Advanced Synthetic Route for 5-Alkylresorcinol: Enhancing Purity and Commercial Scalability for Global Pharma
Advanced Synthetic Route for 5-Alkylresorcinol: Enhancing Purity and Commercial Scalability for Global Pharma
The pharmaceutical and fine chemical industries are constantly seeking robust, cost-effective, and environmentally sustainable pathways for producing critical intermediates. Patent CN101591225A introduces a groundbreaking synthetic methodology for the preparation of 5-alkylresorcinol, a versatile compound serving as a pivotal building block for various bioactive molecules, including potential treatments for immune deficiencies and malignant tumors. This innovative approach leverages a strategic combination of alpha,beta-unsaturated ketones and malonate diesters, initiating a cascade of reactions that culminate in the formation of the target resorcinol derivative. By shifting away from hazardous and expensive traditional reagents, this technology offers a compelling value proposition for reliable pharmaceutical intermediate supplier networks aiming to optimize their supply chains. The process is characterized by its operational simplicity, utilizing common technical grade chemicals that significantly lower the barrier to entry for commercial production while maintaining high standards of chemical integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-alkylresorcinol has been plagued by significant technical and economic hurdles that hinder efficient cost reduction in API manufacturing. Traditional routes, such as those described in US Patent No. 4,249,027, rely on the condensation of unsaturated carboxylic acid esters with 3-ketoglutaric acid diesters. These pathways are notoriously tedious, involving multiple complex steps that result in lower overall productivity and yield. Furthermore, the raw materials required for these legacy methods are often non-commodity chemicals, making them difficult to source reliably and driving up procurement costs. Another prevalent method involves the use of trifluoroacetic anhydride for sulfonation and isomerization, which presents severe safety and environmental challenges due to the reagent's high reactivity and exorbitant cost. Additionally, high-temperature alkali fusion processes, while shorter in step count, suffer from difficult condition control and poor yields during industrial scaling, creating bottlenecks for commercial scale-up of complex phenolic compounds.
The Novel Approach
In stark contrast, the methodology disclosed in CN101591225A revolutionizes the production landscape by introducing a streamlined, three-stage sequence that prioritizes efficiency and safety. This novel approach utilizes alpha,beta-unsaturated ketones and malonate diesters as primary feedstocks, which are abundant, inexpensive, and easily accessible in the global chemical market. The reaction conditions are remarkably mild, typically proceeding under alkaline conditions at moderate temperatures, thereby eliminating the need for extreme thermal inputs or corrosive environments. This gentleness not only preserves the integrity of the molecular structure but also simplifies the engineering requirements for reactor systems. The process flow is succinct, moving directly from the initial condensation to bromination, decarboxylation, and aromatization without the need for isolating unstable intermediates, which drastically reduces processing time and waste generation. This represents a paradigm shift towards reducing lead time for high-purity resorcinol derivatives while adhering to modern green chemistry principles.
Mechanistic Insights into the Cascade Cyclization and Aromatization
The core of this synthetic innovation lies in a sophisticated cascade reaction mechanism that efficiently constructs the resorcinol core. The process begins with a Michael-type addition where the malonate diester acts as a nucleophile, attacking the beta-carbon of the alpha,beta-unsaturated ketone under basic catalysis. This initial step is crucial for establishing the carbon skeleton of the six-membered ring. Following the addition, an intramolecular cyclization occurs, facilitated by the enolate chemistry inherent to the beta-dicarbonyl system, forming a substituted cyclohexenone intermediate (Formula IV). This intermediate is structurally poised for the subsequent transformation, possessing the necessary functionality for oxidative aromatization. The precision of this cyclization step is vital for controlling regioselectivity, ensuring that the alkyl group ends up in the desired 5-position on the final aromatic ring, which is critical for the biological activity of downstream pharmaceutical products.
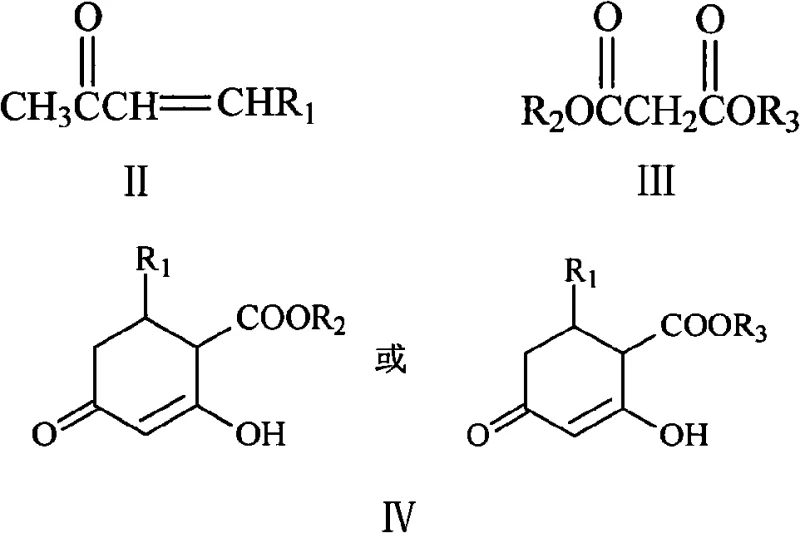
The final transformation involves a tandem bromination-decarboxylation-aromatization sequence that converts the cyclic intermediate into the target 5-alkylresorcinol. Bromination introduces a leaving group that facilitates the subsequent elimination reactions. Upon heating, typically in a polar aprotic solvent like DMF, the molecule undergoes decarboxylation, losing carbon dioxide to drive the equilibrium forward. Simultaneously, the system aromatizes to regain the stability of the benzene ring, resulting in the formation of the diphenolic structure. This mechanism is highly advantageous because it avoids the use of heavy metal catalysts or harsh oxidants that could introduce difficult-to-remove trace impurities. The ability to perform these steps in a telescoped manner, often without isolating the brominated intermediate, minimizes material loss and exposure to potentially hazardous reagents, thereby enhancing the overall safety profile and purity of the final high-purity 5-alkylresorcinol product.
How to Synthesize 5-Alkylresorcinol Efficiently
Implementing this synthetic route requires careful attention to reaction parameters, particularly regarding the choice of base and the control of temperature during the aromatization phase. The patent outlines a robust protocol where the initial condensation of acetone and aldehyde can be performed with various bases such as sodium hydroxide or potassium carbonate, offering flexibility in process design. The subsequent cyclization with dimethyl malonate is optimally conducted in methanol with sodium methoxide, ensuring complete conversion to the cyclic intermediate. For the final aromatization, the use of liquid bromine followed by heating in DMF under an inert atmosphere is key to achieving high yields. Detailed standard operating procedures regarding stoichiometry, addition rates, and workup protocols are essential for reproducibility. The detailed standardized synthesis steps are provided in the guide below.
- Condense acetone with the corresponding aldehyde under alkaline conditions to form the alpha,beta-unsaturated ketone intermediate.
- React the unsaturated ketone with a malonate diester in the presence of a base (e.g., sodium methoxide) to form the cyclic intermediate via Michael addition and cyclization.
- Subject the cyclic intermediate to bromination, followed by heating to induce decarboxylation and aromatization, yielding the final 5-alkylresorcinol product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology offers profound advantages that directly impact the bottom line and supply chain resilience. By replacing exotic and costly reagents like trifluoroacetic anhydride with commodity chemicals such as acetone, simple aldehydes, and dimethyl malonate, manufacturers can achieve substantial cost savings in raw material procurement. These feedstocks are produced on a massive global scale, ensuring consistent availability and shielding the supply chain from the volatility associated with niche specialty chemicals. Furthermore, the elimination of complex purification steps and the ability to recycle solvents like DMF and toluene contribute to a leaner manufacturing process. This efficiency translates into a more competitive pricing structure for the final intermediate, allowing downstream pharmaceutical companies to optimize their own production costs without compromising on quality or regulatory compliance.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the fundamental shift to low-cost, high-volume starting materials. Unlike traditional methods that rely on expensive activating agents or precious metal catalysts, this route utilizes basic inorganic bases and organic esters that are priced at commodity levels. The simplified workflow reduces energy consumption by avoiding high-temperature fusion or cryogenic conditions, leading to lower utility costs per kilogram of product. Additionally, the high total yield reported in the embodiments means less raw material is wasted, maximizing the return on investment for every batch produced. These factors combine to create a highly cost-effective manufacturing model that supports long-term profitability.
- Enhanced Supply Chain Reliability: Supply chain continuity is significantly bolstered by the use of universally available reagents. Acetone and various aliphatic aldehydes are staples of the chemical industry, sourced from multiple suppliers worldwide, which mitigates the risk of single-source dependency. The robustness of the reaction conditions, which tolerate a range of temperatures and do not require ultra-dry or oxygen-free environments for the initial steps, further enhances operational reliability. This flexibility allows for production in diverse geographic locations, reducing logistics lead times and enabling regional manufacturing hubs to serve local markets more effectively. Consequently, procurement teams can negotiate better terms and secure more stable supply agreements.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of hazardous unit operations. The mild reaction conditions reduce the stress on equipment, extending the lifespan of reactors and lowering maintenance costs. From an environmental standpoint, the process generates fewer toxic byproducts compared to alkali fusion or heavy metal-catalyzed routes. The ability to treat and recycle solvents aligns with increasingly stringent global environmental regulations, reducing the burden of waste disposal and potential liability. This sustainability profile not only ensures regulatory compliance but also enhances the corporate social responsibility standing of the manufacturing entity, making it a preferred partner for eco-conscious pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the feasibility and benefits of the process. Understanding these details is crucial for R&D teams evaluating the technology for adoption and for procurement officers assessing the risk-reward profile of switching to this new supply source. The information below highlights the practical aspects of the chemistry and its implications for industrial application.
Q: What are the primary advantages of this new synthetic method over traditional routes?
A: This method utilizes cheap and readily available raw materials like acetone and aldehydes, avoiding expensive reagents like trifluoroacetic anhydride. It features mild reaction conditions, simple operation, and a higher total yield compared to conventional alkali fusion or Wittig reaction pathways.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial suitability. It avoids harsh conditions like high-temperature alkali fusion and uses common technical grade chemicals. The solvents and catalysts employed are reusable, enhancing environmental friendliness and cost-efficiency for commercial scale-up.
Q: What is the purity profile of the resulting 5-alkylresorcinol?
A: The method produces high-purity 5-alkylresorcinol, often obtained as white crystals after recrystallization (e.g., from ethanol or cyclohexane). The specific purification steps, including pH adjustment and washing, effectively remove impurities, ensuring the material meets stringent specifications for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Alkylresorcinol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our team of expert chemists has thoroughly analyzed the potential of the synthetic route described in CN101591225A and is fully prepared to leverage this technology for your specific needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from pilot studies to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5-alkylresorcinol we deliver meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to unlock the full potential of this advanced synthesis method. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to discuss your project needs,索取 specific COA data, and review our comprehensive route feasibility assessments. Let us help you secure a reliable, cost-effective, and sustainable supply of 5-alkylresorcinol that drives your innovation forward.
