylacetylene and N-hydroxyphthalimide. Unlike traditional methods that rely heavily on transition metal complexes, this approach operates under remarkably mild conditions, typically between 25°C and 100°C, thereby reducing energy consumption and operational complexity. For R&D directors and process chemists, this represents a significant leap forward in green chemistry, offering a pathway to high-purity products without the burden of residual metal contamination. The strategic implementation of this technology positions manufacturers to meet increasingly stringent regulatory standards regarding impurity profiles while simultaneously optimizing production costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex oxidative alkylation products has been plagued by the necessity for transition metal catalysts such as copper, palladium, or iron complexes. These conventional methodologies often require rigorous exclusion of air and moisture, demanding specialized equipment and inert atmosphere handling that drives up capital expenditure and operational overhead. Furthermore, the use of stoichiometric oxidants like peroxides or hypervalent iodine reagents generates substantial quantities of hazardous waste, creating significant disposal challenges and environmental liabilities for manufacturing facilities. From a quality control perspective, the presence of metal residues in the final active pharmaceutical ingredient (API) or intermediate is a critical failure point, necessitating expensive and time-consuming purification steps such as scavenging or recrystallization. These legacy processes frequently suffer from poor atom economy and limited scalability, making them ill-suited for the demands of modern commercial production where consistency and cost-efficiency are paramount.
The Novel Approach
In stark contrast, the methodology outlined in patent CN111606876A leverages the inherent reactivity of alkynes under an oxygen atmosphere to achieve oxidative alkylation without any external catalyst or additive. This metal-free paradigm shift eliminates the risk of heavy metal contamination at the source, fundamentally simplifying the downstream purification workflow and ensuring a cleaner impurity profile for the final product. By utilizing molecular oxygen, an abundant and inexpensive reagent, the process achieves excellent atom economy and drastically reduces the chemical footprint associated with the synthesis. The reaction demonstrates exceptional selectivity, yielding a single major product rather than a complex mixture of regioisomers or over-oxidized byproducts, which is a common pitfall in radical-mediated transformations. This streamlined approach not only accelerates the development timeline for new drug candidates but also provides a robust platform for the reliable pharmaceutical intermediate supplier seeking to optimize their manufacturing portfolio for long-term sustainability.
Mechanistic Insights into Oxygen-Mediated Oxidative Alkylation
The core of this innovative synthesis lies in the generation of reactive radical species through the interaction of N-hydroxyphthalimide (NHPI) and molecular oxygen. Under the thermal conditions provided by the oil bath, NHPI undergoes homolytic cleavage or hydrogen abstraction to generate the phthalimide N-oxyl (PINO) radical, a potent hydrogen abstractor. This radical species initiates the reaction cascade by abstracting a hydrogen atom or interacting with the alkyne substrate, phenylacetylene, to form a carbon-centered radical intermediate. The presence of oxygen is critical, as it traps these carbon-centered radicals to form peroxy radicals, which subsequently rearrange or react further to install the oxygen functionality observed in the final 2-(2-oxo-2-phenethoxy)isobenzyl-1,3-dione structure. Understanding this radical chain mechanism is vital for process optimization, as factors such as oxygen flow rate and stirring efficiency directly influence the concentration of dissolved oxygen and, consequently, the reaction kinetics. The absence of metal catalysts suggests that the redox potential is managed entirely through the organic radical cycle, offering a unique window into metal-free oxidation chemistry that is both elegant and highly effective for industrial applications.
From an impurity control standpoint, the simplicity of the mechanistic pathway contributes significantly to the high purity of the isolated product. Traditional metal-catalyzed reactions often suffer from side reactions mediated by the metal center, such as beta-hydride elimination or unwanted coupling events, which generate difficult-to-remove impurities. In this catalyst-free system, the reaction trajectory is more defined, primarily governed by the stability of the radical intermediates and the availability of oxygen. The use of 1,2-dichloroethane (DCE) as a solvent further stabilizes the reaction environment, providing a medium that supports the radical propagation steps without interfering with the substrate. For quality assurance teams, this translates to a more predictable impurity spectrum, facilitating easier method development for HPLC analysis and ensuring that the final material meets the stringent specifications required for high-purity pharmaceutical intermediates. The ability to control the reaction outcome through temperature and oxygen pressure, rather than complex ligand tuning, offers process engineers greater flexibility in scaling the reaction while maintaining consistent product quality.
How to Synthesize 2-(2-Oxo-2-Phenethoxy)Isobenzyl-1,3-Dione Efficiently
The practical execution of this synthesis is designed for ease of operation, requiring standard laboratory or pilot plant equipment without the need for specialized high-pressure reactors or gloveboxes. The process begins by charging the reactor with the solid N-hydroxyphthalimide and liquid phenylacetylene, followed by the addition of the solvent 1,2-dichloroethane. Maintaining an oxygen atmosphere throughout the reaction is crucial, achieved by continuous sparging or maintaining a positive pressure of oxygen in the headspace. The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined below to ensure reproducibility and safety during scale-up operations.
- Combine phenylacetylene and N-hydroxyphthalimide in a reactor with 1,2-dichloroethane solvent under an oxygen atmosphere.
- Heat the mixture in an oil bath between 25°C and 100°C with magnetic stirring for 8 to 16 hours to complete the reaction.
- Extract the reaction mixture with dichloromethane and water, then purify the crude product via column chromatography using ethyl acetate and petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalyst-free technology offers profound strategic benefits that extend beyond simple reagent costs. The elimination of precious metal catalysts removes a volatile cost component from the bill of materials, shielding the manufacturing budget from fluctuations in the global markets for metals like palladium or rhodium. Furthermore, the simplified workflow reduces the number of unit operations required, particularly in the purification stage, leading to significant reductions in solvent consumption and processing time. This efficiency gain directly translates to increased throughput capacity within existing facilities, allowing manufacturers to respond more agilely to market demand without substantial capital investment in new infrastructure. The reliance on commodity chemicals like oxygen and DCE ensures a stable and secure supply chain, minimizing the risk of production delays caused by the shortage of specialized reagents.
- Cost Reduction in Manufacturing: The most immediate financial impact arises from the complete removal of transition metal catalysts and their associated ligands, which are often among the most expensive inputs in fine chemical synthesis. Without the need for metal scavengers or extensive filtration steps to remove catalyst residues, the overall processing cost is drastically simplified, leading to substantial cost savings in the final product pricing. Additionally, the use of molecular oxygen as the oxidant replaces costly stoichiometric oxidizing agents, further driving down the raw material expenses while simultaneously reducing the volume of chemical waste generated. This lean manufacturing approach allows for a more competitive pricing structure, enabling the reliable agrochemical intermediate supplier or pharma partner to offer better value to their downstream customers while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of universally available starting materials and reagents. Phenylacetylene and N-hydroxyphthalimide are commercially accessible bulk chemicals, reducing the dependency on single-source suppliers for exotic catalysts. The robustness of the reaction conditions, which tolerate a range of temperatures and do not require strict anhydrous environments, minimizes the risk of batch failures due to minor deviations in raw material quality or environmental conditions. This reliability ensures consistent delivery schedules and reduces lead time for high-purity pharmaceutical intermediates, a critical factor for clients managing just-in-time inventory systems for API production. The simplified logistics of handling non-hazardous oxidants like oxygen compared to explosive peroxides also streamlines transportation and storage requirements.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage levels is inherently safer and more straightforward due to the absence of exothermic risks associated with strong chemical oxidants or pyrophoric catalysts. The mild thermal profile allows for the use of standard glass-lined or stainless steel reactors without the need for specialized cooling systems to manage runaway reactions. From an environmental perspective, the reduction in heavy metal waste aligns perfectly with global sustainability goals and tightening environmental regulations, reducing the burden on wastewater treatment facilities. This green chemistry credential not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a responsible partner in the sustainable production of complex polymer additives or electronic chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative alkylation technology. These insights are derived directly from the patent data and practical experience in scaling similar metal-free processes, providing clarity for stakeholders evaluating this route for their own production needs. Understanding these nuances is essential for making informed decisions about process adoption and supply chain integration.
Q: What are the primary advantages of this metal-free oxidative alkylation method?
A: The primary advantage is the elimination of transition metal catalysts, which removes the need for expensive and complex heavy metal removal steps often required in pharmaceutical manufacturing. Additionally, using molecular oxygen as the sole oxidant significantly reduces reagent costs and environmental waste compared to traditional stoichiometric oxidants.
Q: What are the optimal reaction conditions described in the patent?
A: The patent specifies mild reaction conditions ranging from 25°C to 100°C in 1,2-dichloroethane (DCE). The reaction proceeds under an oxygen atmosphere with magnetic stirring for a duration of 8 to 16 hours, ensuring high selectivity and yield without the need for harsh additives.
Q: How does this process impact supply chain stability for API intermediates?
A: By utilizing readily available starting materials like phenylacetylene and N-hydroxyphthalimide alongside common solvents, the process minimizes reliance on specialized or scarce catalytic systems. This simplicity enhances supply chain continuity and allows for easier commercial scale-up from laboratory to industrial production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2-Oxo-2-Phenethoxy)Isobenzyl-1,3-Dione Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of catalyst-free synthetic routes in modernizing the production of high-value intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering materials that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging technologies like the one described in CN111606876A, we can offer our partners a superior product profile with minimized impurity risks and optimized cost structures.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific project requirements. Whether you are looking for cost reduction in electronic chemical manufacturing or need a dependable source for complex intermediates, we are ready to provide a Customized Cost-Saving Analysis that highlights the specific economic benefits for your operation. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced capabilities can support your supply chain goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
