Advanced Chiral Synthesis of Octahydro-pyrrolo-pyridine Derivatives for Pharmaceutical Applications
Advanced Chiral Synthesis of Octahydro-pyrrolo-pyridine Derivatives for Pharmaceutical Applications
The pharmaceutical industry continuously demands high-purity chiral intermediates to support the development of next-generation therapeutics, particularly in the fields of metabolic disorders and neurological conditions. Patent CN113549063A discloses a robust and innovative preparation method for optically isomeric octahydro-2H-pyrrolo[3,4-c]pyridine-2-carboxylic acid tert-butyl ester, a critical scaffold found in potent autotaxin inhibitors and dopamine receptor antagonists. This technical insight report analyzes the novel synthetic pathway detailed in the patent, highlighting its superiority over conventional achiral routes. By leveraging specific catalytic hydrogenation conditions and precise functional group manipulations, this method addresses the longstanding challenge of obtaining high enantiomeric excess in fused bicyclic nitrogen heterocycles. For R&D directors and procurement specialists, understanding this chemistry is vital for securing a reliable supply chain of complex pharmaceutical intermediates that meet stringent regulatory standards for chirality and impurity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of octahydro-1H-pyrrolo[3,4-c]pyridine derivatives has been plagued by significant stereochemical challenges. As illustrated in prior art such as patent CN105085525A, traditional routes often rely on non-stereoselective reactions that yield racemic mixtures or achiral compounds like compound 15d. 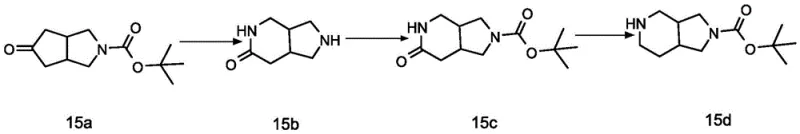 . These conventional methods typically involve harsh conditions, such as the use of sodium azide and methanesulfonic acid, which pose safety risks and complicate waste management. Furthermore, the inability to control the stereochemistry at the bridgehead carbons means that extensive and costly resolution steps are required post-synthesis, drastically reducing overall yield and increasing the cost of goods sold. The lack of a direct route to the specific (3ar, 7as) relative configuration limits the utility of these intermediates for drugs requiring precise spatial arrangement for receptor binding.
. These conventional methods typically involve harsh conditions, such as the use of sodium azide and methanesulfonic acid, which pose safety risks and complicate waste management. Furthermore, the inability to control the stereochemistry at the bridgehead carbons means that extensive and costly resolution steps are required post-synthesis, drastically reducing overall yield and increasing the cost of goods sold. The lack of a direct route to the specific (3ar, 7as) relative configuration limits the utility of these intermediates for drugs requiring precise spatial arrangement for receptor binding.
The Novel Approach
The methodology presented in CN113549063A offers a transformative solution by introducing a highly stereoselective sequence starting from readily available pyridine dicarboxylates. The core innovation lies in the controlled reduction and cyclization strategy that preserves the chiral information established in the early stages. 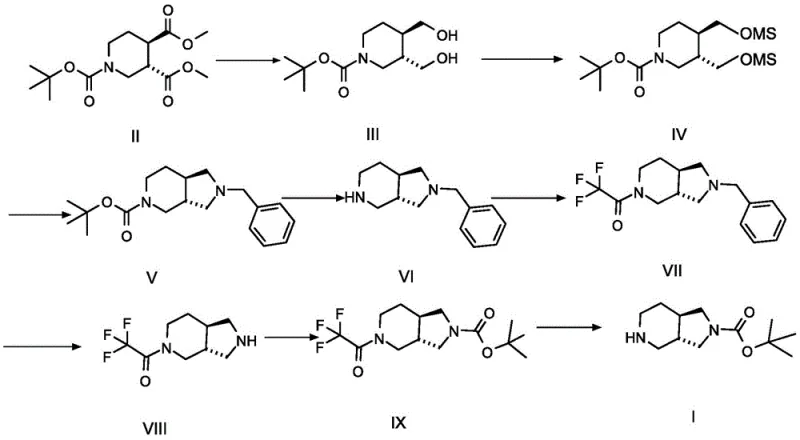 . Unlike previous attempts, this route utilizes a benzylamine-mediated cyclization that effectively constructs the pyrrolidine ring while maintaining the integrity of the piperidine moiety. The process flow from Compound II through to the final Compound I demonstrates a logical progression of functional group interconversions—reduction, mesylation, nucleophilic substitution, and protection—that are amenable to optimization. This approach not only bypasses the need for difficult chiral separations but also achieves an overall yield of approximately 27%, which is substantial for such a complex polycyclic system. For a reliable pharmaceutical intermediate supplier, adopting this route signifies a move towards more efficient and predictable manufacturing processes.
. Unlike previous attempts, this route utilizes a benzylamine-mediated cyclization that effectively constructs the pyrrolidine ring while maintaining the integrity of the piperidine moiety. The process flow from Compound II through to the final Compound I demonstrates a logical progression of functional group interconversions—reduction, mesylation, nucleophilic substitution, and protection—that are amenable to optimization. This approach not only bypasses the need for difficult chiral separations but also achieves an overall yield of approximately 27%, which is substantial for such a complex polycyclic system. For a reliable pharmaceutical intermediate supplier, adopting this route signifies a move towards more efficient and predictable manufacturing processes.
Mechanistic Insights into Stereoselective Hydrogenation and Cyclization
The success of this synthesis hinges on the precise control of stereochemistry during the formation of the saturated bicyclic core. The patent details two distinct pathways for generating the key chiral precursor, Compound II, from dimethyl pyridine-3,4-dicarboxylate (Compound X). 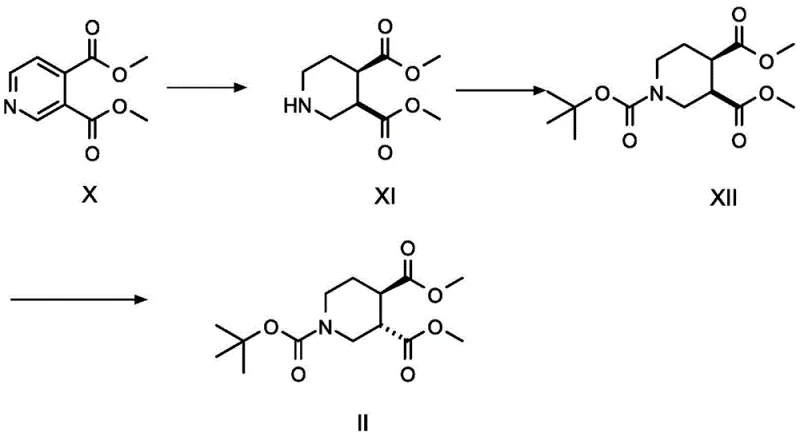 . In one embodiment, Compound X undergoes catalytic hydrogenation using platinum dioxide (PtO2) in the presence of an acid like hydrochloric acid. This condition favors the formation of the cis-product (Compound XI), which is subsequently protected and epimerized using sodium hydride to yield the thermodynamically more stable trans-isomer, Compound II. Alternatively, the use of palladium hydroxide under high pressure (6-8 MPa) can directly yield the trans-product (Compound XIII). This mechanistic flexibility allows manufacturers to tune the process based on available equipment and safety protocols. The subsequent reduction of the diester Compound II to the diol Compound III using lithium aluminum hydride or sodium borohydride is a critical step that must be carefully monitored to prevent over-reduction or side reactions, ensuring the hydroxyl groups are positioned correctly for the ensuing mesylation.
. In one embodiment, Compound X undergoes catalytic hydrogenation using platinum dioxide (PtO2) in the presence of an acid like hydrochloric acid. This condition favors the formation of the cis-product (Compound XI), which is subsequently protected and epimerized using sodium hydride to yield the thermodynamically more stable trans-isomer, Compound II. Alternatively, the use of palladium hydroxide under high pressure (6-8 MPa) can directly yield the trans-product (Compound XIII). This mechanistic flexibility allows manufacturers to tune the process based on available equipment and safety protocols. The subsequent reduction of the diester Compound II to the diol Compound III using lithium aluminum hydride or sodium borohydride is a critical step that must be carefully monitored to prevent over-reduction or side reactions, ensuring the hydroxyl groups are positioned correctly for the ensuing mesylation.
Impurity control is rigorously managed through the strategic use of the benzyl group. During the conversion of the dimesylate (Compound IV) to the cyclic amine (Compound V/VI), benzylamine acts as both a nucleophile and a protecting group. This dual function simplifies the purification landscape, as the benzyl group can be cleanly removed later via catalytic hydrogenolysis using palladium on carbon. The patent specifies that residual benzylamine can be scavenged by reacting with di-tert-butyl dicarbonate in the filtrate, a clever trick that prevents amine impurities from carrying over into the final product. Furthermore, the final deprotection step using base (such as potassium carbonate) to remove the trifluoroacetyl group is highly selective, leaving the tert-butyl carbamate (Boc) group intact. This orthogonality in protecting group chemistry is essential for maintaining high purity specifications, ensuring that the final API intermediate is free from structurally related impurities that could impact drug safety.
How to Synthesize Optically Isomeric Octahydro-pyrrolo-pyridine Efficiently
Implementing this synthesis requires a deep understanding of reaction kinetics and phase separation techniques. The process begins with the preparation of the chiral diester, followed by a reduction step that generates a sensitive diol intermediate. Operators must maintain strict temperature control, typically around 0°C, during the addition of reducing agents to manage exotherms. The subsequent mesylation and cyclization steps require anhydrous conditions and careful stoichiometry to maximize the formation of the bicyclic ring. Detailed standardized operating procedures for each transformation, including work-up and purification protocols, are essential for reproducibility. For a comprehensive guide on executing these reactions with maximum efficiency and safety, please refer to the step-by-step instructions provided below.
- Reduce the diester precursor (Compound II) using lithium aluminum hydride or sodium borohydride to generate the diol intermediate (Compound III).
- Activate the diol via mesylation with methanesulfonyl chloride to form Compound IV, followed by cyclization with benzylamine to create the pyrrolidine ring structure.
- Perform sequential deprotection, trifluoroacetylation, catalytic hydrogenation to remove the benzyl group, and final Boc-protection to yield the target chiral ester.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers significant strategic advantages for procurement managers and supply chain heads looking to optimize their sourcing of complex heterocyclic building blocks. The route is designed with scalability in mind, utilizing reagents and catalysts that are commercially available in bulk quantities. By eliminating the need for expensive chiral chromatography columns or enzymatic resolutions that are often required for similar scaffolds, the process inherently drives down the cost of manufacturing. The ability to produce the target molecule with high optical purity directly from the synthesis reduces the burden on quality control laboratories and minimizes the risk of batch rejection due to failed enantiomeric excess specifications. This reliability is crucial for maintaining continuous supply lines for downstream API production.
- Cost Reduction in Manufacturing: The synthetic pathway significantly lowers production costs by avoiding the use of precious metal catalysts in the final steps, relying instead on recyclable palladium on carbon for hydrogenolysis. The high yields reported in the experimental examples, such as the 96% yield in the reduction step and 95% in the mesylation, indicate a material-efficient process that minimizes raw material waste. Furthermore, the use of common solvents like THF, DCM, and methanol simplifies solvent recovery and recycling operations, contributing to substantial cost savings in utility and waste disposal expenses. The streamlined number of isolation steps also reduces labor hours and equipment occupancy time, enhancing overall plant throughput.
- Enhanced Supply Chain Reliability: The starting materials, specifically dimethyl pyridine-3,4-dicarboxylate, are commodity chemicals with a stable global supply, reducing the risk of raw material shortages. The robustness of the reaction conditions, which tolerate slight variations in temperature and pressure without compromising product quality, ensures consistent batch-to-batch performance. This stability is vital for long-term supply agreements, as it mitigates the risk of production delays caused by sensitive reaction parameters. Additionally, the intermediate compounds are generally stable oils or solids that can be stored or transported if a multi-site manufacturing strategy is employed, providing flexibility in logistics planning.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-kilogram pilot runs and eventually to ton-scale commercial production. The avoidance of highly toxic reagents like sodium azide, which was present in older routes, aligns with modern environmental, health, and safety (EHS) standards. The waste streams generated are primarily aqueous salts and organic solvents that can be treated using standard effluent treatment protocols. This compliance with green chemistry principles not only reduces the environmental footprint but also simplifies the regulatory approval process for the manufacturing site, ensuring uninterrupted operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this chiral intermediate. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these details helps stakeholders make informed decisions regarding the feasibility of integrating this intermediate into their drug development pipelines. For further technical discussions or custom synthesis requests, our team is available to provide deeper insights.
Q: How is stereochemical control achieved in this synthesis route?
A: Stereocontrol is primarily established during the initial hydrogenation of the pyridine dicarboxylate precursor using specific catalysts like PtO2 or Pd(OH)2 under acidic conditions, which favors the formation of the cis- or trans-isomers required for the subsequent chiral integrity of the octahydro-pyrrolo[3,4-c]pyridine core.
Q: What are the critical purification steps to ensure high optical purity?
A: The process utilizes strategic crystallization and column chromatography at key intermediates, particularly after the cyclization and acylation steps. The use of benzylamine as a temporary protecting and cyclizing group allows for effective removal of impurities before the final hydrogenolysis step.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the route avoids extremely hazardous reagents where possible and utilizes standard unit operations such as filtration, solvent exchange, and catalytic hydrogenation. The overall yield reported in examples exceeds 27%, indicating a viable pathway for kilogram-to-ton scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Octahydro-pyrrolo-pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the successful development of novel therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demanding volume requirements of global pharmaceutical partners. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation for chiral HPLC and NMR analysis. Our facility is designed to handle complex chemistries safely and efficiently, making us an ideal partner for the commercialization of this specific octahydro-pyrrolo-pyridine derivative.
We invite you to collaborate with us to leverage this advanced synthesis technology for your upcoming projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume needs and timeline. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your evaluation. Let us help you accelerate your drug development timeline with a supply chain partner dedicated to excellence and innovation.
