Advanced One-Pot Synthesis of 2-Amino-1,2-Dihydroisoquinolines for Scalable Pharmaceutical Manufacturing
Introduction to Novel Isoquinoline Synthesis Technology
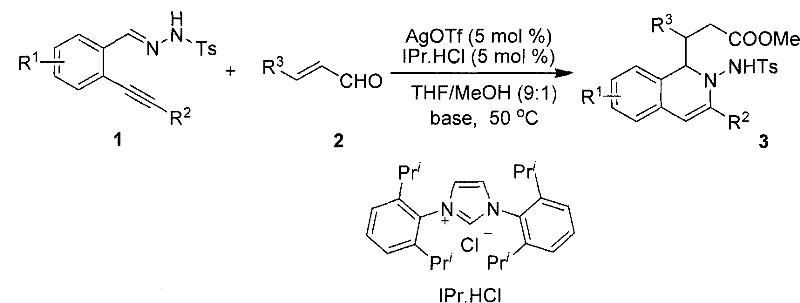
The pharmaceutical industry continuously seeks efficient routes to bioactive heterocyclic scaffolds, and patent CN101786983A presents a significant breakthrough in the synthesis of 2-amino-1,2-dihydroisoquinoline compounds. This intellectual property details a highly efficient, one-pot tandem reaction strategy that merges silver-catalyzed intramolecular cyclization with N-heterocyclic carbene (NHC) mediated polarity inversion addition. By utilizing o-alkynyl benzylhydrazones and alpha,beta-unsaturated aldehydes as starting materials, this methodology bypasses the tedious multi-step sequences traditionally associated with isoquinoline alkaloid construction. The process operates under remarkably mild conditions, typically at 50°C in a THF/MeOH solvent system, achieving reaction efficiencies as high as 90% with exceptional atomic economy. For R&D directors and process chemists, this represents a paradigm shift towards greener, more streamlined manufacturing of complex nitrogen-containing heterocycles that serve as critical intermediates in drug discovery programs targeting various physiological pathways.
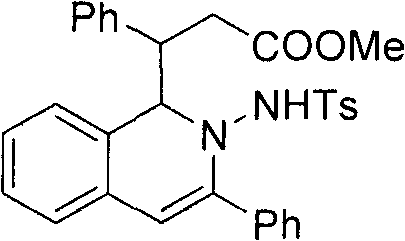
To illustrate the structural diversity accessible through this platform, consider the specific derivative shown below, which incorporates phenyl and ester functionalities essential for downstream biological evaluation. The ability to construct such complex molecular architectures in a single operational step significantly reduces the time-to-market for new therapeutic candidates. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic nuances allows us to offer superior process optimization services, ensuring that clients receive high-purity materials generated through the most advanced synthetic methodologies available today. The versatility of this reaction extends to various substituted substrates, making it a robust tool for generating diverse chemical libraries for high-throughput screening.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2-dihydroisoquinoline skeleton has relied heavily on classical named reactions such as the Bischler-Napieralski, Pictet-Spengler, or Pomeranz-Fritsch methodologies. These traditional pathways are often plagued by significant operational drawbacks that hinder their efficiency in modern industrial settings. For instance, the Bischler-Napieralski reaction typically necessitates the use of strong dehydrating agents like phosphorus oxychloride or polyphosphoric acid under rigorous thermal conditions, which can lead to substrate decomposition and the formation of difficult-to-remove impurities. Furthermore, these conventional routes frequently involve linear synthetic sequences requiring isolation and purification of unstable intermediates, resulting in lower overall yields and increased waste generation. The substrate scope is also frequently limited, particularly when dealing with sensitive functional groups that cannot withstand the harsh acidic or oxidative environments inherent to these older protocols. Consequently, procurement managers often face higher costs and longer lead times due to the inefficiency and complexity of these legacy manufacturing processes.
The Novel Approach
In stark contrast, the technology disclosed in CN101786983A introduces a convergent, catalytic approach that fundamentally redefines the synthesis of these valuable heterocycles. By employing a dual-catalytic system comprising silver trifluoromethanesulfonate (AgOTf) and an N-heterocyclic carbene precursor (IPr.HCl), the reaction achieves a seamless tandem cyclization-addition sequence in a single pot. This novel strategy eliminates the need for stoichiometric amounts of hazardous reagents and avoids the extreme temperatures associated with classical methods, operating instead at a moderate 50°C. The use of a THF/MeOH (9:1) solvent mixture provides an optimal medium for solubility and reactivity, facilitating smooth conversion of starting materials into the desired 2-amino-1,2-dihydroisoquinoline products. This approach not only simplifies the operational workflow by removing intermediate isolation steps but also dramatically improves the atom economy, approaching 100% in ideal scenarios. For supply chain heads, this translates to a more predictable and scalable process that minimizes raw material consumption and waste disposal costs, aligning perfectly with modern green chemistry principles and sustainability goals.
Mechanistic Insights into Ag-NHC Dual Catalytic Cyclization
The success of this transformation lies in the synergistic interplay between the silver Lewis acid and the nucleophilic N-heterocyclic carbene organocatalyst. The mechanism initiates with the activation of the alkyne moiety in the o-alkynyl benzylhydrazone by the silver cation, which facilitates an intramolecular nucleophilic attack by the hydrazone nitrogen to form a cyclic silver-carbene intermediate or a related metallacycle. Concurrently, the NHC catalyst, generated in situ from the imidazolium salt and base, engages with the alpha,beta-unsaturated aldehyde to form a Breslow-type intermediate or an extended enolate species via polarity inversion (umpolung). This activation renders the beta-carbon of the aldehyde nucleophilic, allowing it to attack the electrophilic cyclic intermediate generated by the silver catalyst. This cascade effectively stitches together the two distinct building blocks into the final dihydroisoquinoline framework with high stereochemical control. Understanding this dual-activation mode is crucial for R&D teams aiming to further optimize reaction parameters or adapt the chemistry to novel substrate classes, ensuring maximum efficiency and minimal byproduct formation during scale-up operations.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in maintaining high product purity. Unlike acid-catalyzed cyclizations that often promote polymerization or rearrangement side reactions, this Ag-NHC system operates under neutral to slightly basic conditions, preserving sensitive functional groups such as esters and halides. The specific choice of base, such as cesium carbonate or potassium phosphate, is critical for generating the active carbene species without degrading the hydrazone substrate. The patent data indicates that the reaction proceeds cleanly, as evidenced by TLC monitoring where starting materials disappear completely within 12 to 15 hours. This clean reaction profile simplifies downstream processing, as the crude mixture contains fewer tarry byproducts, allowing for straightforward purification via silica gel chromatography. For quality control laboratories, this means easier validation of purity specifications and reduced risk of genotoxic impurities often associated with harsh synthetic conditions, thereby enhancing the safety profile of the final pharmaceutical intermediate.
How to Synthesize 2-Amino-1,2-Dihydroisoquinoline Efficiently
Implementing this advanced synthetic route requires precise control over reaction parameters to replicate the high yields reported in the patent literature. The process begins with the dissolution of the o-alkynyl benzylhydrazone and the alpha,beta-unsaturated aldehyde in a degassed mixture of tetrahydrofuran and methanol. The sequential addition of the silver catalyst, the NHC precursor, and the inorganic base must be performed under an inert nitrogen atmosphere to prevent oxidation of the sensitive carbene species. Maintaining the temperature at 50°C ensures optimal kinetic energy for the tandem cyclization without triggering thermal decomposition. Detailed standardized operating procedures regarding reagent grades, solvent drying, and workup protocols are essential for consistent batch-to-batch reproducibility. The following guide outlines the critical steps for executing this transformation effectively in a laboratory or pilot plant setting.
- Dissolve o-alkynyl benzylhydrazone and alpha,beta-unsaturated aldehyde in THF/MeOH (9: 1) under nitrogen.
- Add AgOTF (5 mol%), IPr.HCl (5 mol%), and base (e.g., Cs2CO3) sequentially.
- Heat at 50°C for 12-15 hours, then quench, extract, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic technology offers substantial strategic advantages over traditional manufacturing routes. The primary benefit stems from the drastic simplification of the synthetic sequence; by consolidating multiple chemical transformations into a single one-pot operation, the process significantly reduces the consumption of solvents, reagents, and labor hours. This consolidation directly correlates to a reduction in the cost of goods sold (COGS), as fewer unit operations mean lower utility costs and reduced equipment occupancy time. Furthermore, the high atom economy of the reaction ensures that the majority of the mass of the starting materials is incorporated into the final product, minimizing waste generation and the associated environmental compliance costs. This efficiency makes the process highly attractive for large-scale production where margin optimization is critical for competitiveness in the global pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric reagents and the reduction of synthetic steps lead to significant cost savings. By avoiding the use of harsh acids and high-temperature conditions, the process reduces energy consumption and extends the lifespan of reactor vessels and ancillary equipment. The use of commercially available and relatively inexpensive starting materials, such as substituted cinnamaldehydes and hydrazones, further enhances the economic viability of the route. Additionally, the high yields reported (up to 90%) minimize the loss of valuable intermediates, ensuring that raw material investments are maximized. This economic efficiency allows suppliers to offer more competitive pricing structures to their clients while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The robustness of this reaction protocol contributes to a more resilient supply chain. The tolerance of the catalytic system to various functional groups means that a wide range of substrate derivatives can be sourced or synthesized without needing custom process development for each variant. This flexibility reduces the risk of supply disruptions caused by the unavailability of specific niche precursors. Moreover, the mild reaction conditions reduce the safety hazards associated with manufacturing, lowering insurance premiums and regulatory hurdles. The simplicity of the workup procedure, involving standard extraction and chromatography, ensures that production timelines are predictable and short, enabling faster turnaround times for customer orders and improving overall service levels.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or ton scale is facilitated by the homogeneous nature of the catalytic system and the absence of exothermic hazards typical of strong acid reactions. The use of common organic solvents like THF and methanol simplifies solvent recovery and recycling programs, aligning with corporate sustainability initiatives. The high purity of the crude product reduces the burden on waste treatment facilities, as fewer toxic byproducts require disposal. This environmental compatibility is increasingly important for meeting stringent international regulatory standards and maintaining a positive corporate social responsibility profile. The ability to produce high-quality intermediates with a smaller environmental footprint positions manufacturers as preferred partners for eco-conscious pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating this method into their existing supply chains or R&D pipelines. The clarity provided here aims to mitigate risks and accelerate the adoption of this innovative chemical process.
Q: What are the advantages of this Ag-NHC catalytic system over traditional methods?
A: Unlike traditional Bischler-Napieralski or Pictet-Spengler reactions which often require harsh acidic conditions and multi-step sequences, this method utilizes a mild one-pot tandem reaction at 50°C with high atom economy (100%) and yields up to 90%.
Q: Is this process suitable for large-scale production of pharmaceutical intermediates?
A: Yes, the patent explicitly states the method is suitable for large-scale preparation due to simple operation, mild conditions, and easy purification via standard silica gel column chromatography.
Q: What is the substrate scope for this reaction?
A: The reaction tolerates a wide range of substituents including electron-donating (Me, OMe) and electron-withdrawing groups (F, Cl, CF3, Br) on both the alkyne and aldehyde components, allowing for diverse library synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-1,2-Dihydroisoquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Ag-NHC catalytic system described in CN101786983A for the production of high-value pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, leveraging our rigorous QC labs to verify every batch against the highest industry standards. Our infrastructure is designed to handle complex catalytic reactions safely and effectively, guaranteeing a consistent supply of critical building blocks for your drug development programs.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient route can optimize your budget. Please contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to cutting-edge chemical innovation combined with the reliability of a seasoned manufacturing partner, ensuring your supply chain remains robust and competitive in the evolving pharmaceutical landscape.
